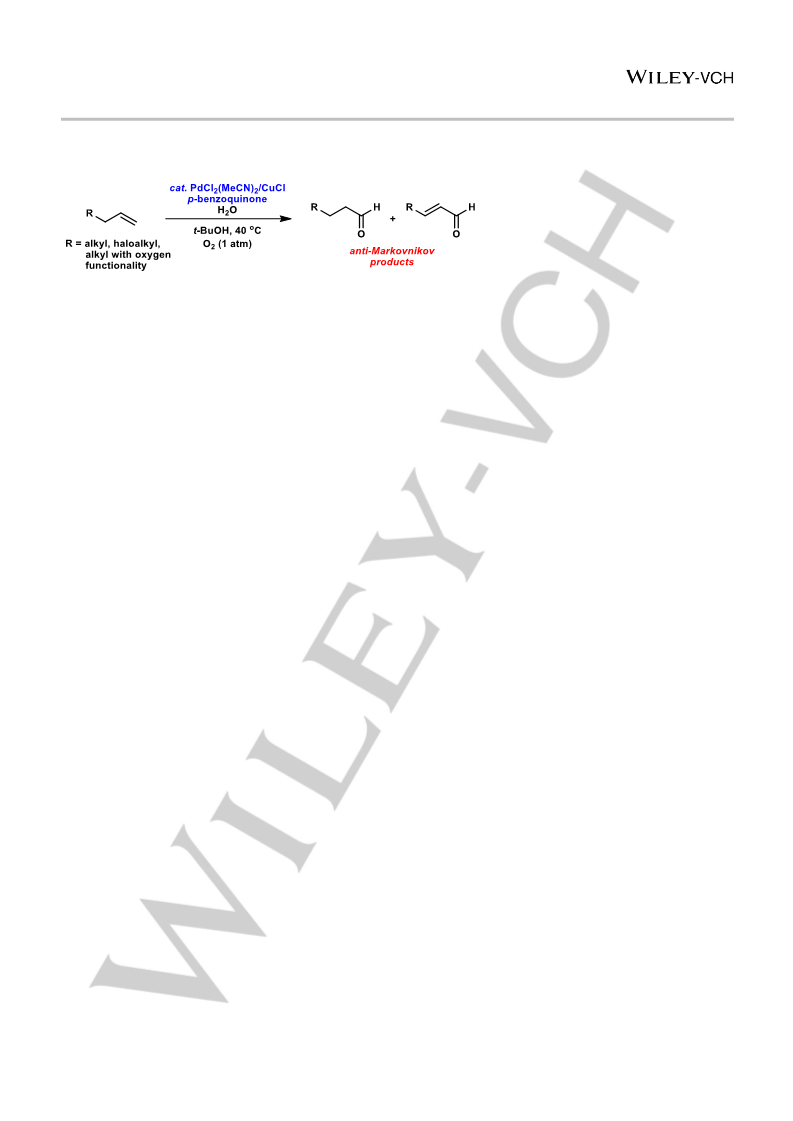
ChemCatChem
10.1002/cctc.202000472
FULL PAPER
4
,4-Dimethylpentanal (2d), 4,4-dimethyl-2-pentenal (3d), and 4,4-
Hz, 2.1 Hz, 1 H), 7.91 (d, J = 9.6 Hz, 1 H), 7.60 (t, J = 4.8 Hz, 1 H) 3.67 (t,
J = 6.3 Hz, 2 H), 2.67–2.60 (m, 2 H), 2.20–2.11 (m, 2 H). 13C{ H} NMR (75
1
dimethyl-2-pentanone (4d). Compound 2d, 3d, and 4d were isolated as
a mixture of corresponding 2,4-dinitrophenylhydrazone derivatives 2d', 3d',
and 4d'. Method B was applied. Purification by silica gel column
MHz, CDCl
28.8. HRMS (ESI): m/z calcd for C10
309.0374. Anal. Calcd for C10 11ClN
Found: C, 41.82; H, 3.80; N, 19.64.
3
) 150.3, 145.0, 138.0, 130.0, 128.9, 123.5, 116.4, 44.0, 29.8,
[M+Na]+ 309.0367, found
: C, 41.90; H, 3.87; N, 19.54.
