10.1002/anie.202003635
Angewandte Chemie International Edition
RESEARCH ARTICLE
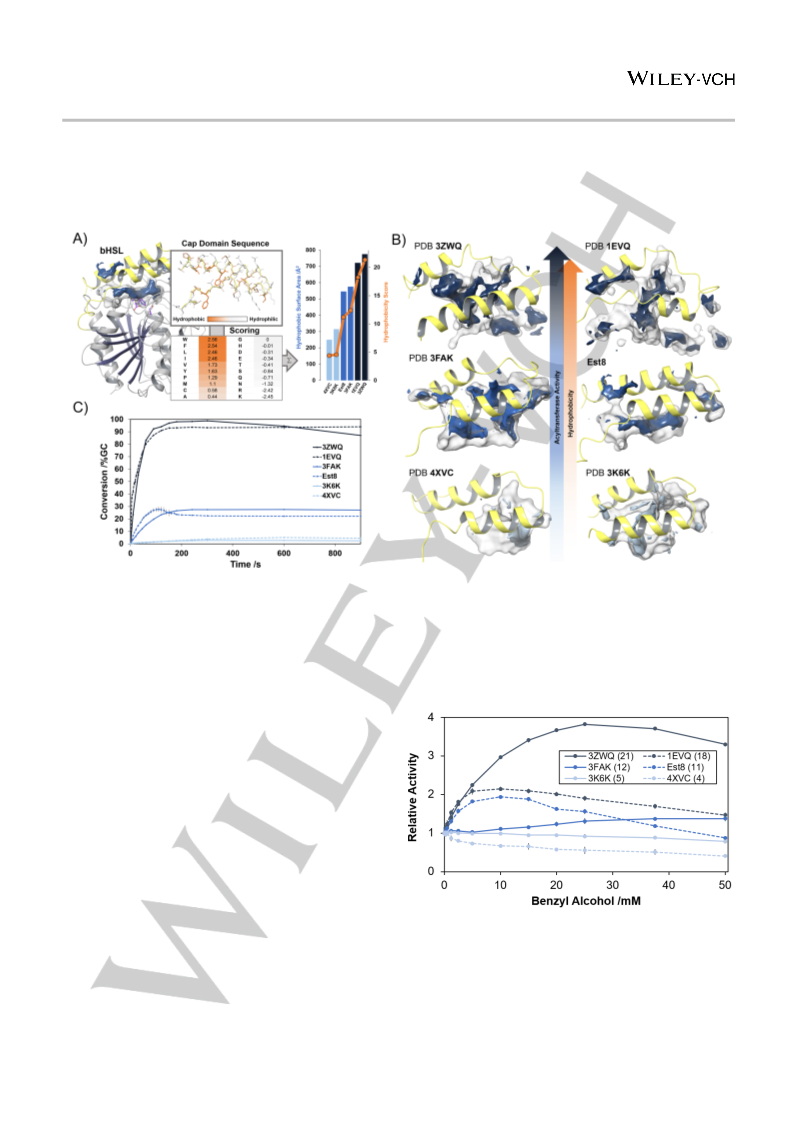
the cap domain is highly variable (Fig. S1). Because of this struc-
tural feature, we postulated that active site hydrophobicity of
bHSLs could be estimated from the amino acid sequence of the
cap domain. For that purpose, we developed a sequence-based
hydrophobicity scoring system for bHSLs (Fig. 2A) using a hydro-
phobicity scale for amino acids.[23] On this scale, hydrophobic res-
idues have positive values, while polar and charged residues
have negative values and thus contribute as penalties. We
summed the values for the 45 N-terminal residues, which form the
cap domain, as visualized in Fig. 2A. Hydrophobic pockets are
thus expected to have high scores while hydrophilic pockets have
low or even negative scores. In order to demonstrate the ability of
the hydrophobicity score to reflect actual active site hydrophobi-
city, we analyzed the crystal structures of five other bHSLs (PDB-
codes: 3ZWQ, 1EVQ, 3FAK, 4XVC, and 3K6K). Hydrophobic
pocket surface areas (visualized in Fig. 2B) calculated from the
crystal structures were plotted against the hydrophobicity scores,
revealing a nearly perfect coherence (correlation coefficient r =
0.98, Fig. 2A).
The same set of bHSLs was experimentally analyzed for pro-
miscuous acyltransferase activity in a model reaction (Fig. 1A) in-
volving the acetylation of 2-phenylethanol in the presence of a
ten-fold excess of vinyl acetate as acyl donor (Fig. 2C). Formation
of the product, 2-phenylethyl acetate, was quantified by gas chro-
matography (GC). For Est8, product hydrolysis became more
dominant than acyl transfer quickly after reaching a kinetic maxi-
mum (~27% conversion) at low concentrations of 2-phenylethyl
acetate. On the way to reaching its thermodynamic equilibrium,
the reaction is rapidly quenched by acidification, indicating high
hydrolase activity. Enzymes with scores lower than Est8, such as
3K6K[24] and 4XVC[25], behaved as conventional hydrolases with
nearly no acyltransferase activity (<5% conversion) in water. An-
other esterase with a score equal to that of Est8, 3FAK[26], showed
acyltransferase activity comparable to Est8 (~ 27% conversion).
