1122
Can. J. Chem. Vol. 83, 2005
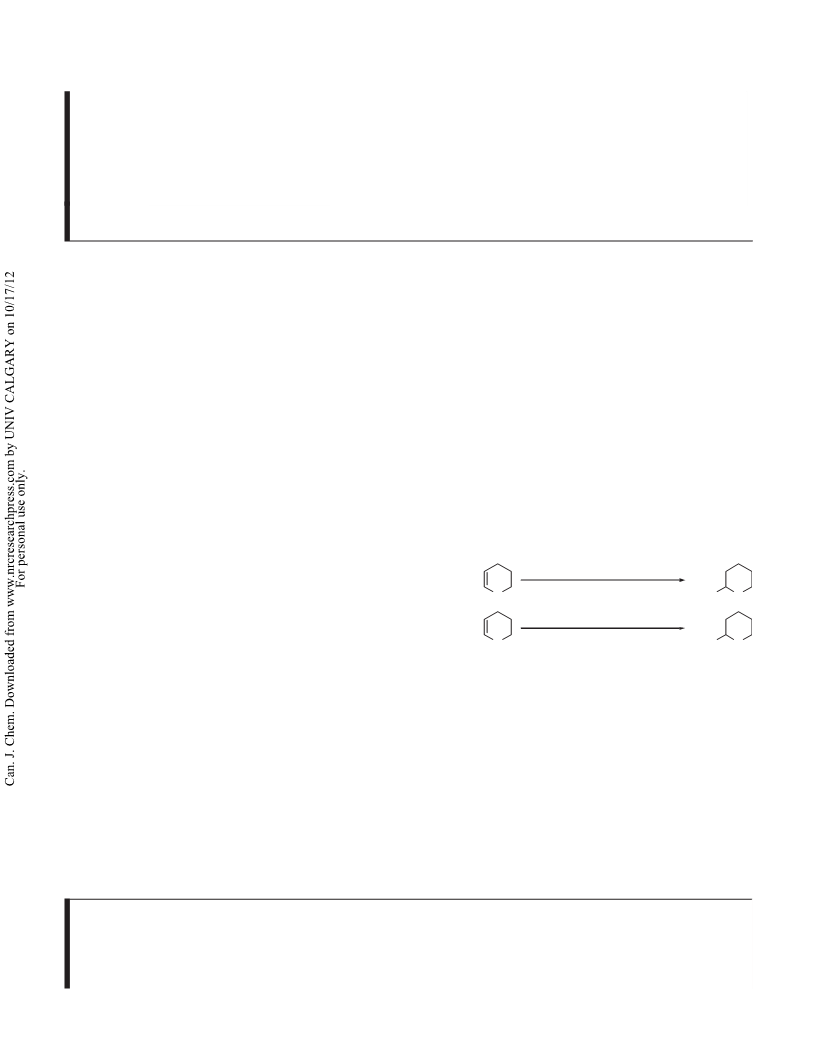
Table 2. Monotetrahydropyranylation of symmetrical diols
catalyzed by Fe2(SO4)3·xH2O in acetone.
Fig. 1. Yield vs. reaction time. Ethylene glycol (5.0 mmol), DHP
(0.45 mL, 5.0 mmol), and Fe2(SO4)3·xH2O (0.05 mmol) were
stirred at room temperature in the presence of acetone (1.5 mL).
MonoTHP ether (#), bisTHP ether (!), monoTHP ether (᭡)
without the addition of acetone.
Yield (%)a
Entry
Reaction time
(min)
Substrate
OH
HO
1
2
55
65
82
80
72b
80
OH
HO
HO
1
O H
3
60
60
aIsolated yield of monoTHP ether; all products were identified
1
by IR, H NMR, and MS.
bBisTHP ether (12%) was also obtained.
40
2
3
20
0
achieved in good yields rather than forming a complex with
Fe(III) or being oxidized by Fe(III). In general, 1 mol% of
Fe2(SO4)3·xH2O is enough for a fairly high yield. However,
with 0.4 mol% of Fe2(SO4)3·xH2O, the same yield was also
achieved under a longer reaction period (Table 1, entry 2).
