L. Osiglio et al. / Journal of Molecular Catalysis A: Chemical 316 (2010) 52–58
57
in multifunctional organic compounds. Besides, alcohols could be
acetylated in the presence of phenol, and even the acetylation of
phenol with electron donor substituents could be selective in pres-
ence of phenols with electron-withdrawing groups.
4. Conclusions
The behavior of borated zirconia, obtained using different prepa-
ration conditions, as catalyst in the acetylation reaction of alcohols
and phenol with acetic acid was studied in the present work. The
characteristics of the catalysts depend on the preparation condi-
tions used, which were finally shown in the acid strength of the
materials.
A good agreement between the results obtained in the acetyla-
tion of 2-phenoxyethanol and the catalyst acidity was observed.
The most appropriate catalyst was borated zirconia calcined at
320 ◦C containing 15 g B2O3/100 g of a support obtained using a
high concentration of ZrOCl2 precursor in the starting solution,
achieving a yield of 80% in acetylated product in 14 h under reac-
tion. Besides, the catalysts kept their activity after three catalytic
cycles. Though the yield was lower for benzylic alcohols and phenol,
the methodology employed is a clean alternative for the acetylation
reaction, tending toward eco-efficiency due to the use of relatively
cheap catalysts, which can easily be prepared in the laboratory and
stored for an extended period of time. In addition, water is obtained
as by-product of the reaction when acetic acid is employed as acy-
lating agent in replacement of the classical agents.
Scheme 2. Steric effect in different alcohols.
calcined at 750 ◦C, no reaction was detected, which may be related
to the structural change toward a crystalline phase of the zirconia
inhibits boron interaction, and also a decrease in SBET took place
(11 m2/g).
The same reaction conditions were used to observe the behavior
of a sample of borated zirconia, calcined at 320 ◦C, as catalyst in the
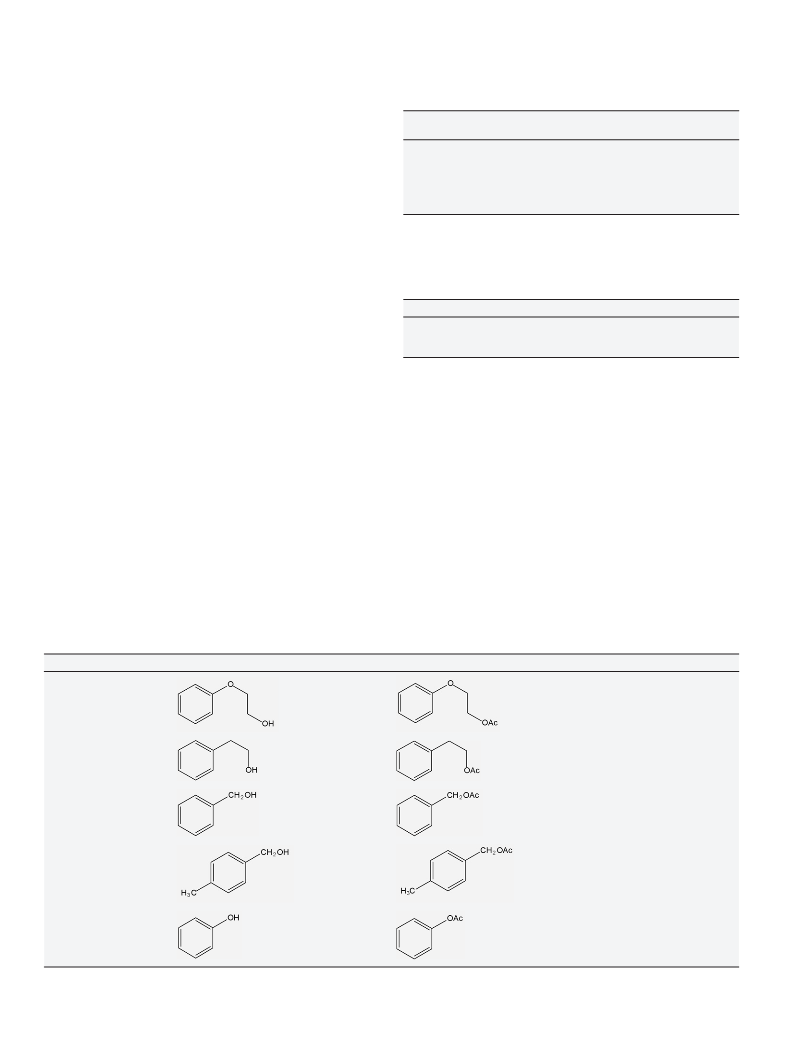
acetylation of other alcohols and phenol with acetic acid (Table 4).
The reactivity of the primary alcohols 2-phenoxyethanol and 2-
phenylethanol (entries 1 and 2) was higher compared to that of the
benzylic alcohol and 4-methyl-benzylic alcohol (entries 3 and 4).
They achieved a yield of acetylated product (67–69%), which was
nearly twice that of the benzylic alcohols (25–35%).
Acknowledgements
be attributed to steric effects that considerably affect the acetyla-
tion rate considering a Fisher esterification mechanism [48]. The
presence of bulky groups, not far from the reaction center, either in
the alcohol or in the acid, slows down the esterification rate, as is
indicated in Scheme 2 for two of the studied alcohols. The aim is to
show that the most sterically hindered alcohols can lead to a lower
yield.
