
Synthetic Communications p. 1022 - 1028 (2010)
Update date:2022-08-10
Topics:
 Shirini, Farhad
Shirini, Farhad
 Zolfigol, Mohammad Ali
Zolfigol, Mohammad Ali
 Aliakbar, Ali-Reza
Aliakbar, Ali-Reza
 Albadi, Jalal
Albadi, Jalal
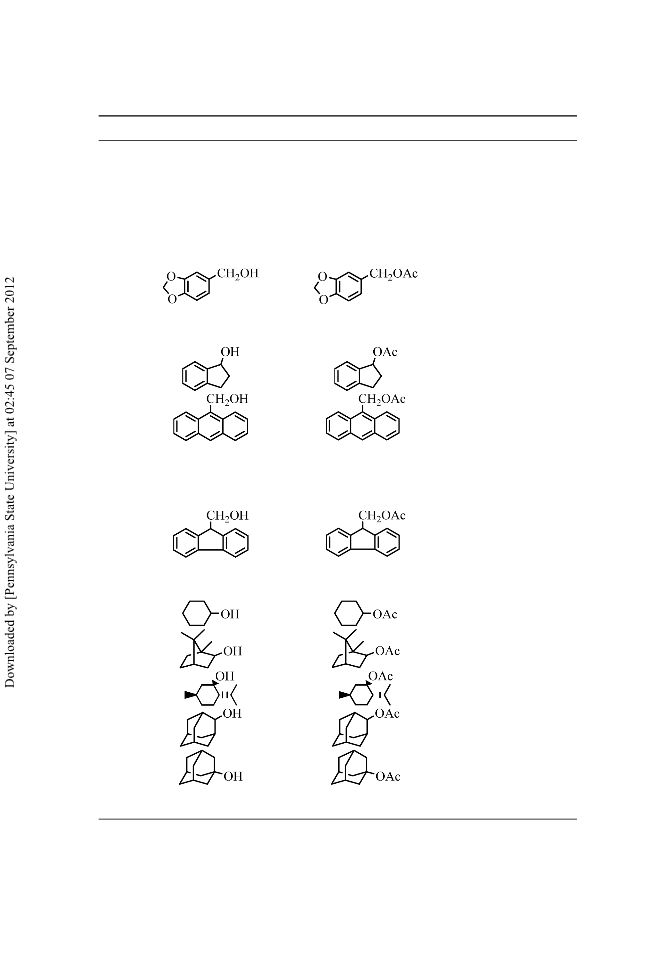
Melamine trisulfonic acid (MTSA) was easily prepared by the reaction of melamine with neat chlorosulfonic acid at room temperature. This reagent can be used as an efficient catalyst for the acetylation of alcohols, phenols, and amines with Ac2O under mild and completely heterogeneous reaction conditions.
View More






website:http://www.shtopchem.com/
Contact:0086-0576-87776998
Address:room no 1608,xuhui business building yude road,xujiahui street, xuhui district
Hefei Highzone Fine Chemical S&T CO.,LTD
Contact:86-0551-63663560
Address:room 1801 NO. 24 Shuguang RD.
Contact:+86-570-4336358
Address:No.87 Building,Tianqian,Sidu Town
Beijing Mesochem Technology Co.,LTD
website:http://www.mesochem.com
Contact:0086-10-57862036
Address:2301, Floor 23, Building 9 Lippo Plaza, Yard 8 Ronghua Middle Road, ETDZ, Beijing, China
Yingkou Sanzheng Organic Chemical Co. Ltd.
Contact:+86-417-3638818
Address:25 Gengxinli Village, Daqing Road, Yingkou, Liaoning, China
Doi:10.1016/j.tetlet.2005.03.200
(2005)Doi:10.1016/S0022-328X(00)83521-4
(1977)Doi:10.1016/j.steroids.2013.07.009
(2013)Doi:10.1016/j.tetlet.2006.12.030
(2007)Doi:10.1021/jo00238a048
(1988)Doi:10.1021/jp0143591
(2002)