H
4 4
11ClN O
O
4 4
chromatography (hexane/ethyl acetate = 10/1) afforded an orange solid
H
1
(89 mg, 0.29 mmol, 58% yield (2d'/3d'/4d' = 40%/8%/10%)). For 2d': H
NMR (300 MHz, CDCl
3
) 11.00 (s, 1 H), 9.09 (d, J = 2.4 Hz, 1 H), 8.28
(
1
dd, J = 9.6 Hz, 2.1 Hz, 1 H), 7.92 (d, J = 9.6 Hz, 1 H), 7.55 (t, J = 5.4 Hz,
5
-Chloropentanal (2h), 5-chloro-2-pentenal (3h), and 5-chloro-2-
H), 2.44–2.37 (m, 2 H), 1.52–1.46 (m, 2 H), 0.96 (s, 9 H). 13C{ H} NMR
1
pentanone (4h). Compounds 2h, 3h, and 4h were isolated as a mixture
of corresponding 2,4-dinitrophenylhydrazone derivatives 2h', 3h', and 4h'.
Method C was applied. Purification by silica gel column chromatography
(75 MHz, CDCl
3
) 153.3, 145.1, 137.6, 129.9, 128.7, 123.5, 116.5, 40.1,
+
3
0.3, 29.2, 28.3. HRMS (ESI): m/z calcd for C13
H N O
18 4 4
[M+H] 295.1406,
found 295.1398.
(
7
hexane/ethyl acetate = 5/1) afforded an orange solid (108 mg, 0.36 mmol,
2% yield (2h'/3h'/4h' = 55%/7%/10%)). For 2h': 1H NMR (300 MHz,
CDCl ) 11.01 (s, 1 H), 9.03 (d, J = 2.4 Hz, 1 H), 8.25 (dd, J = 9.6 Hz, 1.8
4,4-Dimethyl-2-pentenal (3d) and 4,4-dimethyl-2-pentanone (4d).
3
Compounds 3d and 4d were isolated as a mixture of corresponding 2,4-
dinitrophenylhydrazone derivatives 3d' and 4d'. Method B was applied.
Purification by silica gel column chromatography (hexane/ethyl acetate =
Hz, 1 H), 7.88 (d, J = 9.6 Hz, 1 H), 7.58 (t, J = 5.1 Hz, 1 H), 3.59 (t, J = 6.3
Hz, 2 H), 2.51–2.40 (m, 2 H), 1.93–1.77 (m, 4 H). 13C{ H} NMR (75 MHz,
1
CDCl
3
) 151.7, 145.0, 137.6, 129.8, 128.6, 123.3, 116.4, 44.5, 31.7, 31.6,
1
1
6
=
0/1) afforded an orange solid (103 mg, 0.35 mmol, 71% yield (3d'/4d' =
23.3. For 3h': H NMR (300 MHz, CDCl
3
) 11.09 (s, 1 H), 9.03 (d, J = 2.4
1
4%/7%)). For 3d': H NMR (300 MHz, CDCl
3
) 11.10 (s, 1 H), 9.12 (d, J
Hz, 1 H), 8.25 (dd, J = 9.6 Hz, 1.8 Hz, 1 H), 7.88 (d, J = 9.6 Hz, 1 H), 7.81
2.7 Hz, 1 H), 8.30 (ddd, J = 9.6 Hz, 2.4 Hz, 0.6 Hz, 1 H), 7.93 (d, J = 9.6
(d, J = 9.0 Hz, 1 H), 6.46–6.38 (m, 1 H), 6.30–6.20 (m, 1 H), 3.65 (t, J =
6.6 Hz, 2 H), 2.73 (q, J = 6.6 Hz, 2 H). 13C{ H} NMR (75 MHz, CDCl
1
Hz, 1 H), 7.77 (t, J = 4.2 Hz, 1 H), 6.28 (d, J = 1.2 Hz, 1 H), 6.27 (s, 1 H),
3
)
1
1
1
.13 (s, 9 H). 13C{ H} NMR (75 MHz, CDCl
3
) 156.5, 150.8, 144.8, 137.9,
149.3, 145.0, 139.9, 137.6, 130.0, 128.9, 128.6, 123.2, 116.5, 42.9, 35.6.
