ꢀ
JalalꢀAlbadiꢀetꢀal.ꢀ/ꢀChineseꢀJournalꢀofꢀCatalysisꢀ36ꢀ(2015)ꢀ308–313ꢀ
309ꢀ
microscopyꢀ (SEM)ꢀ andꢀ high‐resolutionꢀ transmissionꢀ electronꢀ
microscopyꢀ (HRTEM)ꢀ techniquesꢀ usingꢀ aꢀ HITACHIꢀ S‐4160ꢀ
instrumentꢀ(Japan)ꢀandꢀaꢀJEOLꢀJEM‐2100ꢀ(200ꢀkV)ꢀmicroscopeꢀ
equippedꢀwithꢀanꢀenergyꢀdispersionꢀscanningꢀ(EDS)ꢀanalyticalꢀ
systemꢀ (Japan).ꢀ Aꢀ GBCꢀ 906AAꢀ flameꢀ atomicꢀ absorptionꢀ
spectrophotometerꢀ (Australia)ꢀ wasꢀ usedꢀ toꢀ determineꢀ theꢀ Cuꢀ
contentsꢀofꢀtheꢀcatalysts.ꢀ
CuO-ZnO nanocatalyst
Ac O, CH Cl , r.t.
ROAc
ROH
2
2
2
Schemeꢀ 1.ꢀ Chemoselectiveꢀ acetylationꢀ ofꢀ alcoholsꢀ catalyzedꢀ byꢀ a
CuO‐ZnOꢀnanocatalyst.ꢀ
conditionsꢀ[21–23].ꢀBecauseꢀofꢀtheirꢀattractiveꢀproperties,ꢀnanoꢀ
metalꢀoxidesꢀhaveꢀbeenꢀusedꢀextensivelyꢀasꢀsolidꢀcatalystsꢀinꢀaꢀ
varietyꢀofꢀorganicꢀreactions.ꢀ
2.3.ꢀ ꢀ Generalꢀprocedureꢀforꢀtheꢀacetylationꢀofꢀalcoholsꢀ
Weꢀ previouslyꢀ reportedꢀ theꢀ preparationꢀ ofꢀ severalꢀ
nanocatalystsꢀ andꢀ anꢀ evaluationꢀ ofꢀ theirꢀ catalyticꢀ activityꢀ
towardsꢀ aꢀ wideꢀ varietyꢀ ofꢀ organicꢀ reactionsꢀ [24–29].ꢀ Inꢀ
continuationꢀ ofꢀ ourꢀ researchꢀ inꢀ thisꢀ area,ꢀ weꢀ hereinꢀ wishꢀ toꢀ
