1
3
25
(
1
1
m, 12H); C NMR (100 MHz, CDCl ): δ 24.09, 33.68, 33.73,
4.2.11. N-(4-Fluorophenyl)formamide (3ka). White solid; 26.4
3
ACCEPTED MANUSCRIPT
o
1
19.34, 120.30, 127.08, 127.76, 134.48, 134.68, 145.69, 146.34,
59.19, 163.03.
mg (95% yield); mp 63-64 C; H NMR (400 MHz, CDCl ,
mixture of two rotamers, ratio: 0.7/1): δ 8.63 (brs, 1.4H), 8.35 (s,
3
13
1
H), 7.76 (brs, 1H), 7.53-7.50 (m, 2H), 7.11-6.99 (m, 4.8H); C
NMR (100 MHz, CDCl ): δ 115.88 (d, J = 22.0 Hz), 116.67 (d, J
23.0 Hz), 121.27 (d, J = 8.0 Hz), 121.98 (d, J = 8.0 Hz), 132.87
d, J = 3.0 Hz), 132.99 (d, J = 3.0 Hz), 159.28, 159.72 (d, J =
43.0 Hz), 160.56 (d, J = 243.0 Hz), 163.15.
4
.2.4. p-Butyl-formanilide (3da)
yield); H NMR (400 MHz, CDCl , mixture of two rotamers,
ratio: 1/0.8): δ 8.75 (brs, 1H), 8.64 (d, J = 11.4 Hz, 1H), 8.33 (s,
.8H), 7.84 (brs, 0.8H), 7.45 (d, J = 8.2 Hz, 1.6H), 7.16-7.11 (m,
.6H), 7.01 (d, J = 8.1 Hz, 2H), 2.60-2.54 (m, 3.6H), 1.61-1.52
.
Yellow liquid; 31.5 mg (89%
1
3
3
=
(
2
0
3
(
13
m, 3.6H), 1.39-1.28 (m, 3.6H), 0.94-0.89 (m, 5.4H); C NMR
100 MHz, CDCl ): δ 14.02, 22.34, 33.71, 35.04, 35.14, 119.15,
4.2.12. N-(3-Fluorophenyl)formamide (3la). White solid; 18.9
mg (68% yield); mp 56-57 C; H NMR (400 MHz, CDCl ,
o
1
(
3
3
1
1
20.19, 129.01, 129.67, 134.43, 134.64, 139.61, 140.26, 159.33,
63.18
mixture of two rotamers, ratio: 1/1.2): δ 8.73 (d, J = 11.1 Hz,
1H), 8.64 (brs, 1H), 8.38 (s, 1.2H), 7.71 (brs, 1.2H), 7.50 (d, J =
1
7e
10.6 Hz, 1.2H), 7.38-7.23 (m, 2.4H), 7.20 (d, J = 8.1 Hz, 1.2H),
4
.2.5. N-(4-Ethoxyphenyl)formamide (3ea). White solid; 31.4
13
o
1
6.98-6.72 (m, 4H); C NMR (100 MHz, CDCl
3
): δ 106.05 (d, J =
mg (95% yield); mp 66-67 C; H NMR (400 MHz, CDCl ,
mixture of two rotamers, ratio: 0.9/1): δ 8.52 (brs, 1.8H), 8.29 (s,
H), 7.84 (brs, 1H), 7.43 (d, J = 8.7 Hz, 2H), 7.02 (d, J = 8.6 Hz,
.8H), 6.88-6.82 (m, 3.8H), 4.03-3.97 (m, 3.8H), 1.42-1.37 (m,
.7H); C NMR (100 MHz, CDCl ): δ 14.86, 14.88, 63.78,
3.89, 114.87, 115.53, 121.57, 121.91, 129.60, 130.02, 156.10,
57.01, 159.29, 163.42.