Most strikingly, the enzymes with hydrophobicity scores higher
than that of Est8, specifically 1EVQ[27] and 3ZWQ,[28] were excel-
lent acyltransferases that could nearly fully convert 2-phenyleth-
anol to 2-phenylethyl acetate (Fig. 2C).
The product is then not enzymatically hydrolyzed anymore, and
its concentration remains constant. The dotted line represents the
product hydrolysis that would be observed if the enzyme would
not be inactivated by acidification.
Unlike naturally occurring CoA-dependent acyltransferases,
which often show virtually no hydrolase activity, promiscuous
acyltransferases can hydrolyze both the acyl donor and the trans-
esterification product. Therefore, the product of transesterification
only transiently accumulates, and high conversions can only be
achieved in a kinetically controlled reaction (Fig. 1B). Conven-
tional hydrolases do not show a transient kinetic maximum above
the thermodynamic equilibrium under the same conditions.[13] In
any case, hydrolysis leads to the release of free carboxylic acids,
which progressively acidifies the reaction. When using high sub-
strate concentrations, this can lead to a pH drop that inactivates
the enzyme and the kinetic product cannot be fully hydrolyzed.[12]
To judge the efficiency of promiscuous acyltransferases in a spe-
cific kinetically controlled reaction, the transfer to hydrolysis ratio
(kt/kh; Fig. 1A) became a commonly accepted parameter.[3,14]
The few known promiscuous acyltransferases come with sev-
eral synthetic limitations. Even though some CAL-A-related acyl-
transferases show great efficiency, their application is limited to
long-chain fatty acid esters as acyl donors, with CpLIP2 even be-
ing inhibited by short-chain esters like ethyl acetate.[15] MsAcT is
generally not enantioselective and does not accept more polar
substrates like amino acids.[10,12,16]
Due to the small number of known acyltransferases, the struc-
tural prerequisites for efficient acyltransferase activity in water are
also not fully understood. Jiang et al. discussed the role of the
immediate surrounding of the catalytic triad and proposed a
mechanism for acyltransferases that involves the inactivation of
attacking water molecules by acidic backbone amide hydro-
gens.[17] For MsAcT, it has been speculated that the highly hydro-
phobic microenvironment formed by its oligomeric structure fa-
vors the binding of organic nucleophiles over water.[7,18] Support-
ing this, studies of CAL-A and CpLIP2 revealed that substitution
of active site residues by more hydrophobic residues improves
acyltransferase activity.[8,19]
We recently reported a high-throughput assay for identifying
more enzymes with acyltransferase activity in water.[20] By screen-
ing several hydrolases using this assay, we identified a member
of the family of bacterial hormone-sensitive lipases (bHSLs), Est8,
as a promiscuous acyltransferase.[20,21] Based on a detailed struc-
ture-function analysis of Est8[22] and several homologs, the pre-
sent study aims to clarify molecular prerequisites for promiscuous
acyltransferase activity, enabling sequence-based prediction of
this remarkable phenomenon.
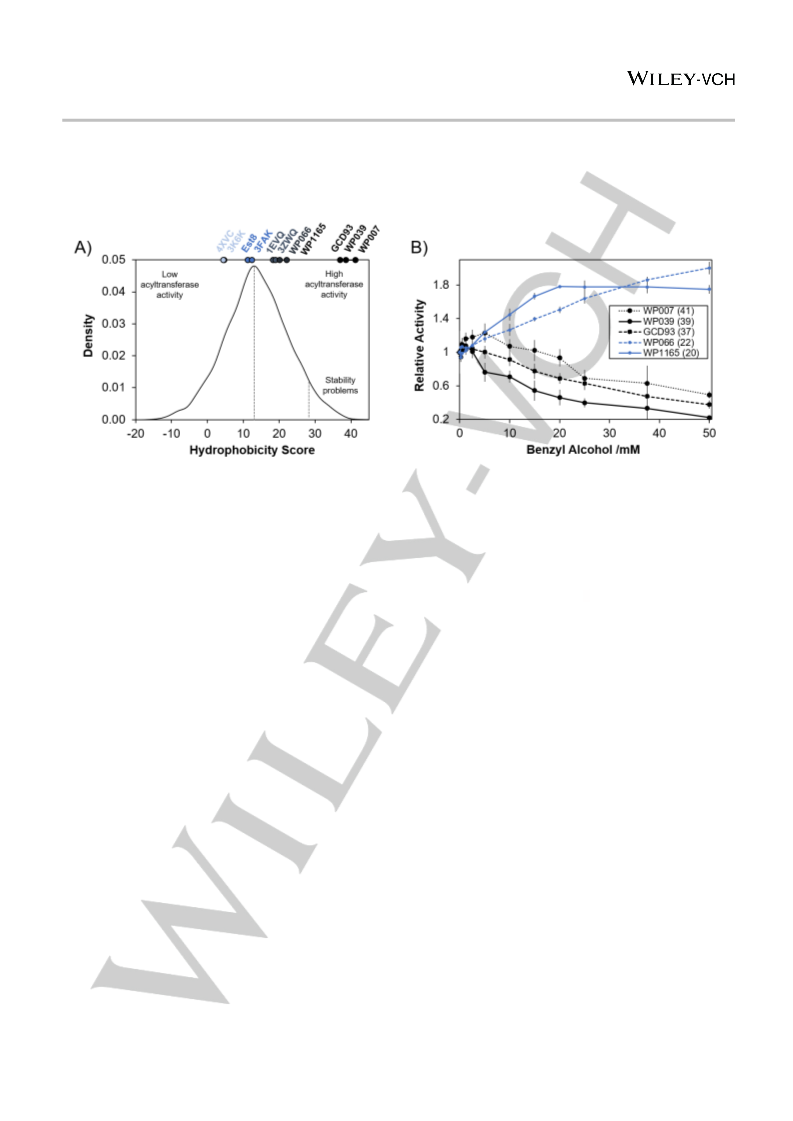
Even though the prediction of promiscuous activities is com-
monly considered to be extremely difficult[29], the active site hy-
drophobicities calculated from crystal structures are hence highly