The deprotection of the THP ether by Fe2(SO4)3·xH2O
was then explored. It was found that the deprotection of
THP ether with the presence of a catalytic amount of
Fe2(SO4)3·xH2O was realized successfully. For example, the
THP ether (1.0 mmol) of the compounds in Table 1 (entries
1, 5, 6, 8, and 10) were treated in anhydr. MeOH (3.0 mL)
with 10 mol% of Fe2(SO4)3·xH2O under reflux. Once the re-
action was completed (indicated by TLC), the corresponding
alcohols were finally isolated in 95%, 85%, 90%, 80%, and
90% yield, respectively. Therefore, a new reagent for both
THP protection and deprotection was found, which will en-
rich the toolbox for organic chemists.
Meanwhile, selective monotetrahydropyranylation of sym-
metrical diols was studied under similar reaction condi-
tions.4 The reaction was carried out by stirring diol
(5.0 mmol), DHP (0.45 mL, 5.0 mmol), and hydrated ferric
sulfate (0.025 g, 0.05 mmol) in the presence of dry acetone
(1.5 mL) and the process was monitored by TLC. After
completion, symmetrical diols were selectively protected as
their corresponding monotetrahydropyranylated products in
good yields (Table 2). It is worth noting that hydroquinone
(Table 2, entry 3) was selectively protected to give the
monotetrahydropyranylation product in 72% yield (along
with 12% of the diprotected product), which has seldom
been achieved in the literature so far.
0
10 20 30 40 50 60 70
Reaction time (min)
Table 3. The reusability of Fe2(SO4)3·xH2O catalyzed
tetrahydropyranylation.
Catalyst
Reaction
Yield
(%)a
Round
Substrate
recovered (g)
time (min)
60
80
1
2
3
4
C6H5CH2OH
C6H5CH2OH
C6H5CH2OH
C6H5CH2OH
0.025
0.024
0.022
0.021
95
92
92
89
110
150
aIsolated yield; all the products were identified by H NMR.
1
was found that the presence of a small amount of acetone fa-
cilitated the reaction procedure significantly (Fig. 1, curve
1). In fact, acetone is very helpful in making the two re-
agents (ethylene glycol and DHP) into a homogeneous mix-
ture, which helps the reaction proceed more smoothly than
in a heterogeneous state (Fig. 1, curve 2).
In the view of green chemistry, efficient recovery and re-
use of the catalyst are highly preferred. In our process,
Fe2(SO4)3·xH2O was easily recovered from the reaction mix-
ture by filtration and subsequent washing with dry acetone.
The recovered catalysts were ready to be used for the next
reaction cycle. For example, benzyl alcohol was protected as
its THP ether by using the recovered catalyst and the yields
were not significantly decreased even after three recovery
cycles (Table 3). However, an increased reaction period is
necessary to achieve comparable yields.
The effects of acetone and reaction time on the yield of
monoprotected diol were then investigated by taking ethyl-
ene glycol as the model substrate. As shown in Fig. 1, at the
primary stage, the reaction mainly gave the monoprotected
product. Even when the yield of monoether reached 82%
(Fig. 1, curve 1), the yield of diprotected diol was still lower
than 10% (Fig. 1, curve 3). However, the yield of diprotected
product increased remarkably after this stage, which implied
that optimization of the reaction time (55 min under stan-
dard reaction conditions) leads to a maximum amount of
monotetrahydropyranylation output. On the other hand, it
On the other hand, the development of new methods for
direct transformation from one protecting group to another
has been gaining importance in recent years. However, the
available method for direct transformation of THP ether to
the corresponding acetate is very limited (18). Two reported
4 Typical procedure for the monotetrahydropyranylation of symmetrical diols: ethylene glycol (5.0 mmol) and DHP (0.45 mL, 5.0 mmol) in
dry acetone (1.5 mL) were stirred for several minutes. Then, Fe2(SO4)3·xH2O (0.025 g, 0.05 mmol) was added. After 55 min, the reaction
was terminated by filtration. The filtrate was concentrated and purified through silica gel column chromatography (2:1 petroleum ether –
1
EtOAc) to give the monoTHP product (0.60 g, 82% yield). The product was identified by GC–MS and H NMR and the spectroscopic data
are comparable with the literature data.
© 2005 NRC Canada

 Li, Lingjun
Li, Lingjun
 Zhu, Lizhi
Zhu, Lizhi
 Zhang, Xinying
Zhang, Xinying
 Zhang, Guisheng
Zhang, Guisheng
 Qu, Guirong
Qu, Guirong