3
2
1
5.0 Hz), 107.74 (d, J = 26.0 Hz), 111.73 (d, J = 21.0 Hz),
12.16 (d, J = 21.0 Hz), 114.18 (d, J = 3.0 Hz), 115.30 (d, J = 3.0
1
1
5
6
1
Hz), 130.39 (d, J = 9.0 Hz), 131.29 (d, J = 10.0 Hz), 138.42 (d, J
= 11.0 Hz), 138.52 (d, J = 10.0 Hz), 159.28, 162.60, 163.03 (d, J
13
3
=
244.0 Hz), 163.49 (d, J = 245.0 Hz).
23
4.2.13. N-(4-Bromophenyl) formamide (3ma). White solid; 29.6
o
1
23
mg (74% yield); mp 111-113 C; H NMR (400 MHz, CDCl
,
3
4
.2.6. N-(4-Methoxyphenyl)formamide (3fa). White solid; 28.1
mg (93% yield); mp 78-79 C; H NMR (400 MHz, CDCl ,
mixture of two rotamers, ratio: 1/1.1): δ 8.65 (brs, 1H), 8.52 (d, J
11.4 Hz, 1H), 8.28 (s, 1.1H), 8.01 (brs, 1.1H), 7.45 (d, J = 8.9
Hz, 2.2H), 7.04 (d, J = 8.8 Hz, 2H), 6.83-6.89 (m, 4.2H), 3.79 (s,
H), 3.77 (s, 3.3H); C NMR (100 MHz, CDCl ): δ 55.52, 55.60,
14.24, 114.93, 121.53, 121.95, 129.76, 130.16, 156.71, 157.61,
59.38, 163.46.
o
1
mixture of two rotamers, ratio: 0.7/1): δ 8.67 (brs, 1.4H), 8.37 (s,
3
1
1
1
H), 7.70 (brs, 1H), 7.52-7.39 (m, 5.4H), 6.99 (d, J = 8.4 Hz,
13
.4H); C NMR (100 MHz, CDCl ): δ 117.62, 118.39, 120.44,
3
=
21.66, 132.22, 132.92, 135.94, 136.02, 159.14, 162.55.
13
23
3
1
1
4.2.14. N-(4-Chlorophenyl)formamide (3na). White solid; 21.5
3
o
1
mg (69% yield); mp 98-99 C; H NMR (400 MHz, CDCl ,
3
mixture of two rotamers, ratio: 1/1.4): δ 8.65 (d, J = 11.3 Hz,
1
H), 8.38 (s, 1.4H), 8.06 (brs, 1H), 7.50 (d, J = 8.7 Hz, 2.8H),
4
.2.7. N-(3-Methoxyphenyl)formamide (3ga). Yellow liquid; 28.7
13
1
7.35-7.29 (m, 5.8H), 7.04 (d, J = 8.6 Hz, 2H); C NMR (100
MHz, CDCl ): δ 120.24, 121.33, 129.29, 130.00, 130.92, 135.39,
1
mg (95% yield); H NMR (400 MHz, CDCl , mixture of two
rotamers, ratio: 1/0.9): δ 8.70 (d, J = 11.3 Hz, 1H), 8.37 (s, 0.9H),
3
3
35.51, 159.06, 162.54.
8
8
3
1
1
.28 (brs, 1H), 7.46 (brs, 0.9H), 7.30-7.20 (m, 3H), 7.01 (d, J =
.0 Hz, 0.9H), 6.74-6.68 (m, 2.7H), 6.62 (s, 1H), 3.81 (s, 3H),
4.2.15. N-Formyl p-aminobenzoic acid methyl ester (3oa). White
solid; 18.6 mg (52% yield); mp 121-122 C; H NMR (400 MHz,
CDCl , mixture of two rotamers, ratio: 1/1.3): δ 8.87 (d, J = 9.9
Hz, 1H), 8.66 (brs, 1H), 8.44 (s, 1.3H), 8.03 (t, J = 9.2 Hz, 4.6H),
13
o
1
.80 (s, 2.7H); C NMR (100 MHz, CDCl ): δ 55.46, 55.52,
3
05.05, 105.99, 110.53, 110.69, 111.04, 112.14, 129.94, 130.74,
38.06, 138.19, 159.16, 160.29, 160.82, 162.69.