predictive of acyltransferase activity in water. Consequently, the
hydrophobicity score, which accurately reflects active site hydro-
phobicity, can be used to predict promiscuous acyltransferase ac-
tivity within this enzyme family.
In order to gain more insights into the acyltransferase effi-
ciency of the newly identified acyltransferases, we needed a more
sophisticated assay that provides information about the relative
rates of acyl transfer to acyl donor hydrolysis. However, our oligo-
carbonate-assay[20] provides only qualitative information. Mes-
trom et al. recently reported a coupled spectrophotometric acyl-
transferase assay which we could not use since the employed al-
cohol dehydrogenase precipitates in the presence of organic nu-
cleophiles other than benzyl alcohol. Therefore, we developed a
robust and versatile acyltransferase assay using para-nitrophenyl
esters as acyl donors. Our assay has the advantage of being com-
patible with virtually any organic nucleophile as acyl acceptor.
Upon formation of the acyl-enzyme intermediate, para-nitrophe-
nolate (pNP) is released and can be quantified by measuring the
increase in absorbance at 405 nm. In the absence of any other
nucleophile than water, the half-life of the acyl-enzyme intermedi-
ate depends only on the rate of hydrolysis. However, if an organic
nucleophile is preferred over water, the half-life of the acyl-en-
zyme intermediate is expected to be shorter in its presence, re-
sulting in an overall faster release of pNP (Fig. S3A). To show that
the increased release of pNP is directly correlated with enzymatic
transesterification, and not due to a nonspecific acceleration of
pNPA hydrolysis, the formation of the transesterification product,
benzyl acetate, was confirmed by GC analysis. A direct correla-
tion between the rate of pNP release at different benzyl alcohol
concentrations and benzyl acetate formation was observed (Fig.
Results and Discussion
Est8 is the first member of the family of bacterial hormone-sensi-
tive lipases (bHSLs) for which promiscuous acyltransferase activ-
ity in water has been reported. In order to elucidate the structural
basis for this activity, we solved the crystal structure of Est8 (PDB-
code: 6Y9K; Table S1). We observed that its substrate-binding
pocket consists of a well-defined tunnel, which leads from the pro-
tein surface to the active site harboring a catalytic triad (Ser146
,
Glu240, His270, Fig. 2A). Remarkably, this tunnel is highly hydro-
phobic due to the presence of several methionine residues that
are part of the N-terminal cap domain, a helix-turn-helix motif cov-
ering the active site. As discussed for CpLIP2, CAL-A, and MsAcT,
we assumed that active site hydrophobicity also plays a role for
the acyltransferase activity of Est8. As typical for bHSLs, the cap-
domain forms the most significant part of Est8's substrate-binding
pocket. In contrast to the conserved C-terminal catalytic domain,
2
This article is protected by copyright. All rights reserved.

 Badenhorst, Christoffel P. S.
Badenhorst, Christoffel P. S.
 Becker, Ann-Kristin
Becker, Ann-Kristin
 Berndt, Leona
Berndt, Leona
 Bornscheuer, Uwe T.
Bornscheuer, Uwe T.
 Godehard, Simon P.
Godehard, Simon P.
 Lammers, Michael
Lammers, Michael
 Müller, Henrik
Müller, Henrik
 Palm, Gottfried J.
Palm, Gottfried J.
 Reisky, Lukas
Reisky, Lukas