1
1
29.9, 129.1, 123.5, 121.8, 116.5, 34.2, 29.0. For 4d': H NMR (300 MHz,
For 4h': H NMR (300 MHz, CDCl
3
) 11.01 (s, 1 H), 9.03 (d, J = 2.4 Hz, 1
CDCl
3
) 11.01 (s, 1 H), 9.11 (d, J = 2.7 Hz, 1 H), 8.29 (dd, J = 9.6 Hz, 2.4
H), 8.25 (dd, J = 9.6 Hz, 1.8 Hz, 1 H), 7.88 (d, J = 9.6 Hz, 1 H), 3.65 (t, J =
6.6 Hz, 2 H), 2.61 (t, J = 7.2 Hz, 2 H), 2.18–2.13 (m, 2 H), 2.08 (s, 3 H).
C{ H} NMR (75 MHz, CDCl ) 156.4, 144.6, 137.6, 129.9, 128.6, 123.3,
3
Hz, 1 H), 7.94 (d, J = 9.6 Hz, 1 H), 2.34 (s, 2 H), 2.10 (s, 3 H), 1.03 (s, 9
H). 13C{ H} NMR (75 MHz, CDCl
1
13
1
3
) 157.0, 145.1, 137.6, 130.0, 129.0,
1
C
23.5, 116.5, 52.3, 32.0, 29.9, 18.5. HRMS for 3d' (ESI): m/z calcd for
116.2, 44.2, 35.9, 28.5, 16.3. HRMS for 2h' and 4h' (ESI): m/z calcd for
[M+H]+ 293.1250, found 293.1249. Anal. Calcd for 3d'
C H13ClN O
11 4 4
[M+Na] 323.0523, found 323.0514.
+
H N O
13 16 4 4
13 16 4 4
C H N O : C, 53.42; H, 5.52; N, 19.17. Found: C, 53.04; H, 5.42; N,
18.98.
4-Bromobutanal (2i). Compound 2i was isolated as a pure (E)-2,4-
dinitrophenylhydrazone derivative 2i'. Method C was applied. H O (13.5
2
4
-Phenylbutanal (2e) and 4-phenyl-2-butanone (4e). Compounds 2e
mixture of corresponding 2,4-
dinitrophenylhydrazone derivatives 2e' and 4e'. Method A was applied.
Cy P) Rh(H)Cl (29.4 mg, 0.04 mmol) was used. Purification by silica gel
L, 0.75 mmol) was used. Purification by silica gel column chromatography
and 4e were isolated as
a
(hexane/ethyl acetate = 5/1) afforded a yellow solid (111 mg, 0.34 mmol,
67% yield). 1H NMR (300 MHz, CDCl
) 11.07 (s, 1 H), 9.12 (d, J = 2.7
3
(
3
3
2
Hz, 1 H), 8.31 (dd, J = 9.3 Hz, 2.1 Hz, 1 H), 7.91 (d, J = 9.6 Hz, 1 H), 7.58
column chromatography (hexane/ethyl acetate = 10/1) afforded an orange
solid (90 mg, 0.28 mmol, 55% yield (2e'/4e' = 45%/10%)). The spectral
data for 2e' was in accordance with those reported in the literature.[24] For
(t, J = 4.8 Hz, 1 H) 3.53 (t, J = 6.3 Hz, 2 H), 2.67–2.61 (m, 2 H), 2.28–2.19
(m, 2 H). 13C{ H} NMR (75 MHz, CDCl
1
3
) 150.1, 145.0, 138.0, 130.0,
128.9, 123.5, 116.4, 32.4, 31.0, 28.9. HRMS (ESI): m/z calcd for
1
[M+H]+ 331.0042, found 331.0053. Anal. Calcd for
: C, 36.27; H, 3.35; N, 16.92. Found: C, 36.53; H, 3.31; N,
4
8
5
e': H NMR (300 MHz, CDCl
3
) 10.96 (s, 1 H), 9.04 (d, J = 2.7 Hz, 1 H),
C
10
H
11BrN
4
O
4
.22 (dd, J = 9.5 Hz, 2.7 Hz, 1 H), 7.83 (d, J = 9.6 Hz, 1 H), 7.28–7.14 (m,
H), 2.94 (t, J = 7.2 Hz, 2 H), 2.68 (t, J = 7.5 Hz, 2 H), 2.02 (s, 3 H). 13C{ H}
C
10
H11BrN
4
O
4
1
17.12.
NMR (75 MHz, CDCl
3
) 157.1, 145.0, 140.7, 137.7, 129.8, 128.7, 128.4,
126.0, 123.4, 116.4, 40.4, 32.2, 16.2.