reportꢀ theꢀ preparationꢀ ofꢀ aꢀ CuO‐ZnOꢀ nanocatalyst,ꢀ andꢀ itsꢀ
applicationꢀforꢀtheꢀchemoselectiveꢀacetylationꢀofꢀalcoholsꢀwithꢀ
aceticꢀanhydrideꢀatꢀroomꢀtemperatureꢀ(Schemeꢀ1).ꢀ
Aꢀ heterogenousꢀ mixtureꢀ ofꢀ alcoholꢀ (1ꢀ mmol),ꢀ aceticꢀ
anhydrideꢀ(1.2ꢀmmol)ꢀandꢀtheꢀ20CuO‐ZnOꢀnanocatalystꢀ(0.05ꢀ
g)ꢀinꢀCH
2
Cl ꢀ(10ꢀmL)ꢀwasꢀstirredꢀatꢀroomꢀtemperature.ꢀUponꢀ
2
completionꢀofꢀtheꢀreaction,ꢀasꢀdeterminedꢀbyꢀTLC,ꢀtheꢀmixtureꢀ
wasꢀ filteredꢀ toꢀ recoverꢀ theꢀ catalyst.ꢀ Theꢀ catalystꢀ wasꢀ washedꢀ
withꢀCH
usedꢀ inꢀ consecutiveꢀ runs.ꢀ Theꢀ combinedꢀ organicꢀ layersꢀ wereꢀ
washedꢀ sequentiallyꢀ withꢀ 5%ꢀ (w/v)ꢀ NaHCO ꢀ solutionꢀ andꢀ
waterꢀ andꢀ thenꢀ driedꢀ overꢀ MgSO .ꢀ Theꢀ solventꢀ wasꢀ thenꢀ
2 2
Cl ꢀ(5ꢀmL)ꢀandꢀthenꢀdriedꢀatꢀXꢀ°CꢀforꢀYꢀhꢀbeforeꢀbeingꢀ
2.ꢀ ꢀ Experimentalꢀ
3
4
2
.1.ꢀ ꢀ Preparationꢀofꢀcatalystꢀ
removedꢀinꢀvacuoꢀtoꢀgiveꢀtheꢀcrudeꢀproductꢀasꢀaꢀresidue,ꢀwhichꢀ
wasꢀ purifiedꢀ byꢀ columnꢀ chromatographyꢀ overꢀ silicaꢀ gelꢀ toꢀ
affordꢀtheꢀpureꢀdesiredꢀproductsꢀinꢀhighꢀyield.ꢀ
Allꢀofꢀtheꢀchemicalsꢀusedꢀinꢀthisꢀstudyꢀwereꢀpurchasedꢀfromꢀ
Merckꢀ (Germany).ꢀ Theꢀ catalystꢀ wasꢀ preparedꢀ usingꢀ aꢀ
