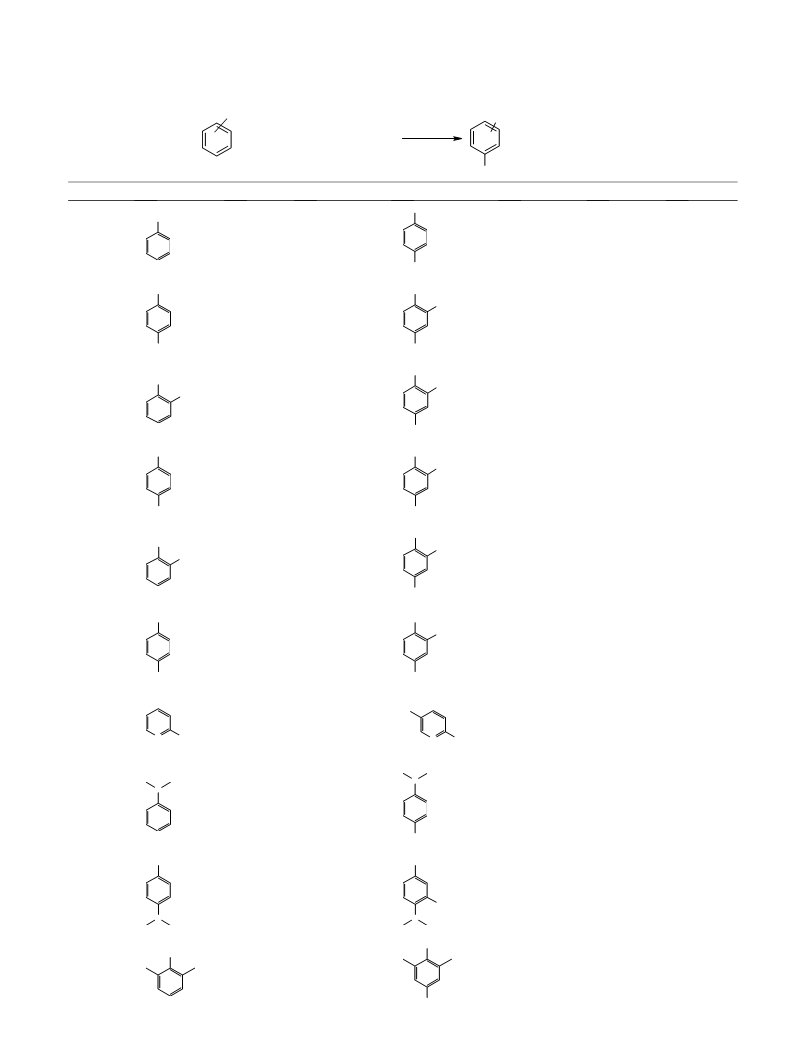
M. M. Heravi et al. / Tetrahedron Letters 46 (2005) 8959–8963
8963
hedron Lett. 2005, 46, 4456; (c) Ghorbani-Vaghei, R.;
Jalili, H. Synthesis 2005, 7, 1099.
10. Grag, D.; Kothans, S. J. Chem. Sci. 2004, 116, 333.
11. Heravi, M. M.; Ajami, D.; Nooshabadi, M. A. Iran J.
Chem. Eng. 1999, 18, 88.
12. Beheshtiha, Y. Sh.; Heravi, M. M.; Sarmad, N.; Hekmat-
Shoar, R. J. Chem. Res. 2001, 79.
13. Dehkordi, M. B.; Heravi, M. M.; Khosrofar, P.; Ziafati,
A.; Ghassemzadeh, M. Phosphorus, Sulfur, Silicon 2004,
179, 2595.
HMTAB (0.5 mmol). The reaction mixture was stirred
until the yellow colour of the complex disappeared.
The reaction was monitored by TLC. After completion
of the reaction the mixture was filtered and the solid resi-
due was washed with dichloromethane. The dichloro-
methane solution was washed successively with dilute
(3%) aqueous sodium carbonate solution and water.
The dried (MgSO4) organic fraction was concentrated
to afford the brominated compound in excellent yields.
14. Dehkordi, M. B.; Heravi, M. M.; Ziafati, A.; Khosrofar,
P.; Ghassemzadeh, M. Phosphorus, Sulfur, Silicon 2004,
179, 1493.
15. (a) Garg, D.; