
Phosphorus, Sulfur and Silicon and the Related Elements p. 181 - 186 (2010)
Update date:2022-08-16
Topics:
 Khazaei, Ardeshir
Khazaei, Ardeshir
 Rostami, Amin
Rostami, Amin
 Rosta, Zahra
Rosta, Zahra
 Alavi, Ali
Alavi, Ali
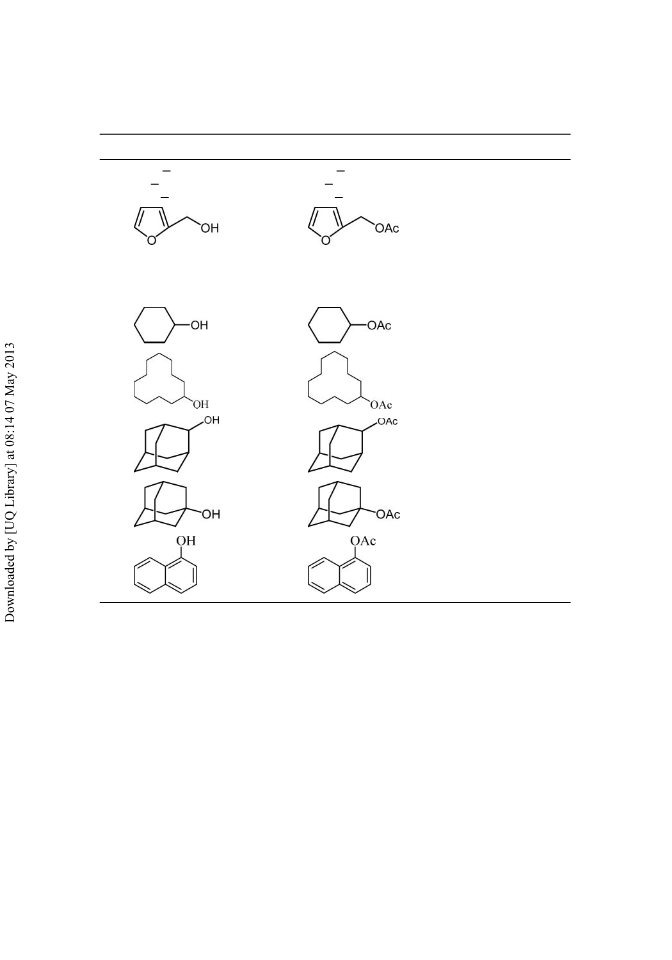
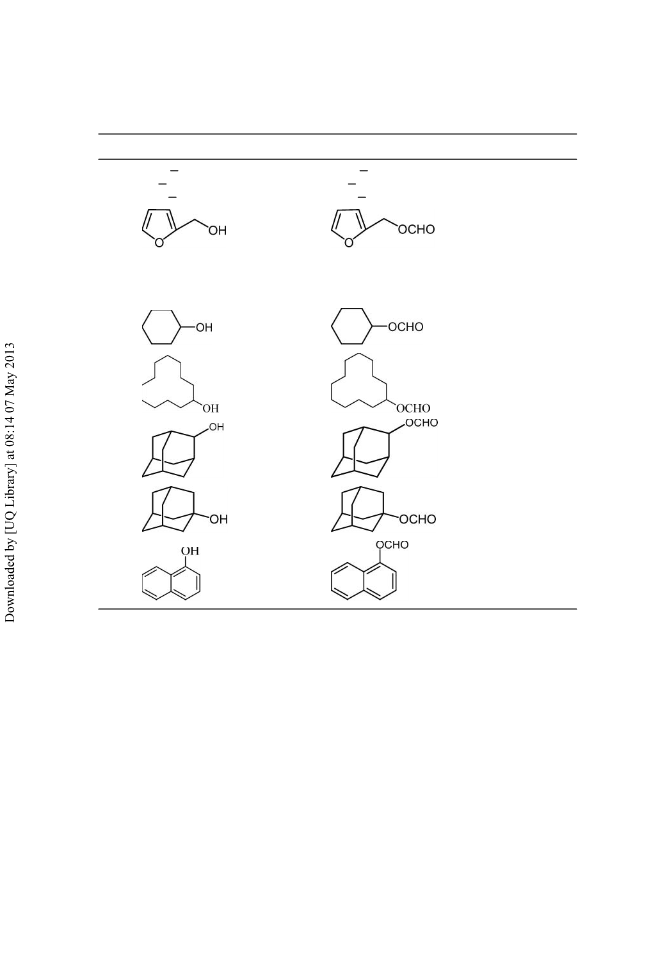
An efficient method for the acylation and formylation of alcohols and phenols by using an acylating/formylating agent (acetic anhydride and formic acid) in the presence of a catalytic amount of N,N′-dibromo-N,N′-1, 2-ethanediylbis(benzene sulfonamide) under mild and solvent-free conditions at room temperature in good to excellent yields is described. The use of protic acids and metal Lewis acids is avoided.
View More




Kaiping Genuine Biochemical Pharmaceutical Co.,Ltd.
Contact:+86-750-2881198
Address:No.1, Xinke Road, Shatang Town, Kaiping, Guangdong Province, P.R.China
Contact:+86-(0)21-3770 9035
Address:Room 301, Building 2, Meijiabang Road 1508, Shanghai China
Fujian Huitian Biological Pharmacy Co., Ltd.
Contact:0086-598-8300831; 8339920
Address:No.46,Taijiang Road,Sanming City,Fujian,China
SuZhou Hua-Emy Chemical Import and Export Co., LTD.
Contact:+86-512-88804994; +86-512-88804550;
Address:710, Building B, International Trade Center, 12 Huanghelu, Changshu, Jiangsu,China
Contact:+86 0310 3166373
Address:shanghai
Doi:10.1016/S0040-4039(01)89618-0
(1967)Doi:10.1021/cm202608f
(2012)Doi:10.1039/C29690000551
(1969)Doi:10.1021/acs.joc.0c01307
(2020)Doi:10.1039/a808072k
(1999)Doi:10.1016/j.dyepig.2015.06.008
(2015)