co‐precipitationꢀ method.ꢀ Briefly,ꢀ aꢀ Na CO ·H Oꢀ solutionꢀ (0.5ꢀ
mol/L)ꢀwasꢀaddedꢀtoꢀaꢀmixtureꢀofꢀ0.03ꢀmol/LꢀCu(NO ·3H Oꢀ
andꢀ Zn(NO ·6H Oꢀ inꢀ waterꢀ inꢀ aꢀ drop‐wiseꢀ mannerꢀ withꢀ
2
3
2
3.ꢀ ꢀ Resultsꢀandꢀdiscussionꢀ
)
3 2
2
)
3 3
2
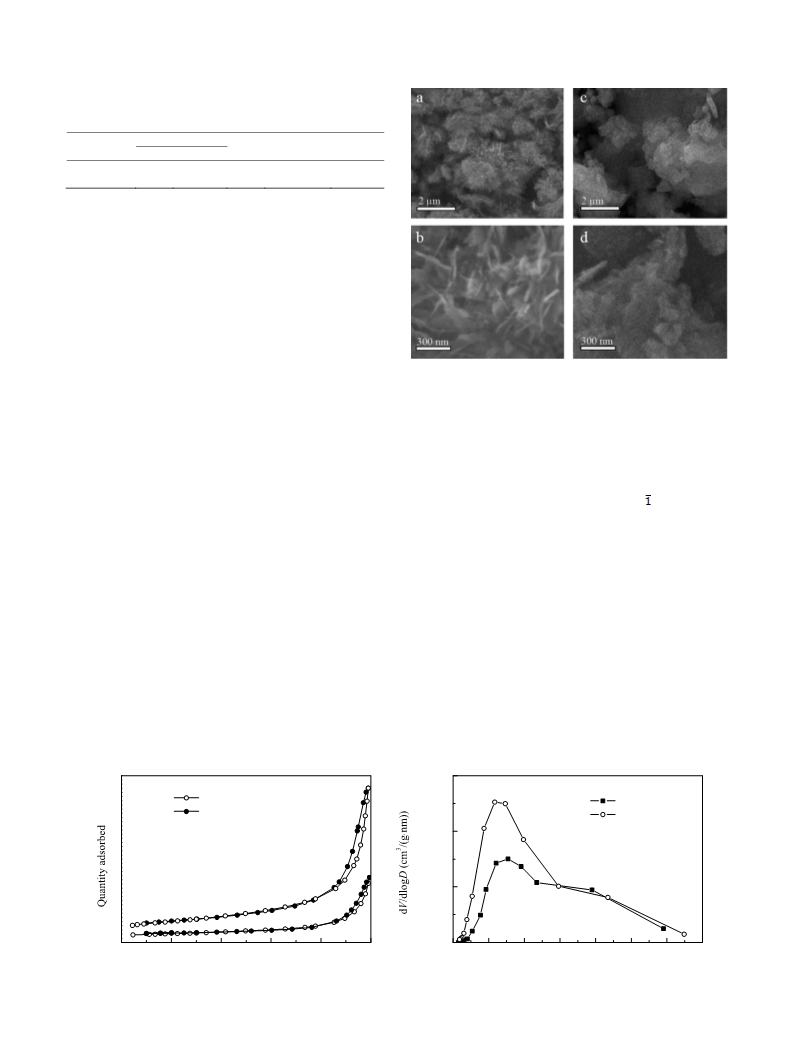
Theꢀ catalystꢀ wasꢀ characterizedꢀ byꢀ XRD,ꢀ N
2
ꢀ adsorption‐
ꢀ
continuousꢀstirring,ꢀandꢀtheꢀresultingꢀsuspensionꢀwasꢀheldꢀatꢀ
pHꢀ=ꢀ8.5ꢀforꢀ15ꢀminꢀatꢀ65ꢀ°C.ꢀTheꢀmixtureꢀwasꢀthenꢀfilteredꢀandꢀ
theꢀ filter‐cakeꢀ washedꢀ withꢀ warmꢀ deionizedꢀ water.ꢀ Theꢀ
precipitateꢀwasꢀsubsequentlyꢀcollectedꢀandꢀdriedꢀatꢀ100ꢀ°Cꢀforꢀ
desorption,ꢀSEM,ꢀTEM,ꢀandꢀEDSꢀanalyses.ꢀ
Figureꢀ 1ꢀ showsꢀ theꢀ XRDꢀ patternsꢀ ofꢀ theꢀ pureꢀ ZnOꢀ andꢀ
20CuO‐ZnOꢀsamples.ꢀSeveralꢀpeaksꢀinꢀtheꢀXRDꢀpatternsꢀwereꢀ
attributedꢀ toꢀ ZnOꢀ overꢀ aꢀ wideꢀ rangeꢀ ofꢀ 2θꢀ values,ꢀ includingꢀ
thoseꢀ atꢀ 31.7°,ꢀ 34.3°,ꢀ 36.3°,ꢀ 47.3°,ꢀ 56.4°,ꢀ 62.8°,ꢀ 66.2°,ꢀ 67.8°,ꢀ
69.0°,ꢀ72.5°,ꢀandꢀ76.9°,ꢀwhichꢀwereꢀcharacteristicsꢀofꢀtheꢀ(100),ꢀ
(002),ꢀ (101),ꢀ (102),ꢀ (110),ꢀ (103),ꢀ (200),ꢀ (112),ꢀ (201),ꢀ (004),ꢀ
andꢀ(202)ꢀplanesꢀofꢀZnOꢀ(JCPDSꢀ89‐1397).ꢀ20CuO‐ZnOꢀcatalystꢀ
revealedꢀ diffractionꢀ peaksꢀ atꢀ 2θꢀ =ꢀ 35.5°,ꢀ 35.6°,ꢀ 38.7°,ꢀ andꢀ
