RSC Advances
Paper
reference. HRMS data were obtained using an Agilent 1290LC-
6540 Accurate Q-TOF mass spectrometer.
2.3 Preparation of Derivative 2
Di-O-isopropylidene-2,3:4,5-b-D-fructopyranosyl
tri-
uoromethanesulfonate (736.70 mg, 1.88 mmol) and phenyl-
alanine ethyl ester (544.27 mg, 2.82 mmol) were mixed in
dimethylformamide (30 mL) and then heated at 120 ꢀC for 6 h.
The progress of the reaction was monitored by thin-layer chro-
matography (TLC) with petroleum ether–ethyl acetate (3 : 1,
v : v). When the starting materials were consumed, the solvent
was evaporated under reduced pressure. The crude residue was
puried on a silica gel column with petroleum ether–ethyl
acetate (3 : 1, v : v) to give the target compound as a yellow oil
(342.20 mg, 41.85%). 1H NMR (400 MHz, CDCl3 containing
0.03% TMS, 25 ꢀC): d: 7.28–7.15 (m, 5H, Ar), 4.58 (dd, J ¼ 7.9 Hz,
J ¼ 2.6 Hz, 1H, CH), 4.38 (d, J ¼ 2.6 Hz, 1H, CH), 4.22 (dd, J ¼
7.9 Hz, J ¼ 1.0 Hz, 1H, CH), 4.06 (q, J ¼ 7.2 Hz, 2H, CH2), 3.81
(dd, 2J ¼ 64.6 Hz, 3J ¼ 12.9 Hz, 2H, CH2), 3.66 (t, J ¼ 6.8 Hz, 1H,
CH), 2.97 (m, 2H, CH2), 2.84 (s, 2H, CH2), 1.51 (s, 3H, CH3), 1.46
(s, 3H, CH3), 1.38 (s, 3H, CH3), 1.34 (s, 3H, CH3), 1.13 (t, J ¼
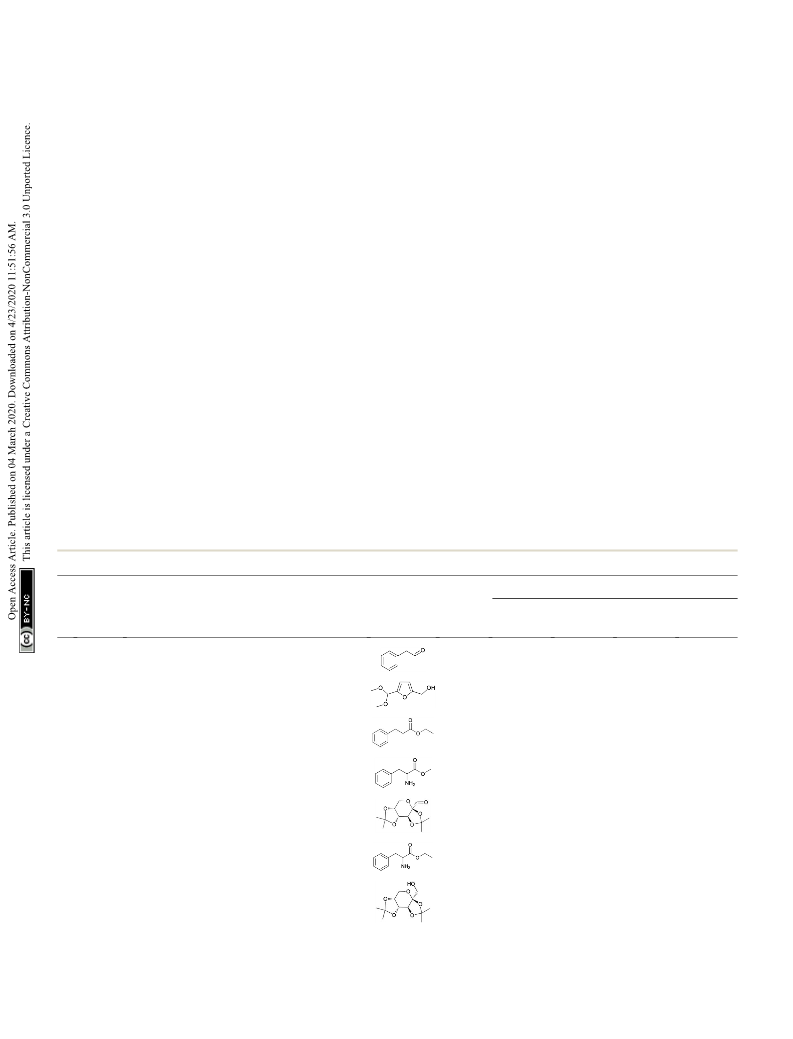
Fig. 1 Chemical structures of Derivative 1 and Derivative 2.