3
2
3
7.83 (brs, 1.3H), 7.65 (d, J = 8.4 Hz, 2.6H), 7.15 (d, J = 8.3 Hz,
4
.2.8. N-(2-Methoxyphenyl)formamide (3ha). White solid; 24.2
13
o
1
2H), 3.92 (s, 3H) , 3.91 (s, 3.9H); C NMR (100 MHz, CDCl
): δ
3
mg (80% yield); mp 81-82 C; H NMR (400 MHz, CDCl ,
mixture of two rotamers, ratio: 1/2.2): δ 8.74 (d, J = 11.6 Hz,
H), 8.45 (d, J = 1.6 Hz, 2.2H), 8.36 (dd, J = 8.0, 1.5 Hz, 2.2H),
.85 (brs, 2.2H), 7.73 (brs, 1H), 7.22-7.17 (d, J = 7.9 Hz, 1H),
.16-7.04 (m, 3H), 7.00-6.87 (m, 6.6H), 3.88 (s, 6.6H), 3.86 (s,
H); C NMR (100 MHz, CDCl ): δ 55.83, 110.15, 111.39,
16.74, 120.56, 121.17, 121.21, 124.38, 125.32, 126.31, 126.86,
47.89, 148.84, 158.85, 161.57.
3
5
1
2.24, 52.33, 117.31, 119.25, 126.26, 126.73, 131.03, 131.68,
41.09, 141.18, 159.28, 162.17, 166.46, 166.67.
1
7
7
3
1
1
17b
4.2.16. N-(4-Cyanophenyl)formamide (3pa). White solid; 12.9
o
1
mg (44% yield); mp 185-187 C; H NMR (400 MHz, DMSO-d ,
6
13
mixture of two rotamers, ratio: 0.33/1): δ 10.64 (brs, 1H), 10.52
(d, J = 10.3 Hz, 0.33H), 8.99 (d, J = 10.6 Hz, 0.33H), 8.37 (d, J =
1.3 Hz, 1H), 7.92-7.65 (m, 4.62H), 7.38 (d, J = 8.5 Hz, 0.66H);
3
13
24
C NMR (100 MHz, DMSO-d
6
): δ 105.44, 117.06, 118.93,
4
3
.2.9. N-(3,5-Dimethoxyphenyl)formamide (3ia). White solid;
o 1
119.30, 133.42, 133.80, 142.30, 160.42, 162.67.
1.2 mg (86% yield); mp 88-89 C; H NMR (400 MHz, CDCl ,
3
26
mixture of two rotamers, ratio: 1/0.7): δ 8.70 (d, J = 11.3 Hz,
4.2.17. N-(1-Naphthyl)formamide (3qa). White solid; 21.6 mg
o 1
1
6
2
4
9
1
H), 8.59 (brs, 1H), 8.34 (d, J = 1.9 Hz, 0.7H), 7.73 (brs, 0.8H),
.79 (d, J = 2.2 Hz, 1.4H), 6.28 (t, J = 2.1 Hz, 1H), 6.25 (t, J =
.2 Hz, 0.7H), 6.24 (d, J = 2.1 Hz, 2H), 3.78 (s, 6H), 3.77 (s,
.2H); C NMR (100 MHz, CDCl ): δ 55.49, 55.56, 96.85,
7.06, 97.24, 98.40, 138.66, 138.76, 159.36, 161.13, 161.75,
62.77.