The acetylation of a sterically hindered tertiary alcohol (triph-
enylmethanol) was also performed, in the same conditions as
were detected.
Similar differences in the behavior of primary and benzylic alco-
hols in the acetylation with acetic acid were reported by Mirkhani
et al. [49], using a cerium polyoxometallate as catalyst.
On the other hand, the yield in the phenol acetylation (Table 4,
entry 5) was still lower than that of benzylic alcohols, reaching
a yield of 10%. This behavior can be attributed to its low nucle-
ophilicity, due to the electronic delocalization by resonance in the
benzene ring. An additional experiment with 4-nitrophenol, which
is less nucleophilic due to the electron-withdrawing group NO2, in
identical conditions to phenol acetylation, only gave traces of the
acetylated product.
The authors thank E. Ponzi, G. Valle and E. Soto for their exper-
imental contribution, and UNLP, CONICET and ANPCyT for the
financial support.
References
[1] T. Green, P. Wutz, Protective Groups in Organic Synthesis, 2nd ed., Wiley, New
York, 1991.
[2] P. Kumar, R. Pandey, M. Bodas, S. Dagade, M. Dongare, A. Ramaswamy, J. Mol.
Catal. A: Chem. 181 (2002) 207.
[3] T. Reddy, M. Narasimhulu, N. Suryakiran, Ch. Mahesh, K. Ashalatha, Y.
[4] M. Heravi, F. Behbahani, V. Zadsirjan, H. Oskooiec, J. Braz. Chem. Soc. 17 (2006)
1045.
[5] M. Alizadeh, T. Kermani, R. Tayebee, Monatsh. Chem. 138 (2007) 165.
[6] R. Zhdanov, S. Zhenodarova, Synthesis (1975) 222.
[7] D. Horton, Org. Synth. V (1973) 1.
[8] E. Scriven, Chem. Soc. Rev. 12 (1983) 1296.
[9] G. Hofle, V. Steglich, H. Vorbruggen, Angew. Chem. Int. Ed. Engl. 17 (1978) 569.
[10] T. Sano, K. Ohashi, T. Oriyama, Synthesis (1999) 1141.
[11] E. Vedejs, S. Diver, J. Am. Chem. Soc. 115 (1993) 3358.
[12] A. Cope, E. Herrick, Org. Synth. 4 (1963) 304.
[13] R. Baker, F. Bordwell, Org. Synth. 3 (1955) 141.
[14] J. Iqbal, R. Srivastva, J. Org. Chem. 57 (1992) 2001.
[15] S. De, Tetrahedron Lett. 45 (2004) 2919.
[16] S. Chandrasekhar, T. Ramachander, M. Takhi, Tetrahedron Lett. 39 (1998) 3263.
[17] K. Ishihara, M. Kubota, H. Kurihara, H. Yamamoto, J. Am. Chem. Soc. 117 (1995)
4413.
[18] K. Chauhan, C. Frost, I. Love, D. Waite, Synlett (1999) 1743.
[19] P. Saravanan, V. Singh, Tetrahedron Lett. 40 (1999) 2616.
[20] V. Choudhary, K. Patil, S. Jana, J. Chem. Sci. 116 (2004) 175.
[21] R. Ballini, G. Bosica, S. Carloni, L. Ciaralli, R. Maggi, G. Sartori, Tetrahedron Lett.
39 (1998) 6049.
[22] T. Li, A. Li, J. Chem. Soc. Perkin Trans. 1 (1998) 1913.
[23] G. Romanelli, D. Bennardi, J. Autino, G. Baronetti, H. Thomas, Eur. J. Chem. 5
(2008) 641.
[24] S. Velusamy, S. Borpuzari, T. Punniyamurthy, Tetrahedron 61 (2005) 2011.
[25] A. Vogel, Textbook of Practical Organic Chemistry, 5th ed., Longman Group Ltd.,
London, 1996.
In sum, the reactivity order toward acetylation with acetic
acid with the studied catalysts was as follows: primary alco-
hols > benzylic alcohols > phenol.
It should be noted that in all the tests performed, the selectivity
was high because after the isolation and purification by column, all
It is worth mentioning that in the acetylation of benzylic alcohol
with acetic acid in ionic liquids with metallic triflates as catalyst,
yields between 21 and 64% at 12 h of reaction have been reported
[50]. Unlike the materials employed in the present work, the sol-
vents and catalysts are expensive, and though in this case the
reaction was carried out at low temperature, an activity decrease
with the reuse using triflates as catalyst was observed. In addition,
borated zirconia could be appropriate to the selective acetylation
[26] S. Wright, D. Hageman, A. Wright, L. McCure, Tetrahedron Lett. 38 (1997) 7345.
[27] J. Otera, N. Danoh, H. Nozaki, J. Org. Chem. 56 (1991) 5307.
[28] Z. Zhao, J. Mol. Catal. A: Chem. 154 (2000) 131.
[29] G. Yadav, P. Metha, Ind. Eng. Chem. Res. 33 (1994) 2198.
[30] B. Saha, S. Chopade, S. Mahajani, Catal. Today 60 (2000) 147.
[31] P. Procopiu, S. Baugh, S. Flask, G. Ingliss, J. Org. Chem. 63 (1998) 2342.

 Osiglio, Lilian
Osiglio, Lilian
 Romanelli, Gustavo
Romanelli, Gustavo
 Blanco, Mirta
Blanco, Mirta