13
ꢀ
7.2 Hz, 3H, CH3). C NMR (100 MHz, CDCl3, 25 C): d: 174.21
(CO), 137.19 (C, Ar), 129.27 (C, Ar), 128.36 (C, Ar), 126.64 (C, Ar),
108.91 (C), 108.15 (C), 103.48 (C), 71.12 (CH), 71.07 (CH), 70.40
(CH), 63.23 (CH2), 61.24 (CH2), 60.46 (CH), 52.94 (CH2), 39.26
(CH2), 26.55 (CH3), 25.89 (CH3), 25.56 (CH3), 24.06 (CH3), 14.11
(CH3). HRMS ESI-TOF: m/z ¼ 458.2154 [M + Na]+ (calcd for
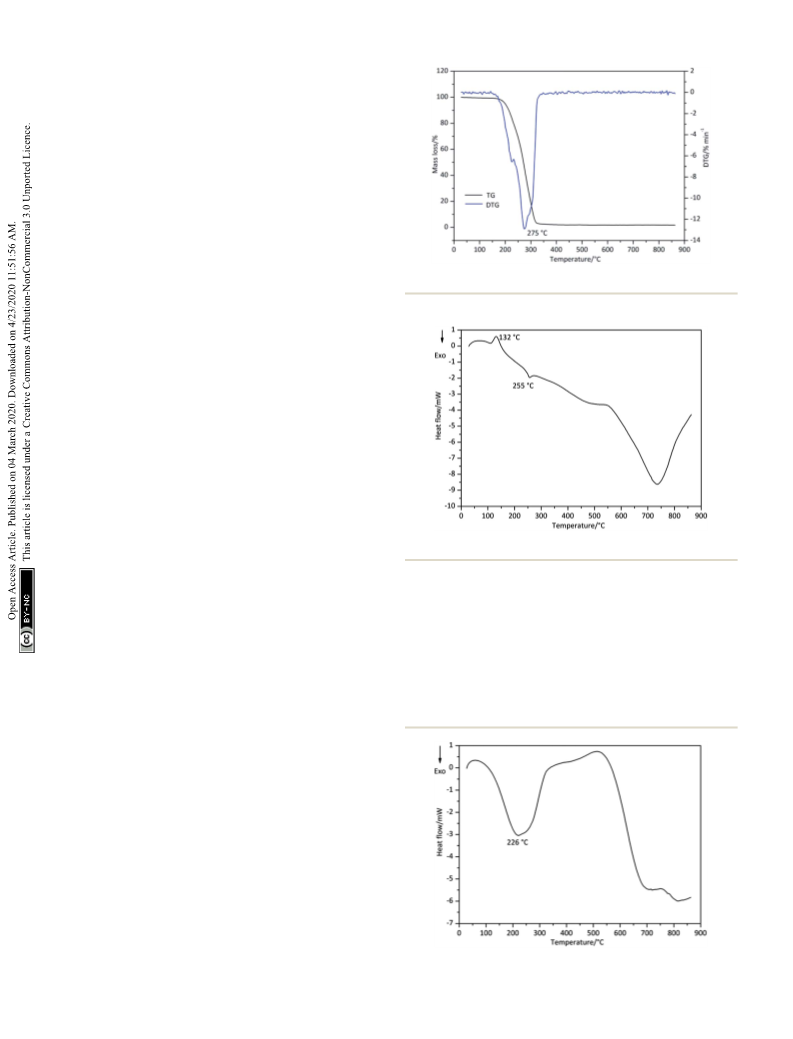
Fig. 2 TG and DTG curves of Derivative 1.
