
Catalysis Letters p. 2734 - 2745 (2018)
Update date:2022-08-11
Topics:
 Veisi, Hojat
Veisi, Hojat
 Vafajoo, Saba
Vafajoo, Saba
 Bahrami, Kiumars
Bahrami, Kiumars
 Mozafari, Bita
Mozafari, Bita
To fabricate SiO2/PDA–SO3H nanocatalyst, a suitable method is designed for the loading of sulfonic acid groups on the surface of polydopamine (PDA)-encapsulated SiO2 nanoparticles. To bridge the gap between heterogeneous and homogeneous catalysis, surface functionalization of silica gel is an elegant procedure. The morphology, structure, and physicochemical features were specified using different analytical techniques including field emission scanning electron microscopy (FESEM), Fourier transformed infrared spectroscopy (FT-IR), high resolution-transmission electron microscopy (HR-TEM), energy dispersive X-ray spectroscopy (EDS), wavelength-dispersive X-ray spectroscopy (WDX), X-ray photoelectron spectroscopy (XPS), and back titration. The SiO2/PDA–SO3H nanoparticles are efficient nanocatalysts for the acetylation of many alcohols, phenols, and amines with acetic anhydride under solvent-free conditions in good to excellent yields. Moreover, the reuse and recovery of the catalyst was shown seven times without detectible loss in activity. Graphical Abstract: [Figure not available: see fulltext.]
View More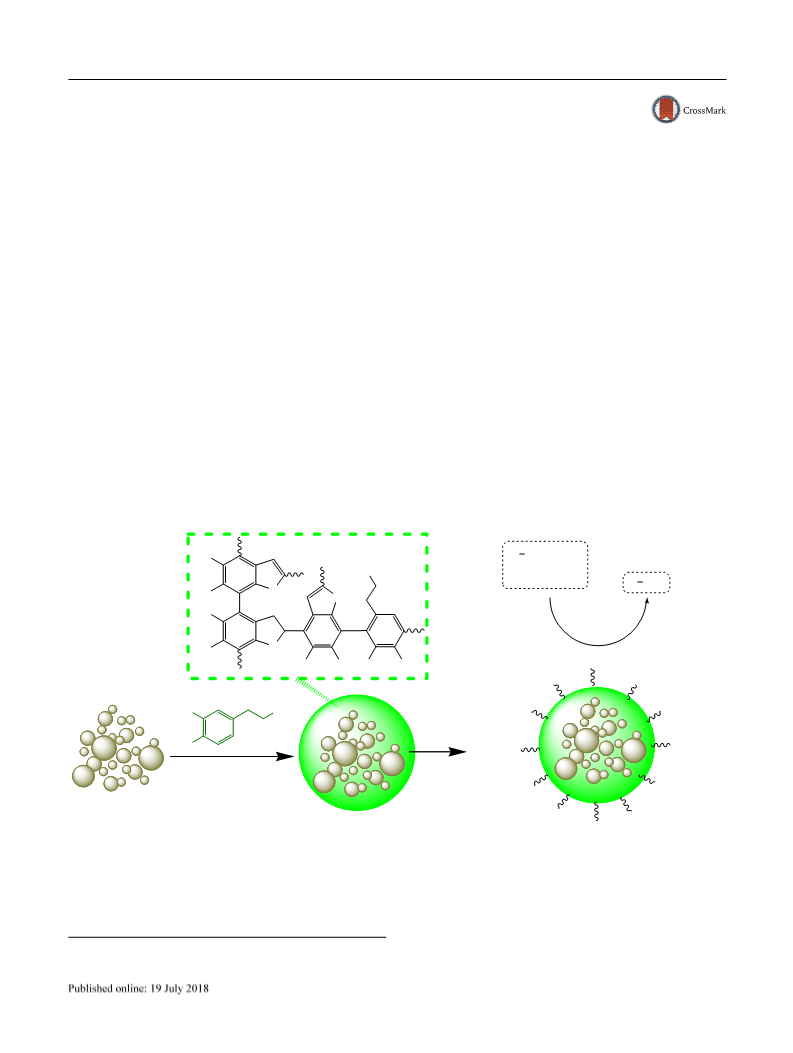










Nantong Advance Chemical Co., Ltd
Contact:86-13818730908
Address:Fine Chemical Industry Garden,Beixin Town,Qidong
Contact:+86-20-62802632;62802633
Address:Room 330 GIGCAS Building,No.511 Kehua Street,Tianhe District
Contact:86-25-51817806
Address:No. 216, middle longpan road, jincheng tower, floor 21-22, nanjing ,china
website:http://www.hanwayschem.com
Contact:+86-18502787239(whatsapp)
Address:18-1-802, Green Garden, Jianghan District, Wuhan 430023, China
Qingdao XinYongAn Chemicals Co., Ltd
Contact:+86-532-81107967
Address:Chengyang dual-port industrial park by the sea,Qingdao
Doi:10.1039/c6ra21203d
(2016)Doi:10.1002/hlca.19390220193
(1939)Doi:10.1021/acs.joc.6b00647
(2016)Doi:10.1016/0025-5408(91)90018-H
(1991)Doi:10.1007/s12272-016-0737-5
(2016)Doi:10.1007/BF00486799
(1991)