12ꢀhꢀfollowedꢀbyꢀcalcinationꢀatꢀ300ꢀ°Cꢀforꢀ3ꢀh.ꢀAꢀbatchꢀofꢀtheꢀ
ZnOꢀ supportꢀ wasꢀ alsoꢀ preparedꢀ underꢀ theseꢀ conditionsꢀ forꢀ
supplementaryꢀ tests.ꢀ Theꢀ resultingꢀ catalystꢀ wasꢀ namedꢀ
20CuO/ZnO,ꢀ whereꢀtheꢀ20ꢀrefersꢀtoꢀtheꢀweightꢀpercentageꢀofꢀ
CuOꢀinꢀtheꢀcatalyst.ꢀ
38.92°,ꢀ whichꢀ wereꢀ attributedꢀ toꢀ theꢀ (002),ꢀ (11 ),ꢀ (111)ꢀ andꢀ
2
.2.ꢀ ꢀ Characterizationꢀ
(200)ꢀplanesꢀofꢀcubicꢀCuO,ꢀrespectivelyꢀ(JCPDSꢀ05‐0661).ꢀ
TheꢀpeaksꢀintensitiesꢀforꢀtheꢀZnOꢀcrystalsꢀinꢀtheꢀ20CuO‐ZnOꢀ
catalystꢀwereꢀlowerꢀandꢀbroaderꢀthanꢀthoseꢀofꢀtheꢀZnOꢀsupport.ꢀ
ThisꢀresultꢀindicatedꢀthatꢀtheꢀZnOꢀcrystalsꢀwereꢀlessꢀcrystallineꢀ
andꢀsmallerꢀinꢀsizeꢀfollowingꢀtheꢀadditionꢀofꢀCuO,ꢀwhichꢀcouldꢀ
beꢀattributedꢀtoꢀtheꢀincorporationꢀofꢀCuOꢀintoꢀtheꢀstructureꢀofꢀ
Productsꢀ wereꢀ characterizedꢀ byꢀ aꢀ comparisonꢀ ofꢀ theirꢀ
spectroscopicꢀdataꢀ(e.g.,ꢀNMRꢀandꢀIR)ꢀandꢀphysicalꢀpropertiesꢀ
withꢀ thoseꢀ reportedꢀ inꢀ theꢀ literatureꢀ forꢀ theꢀ authenticꢀ
compounds.ꢀ Theꢀ IRꢀ spectraꢀ wereꢀ recordedꢀ onꢀ aꢀ PerkinElmerꢀ
7
81ꢀ spectrophotometerꢀ (USA).ꢀ Allꢀ ofꢀ theꢀ NMRꢀ spectraꢀ wereꢀ