2. Materials and methods
2.1 Materials
C
23H33NO7Na, 458.2155).
Methanol, dimethylformamide, dichloromethane, acetone,
ethyl acetate, triuoroacetic acid, and petroleum ether were
purchased from Tianjin Kermel Chemical Reagent Co., Ltd
(China). The above reagents were of analytical grade. Deionized
water (ltered through a 0.22 mm membrane lter, >18.2 MU
cm) was puried using a Milli-Q system from Millipore. Silica
gel was purchased from Qingdao Haiyang Chemical Reagent
Co., Ltd (China). Phenylalanine ethyl ester, di-O-isopropylidene-
2,3:4,5-b-D-fructose and di-O-isopropylidene-2,3:4,5-b-D-fructo-
pyranosyl triuoromethanesulfonate were synthesized accord-
ing to ref. 18 and 19, respectively.
2.4 Preparation of Derivative 1
Derivative 1 was prepared through the hydrolysis of Derivative 2.
The reaction procedure is described below. Derivative 2
(119.10 mg, 0.27 mmol) was stirred in 3.97 mL of triuoroacetic
acid–water (v : v 9 : 1) for 1 h at room temperature. Then, the
solvent was removed on a rotary evaporator at 45 ꢀC. The crude
residue was puried on a silica gel column with ethyl acetate to
give the target compound as a white oil (65.67 mg, 68.44%). 1H
ꢀ
NMR (400 MHz, MeOD, 25 C): d: 7.33–7.30 (m, 2H, Ar), 7.28–
7.22 (m, 3H, Ar), 4.20–4.15 (m, 1H, CH), 4.11 (q, J ¼ 7.1 Hz, 2H,
CH2), 3.98–3.95 (m, 1H, CH), 3.87 (m, 1H, CH), 3.81–3.77 (m,
1H, CH), 3.69–3.63 (m, 2H, CH2), 3.28–3.08 (m, 2H, CH2), 3.15
(s, 2H, CH2), 1.10 (t, J ¼ 7.1 Hz, 3H, CH3). 13C NMR (100 MHz,
MeOD, 25 ꢀC): pyranose form d: 171.47 (CO), 136.36 (C, Ar),
2.2 Structure identication
1H NMR and 13C NMR spectra data were recorded on a Brucker 130.49 (C, Ar), 130.06 (C, Ar), 128.83 (C, Ar), 97.26 (C), 71.46
400 MHz spectrometer. Each sample was dissolved in CDCl3 or (CH), 71.25 (CH), 70.81 (CH), 64.93 (CH), 63.23 (CH2), 63.14
CD3OD containing tetramethylsilane (TMS) as the internal (CH2), 53.56 (CH2), 37.81 (CH2), 14.22 (CH3). Furanose form d:
Table 1 Thermal analysis data for Derivative 1 and Derivative 2
DSC
TG–DTG
Tp/ꢀC
Sample name
Tonset/ꢀC
Tpeak/ꢀC
Tend/ꢀC
DH/kJ molꢁ1
Trange/ꢀC
Derivative 1 (peak 1)
Derivative 1 (peak 2)
Derivative 2
110
247
81
132
255
226
146
272
351
25
132
254
275
103–193
217–279
160–341
ꢁ13
ꢁ852
9310 | RSC Adv., 2020, 10, 9309–9317
This journal is © The Royal Society of Chemistry 2020

 Li, Rui
Li, Rui
 Zhang, Shiyi
Zhang, Shiyi
 Zhang, Yudan
Zhang, Yudan
 Zhao, Mingqin
Zhao, Mingqin