5-Bromopentanal (2j). Compound 2j was isolated as 2,4-
dinitrophenylhydrazone derivatives 2j'. Method C was applied. H O (13.5
2
6-Oxohexenoic acid benzyl ester (2f) and 5-oxohexenoic acid benzyl
L, 0.75 mmol) was used. Purification by silica gel column chromatography
ester (4f). Compounds 2f and 4f were isolated as
a
mixture of
(hexane/ethyl acetate = 5/1) afforded a yellow solid (92 mg, 0.27 mmol,
corresponding 2,4-dinitrophenylhydrazone derivatives 2f' and 4f'. Method
A was applied. BQ (54 mg, 0.50 mmol) was used. Purification by silica
gel column chromatography (hexane/ethyl acetate = 5/1) afforded an
54% yield). 1H NMR (300 MHz, CDCl
3
) 11.04 (s, 1 H), 9.12 (d, J = 2.4
Hz, 1 H), 8.30 (ddd, J = 9.6 Hz, 2.7 Hz, 0.6 Hz, 1 H), 7.93 (d, J = 9.6 Hz, 1
H), 7.55 (t, J = 4.8 Hz, 1 H) 3.47 (t, J = 6.3 Hz, 2 H), 2.52–2.45 (m, 2 H),
1
2.03–1.94 (m, 2 H), 1.89–1.76 (m, 2 H). 13C{ H} NMR (75 MHz, CDCl
1
orange oil (151 mg, 0.38 mmol, 76% yield (2f'/4f' = 54%/22%)). For 2f': H
3
)
NMR (300 MHz, CDCl
3
) 11.91 (s, 1 H), 8.98 (d, J = 2.1 Hz, 1 H), 8.17
151.3, 145.1, 137.9, 130.2, 123.5, 116.5, 33.1, 31.9, 31.6, 24.6. HRMS
+
(dd, J = 9.6 Hz, 2.1 Hz, 1 H), 7.81 (d, J = 9.6 Hz, 1 H), 7.44 (t, J = 5.1 Hz,
(ESI): m/z calcd for C11
Calcd for C11 13BrN
3.82; N, 15.99.
H
13BrN
4
O
4
[M+H] 345.0199, found 345.0198. Anal.
1
H), 7.26 (s, 5 H), 5.04 (s, 2 H), 2.43–2.34 (m, 4 H), 1.74–1.57 (m, 4 H).
C{ H} NMR (75 MHz, CDCl
H
4 4
O : C, 38.28; H, 3.80; N, 16.23. Found: C, 38.37; H,
1
3
1
3
) 173.0, 151.9, 144.9, 137.5, 135.8, 129.7,
1
28.5, 128.4, 128.11, 128.07, 128.0, 123.2, 116.3, 66.1, 37.9, 33.7, 25.4,
1
24.2. For 4f': H NMR (300 MHz, CDCl
3
) 11.91 (s, 1 H), 8.98 (d, J = 2.1
Theoretical Calculations. Calculations were carried out using density
Hz, 1 H), 8.13 (dd, J = 13.5 Hz, 2.1 Hz, 1 H), 7.81 (d, J = 9.6 Hz, 1 H), 7.26
s, 5 H), 5.04 (s, 2 H), 2.43–2.34 (m, 4 H), 1.97 (s, 3 H), 1.74–1.57 (m, 2
functional theory (DFT)-optimized geometries using the Gaussian 09
Revision E.01[25] implementation of B3LYP [Becke three-parameter
(
13
1
H). C{ H} NMR (75 MHz, CDCl
3
) 172.8, 156.9, 144.9, 137.4, 135.7,
exchange functional (B3) and the Lee–Yang–Parr correlation functional
1
3
4
29.8, 128.8, 128.4, 128.11, 128.07, 128.03, 127.99, 123.1, 116.2, 66.1,
consisted of the combination of LANL2DZ for Pd, 6-311G(d) for Cu, and
3.2, 32.0, 20.9, 15.9. HRMS (ESI): m/z calcd for C19
H N O
20 4 6
[M+H]+
01.1461, found 401.1464.
6
-31G(d) for other atoms. The frequency calculation was performed using
M06[ and the basis set consisted of the combination of the Stuttgart-
Dresden-Bonn energy-consistent pseudopotential (SDD)[30] for Pd, 6-
311G(d) for Cu, and 6-311+G(d,p) for other atoms. The SMD method[31]
4-Chlorobutanal (2g). Compound 2g was isolated as 2,4-
dinitrophenylhydrazone derivatives 2g'. Method
C
was applied.
Purification by silica gel column chromatography (hexane/ethyl acetate =
was applied to incorporate a solvent effect (t-BuOH). Frequency
calculations on optimized species established that the energy minima
possessed only real frequencies and the transition states possessed a
1
5/1) afforded a yellow solid (91 mg, 0.32 mmol, 63% yield). H NMR (300
MHz, CDCl ) 11.06 (s, 1 H), 9.11 (d, J = 2.4 Hz, 1 H), 8.30 (dd, J = 9.6
3
8
This article is protected by copyright. All rights reserved.

 Komori, Saki
Komori, Saki
 Yamaguchi, Yoshiko
Yamaguchi, Yoshiko
 Murakami, Yuka
Murakami, Yuka
 Kataoka, Yasutaka
Kataoka, Yasutaka
 Ura, Yasuyuki
Ura, Yasuyuki