recordedꢀ onꢀ aꢀ Brukerꢀ Avanceꢀ 400ꢀ MHzꢀ NMRꢀ spectrometerꢀ
Germany).ꢀ Yieldsꢀ referꢀ toꢀ isolatedꢀ pureꢀ products.ꢀ X‐rayꢀ
ZnO
CuO
(
diffractionꢀ(XRD)ꢀanalysesꢀwereꢀconductedꢀonꢀaꢀBrukerꢀAXSꢀD8ꢀ
advancedꢀdiffractometerꢀtoꢀdetermineꢀtheꢀstructuralꢀpropertiesꢀ
ofꢀtheꢀcatalyst.ꢀSamplesꢀforꢀXRDꢀanalysisꢀwereꢀscannedꢀatꢀ2θꢀ
valuesꢀinꢀtheꢀrangeꢀofꢀ10°–80°ꢀatꢀaꢀrateꢀofꢀ0.05°/sꢀusingꢀCuꢀK
radiationꢀ (λꢀ =ꢀ 1.5406ꢀ Å).ꢀ Theꢀ specificꢀ surfaceꢀ areaꢀ ofꢀ theꢀ
samplesꢀ wasꢀ determinedꢀ byꢀ N ꢀ adsorption‐desorptionꢀ
α
ꢀ
(2)
2
experimentsꢀusingꢀtheꢀBETꢀmethod.ꢀBETꢀtestsꢀwereꢀcarriedꢀoutꢀ
usingꢀ anꢀ automatedꢀ gasꢀ adsorptionꢀ analyzerꢀ (Tristarꢀ 3020,ꢀ
Micromeritics,ꢀUSA).ꢀPriorꢀtoꢀtheꢀtests,ꢀsamplesꢀofꢀtheꢀcatalystꢀ
wereꢀ degassedꢀ underꢀ vacuumꢀ atꢀ 150ꢀ °Cꢀ forꢀ 2ꢀ h.ꢀ Theꢀ
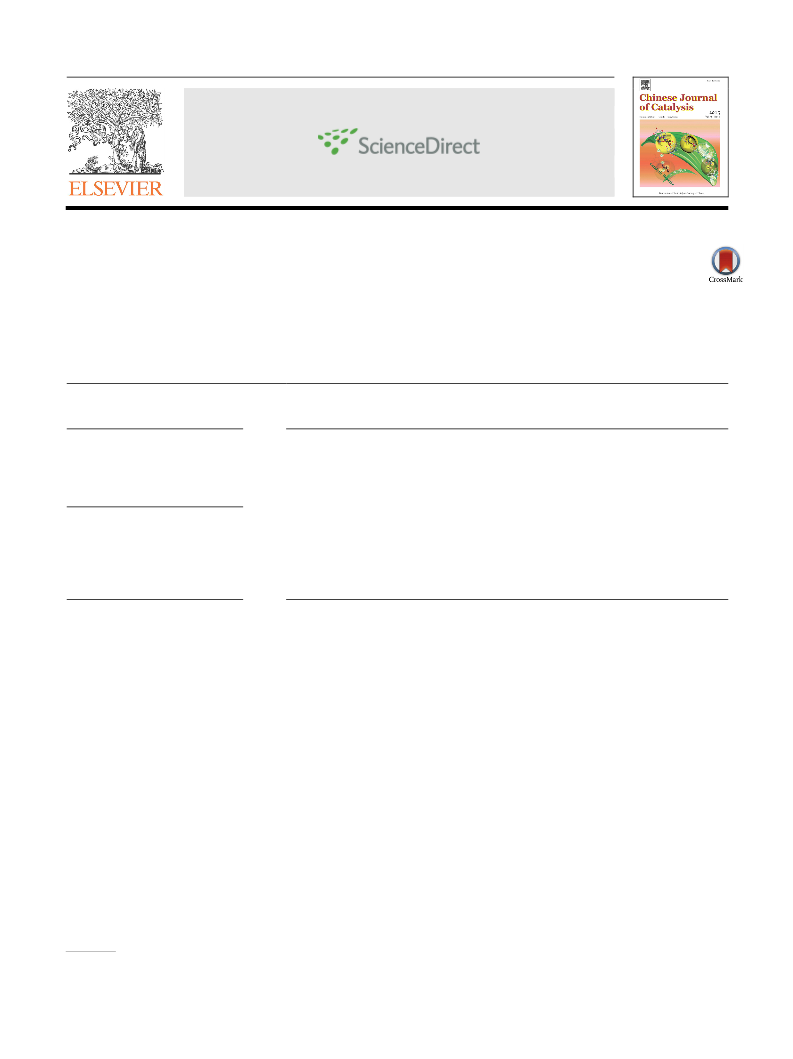
morphologicalꢀcharacteristicsꢀofꢀtheꢀcatalystꢀwereꢀinvestigatedꢀ
comprehensivelyꢀ byꢀ fieldꢀ emissionꢀ scanningꢀ electronꢀ
(
1)
20
30
40
50
60
70
80
o
2
/( )
Fig.ꢀ1.ꢀXRDꢀpatternsꢀofꢀZnOꢀ(1)ꢀandꢀ20CuO‐ZnOꢀ(2)ꢀnanocatalyst.ꢀ

 Albadi, Jalal
Albadi, Jalal
 Alihosseinzadeh, Amir
Alihosseinzadeh, Amir
 Mardani, Mehdi
Mardani, Mehdi