(63% yield); mp 137-138 C; H NMR (400 MHz, CDCl3,
mixture of two rotamers, ratio: 1/0.4): δ 8.64-8.62 (m, 1.4H),
8.45 (brs, 1H), 8.03-7.99 (m, 1.4H), 7.91-7.85 (m, 2H), 7.79 (d, J
= 8.2 Hz, 1H), 7.73 (d, J = 8.2 Hz, 0.4H), 7.62-7.45 (m, 4.4H),
13
3
13
7.32 (d, J = 7.3 Hz, 1H); C NMR (100 MHz, CDCl ): δ 119.07,
3
120.59, 121.01, 121.53, 125.62, 125.80, 126.23, 126.30, 126.60,
1
26.78, 126.91, 127.09, 127.12, 127.88, 128.62, 128.92, 131.18,
4
2
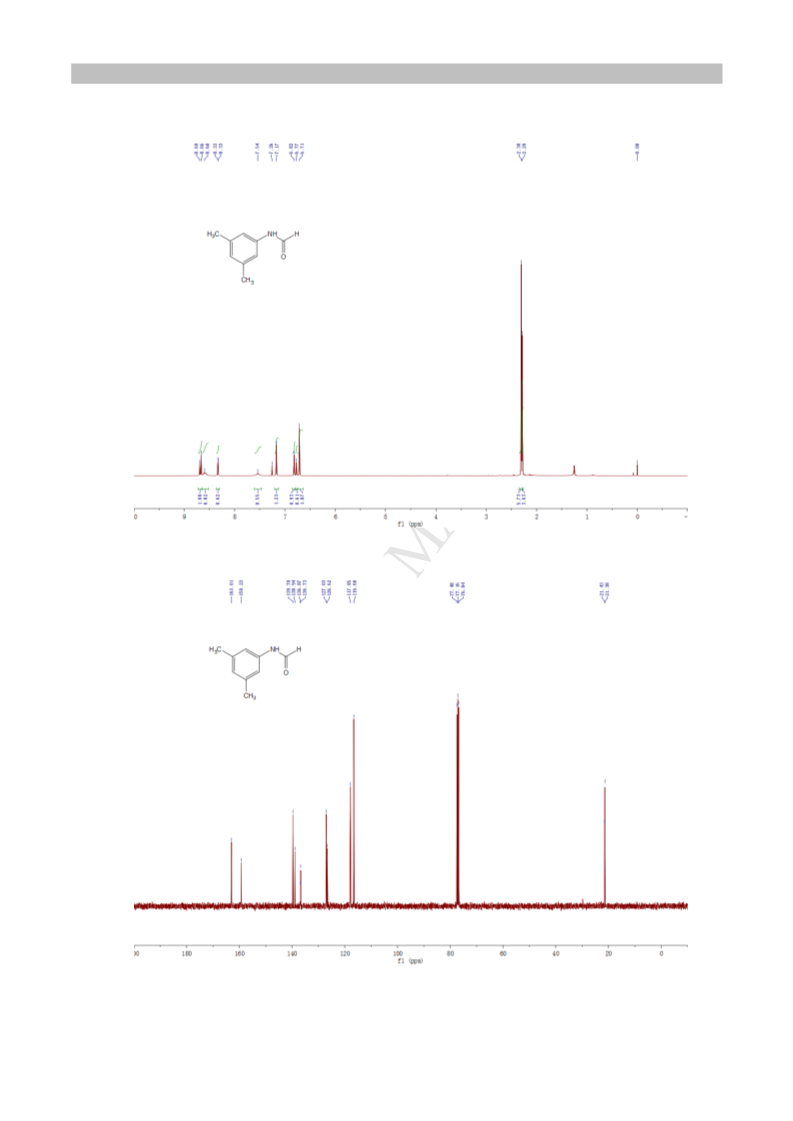
.2.10. Formic acid-(3,5-dimethyl-anilide) (3ja). White solid;
o
1
132.37, 134.18, 134.39, 159.90, 164.45.
6.3 mg (88% yield); mp 67-69 C; H NMR (400 MHz, CDCl ,
3
mixture of two rotamers, ratio: 1/0.6): δ 8.68 (d, J = 11.4 Hz,
4.2.18. N-(2-Naphthyl)formamide (3ra). White solid; 19.9 mg
o
1
1
7
6
1
1
H), 8.60 (brs, 1H), 8.33 (d, J = 1.8 Hz, 0.6H), 7.54 (brs, 0.6H),
(58% yield); mp 124-126 C; H NMR (400 MHz, CDCl3,
mixture of two rotamers, ratio: 1/1): δ 8.85 (d, J = 10.5 Hz, 1H),
8.84 (brs, 1H), 8.43 (s, 1H), 8.22 (s, 1H), 7.83-7.75 (m, 7H),
7.51-7.38 (m,
.17 (s, 1.2H), 6.82 (s, 1H), 6.77 (s, 0.6H), 6.71 (s, 2H), 2.30 (s,
13
H), 2.29 (s, 3.6H); C NMR (100 MHz, CDCl ): δ 21.38, 21.43,
3
16.60, 117.85, 126.62, 127.03, 136.73, 136.87, 138.94, 139.70,
59.23, 163.01.

 Gu, Da-Wei
Gu, Da-Wei
 Guo, Xun-Xiang
Guo, Xun-Xiang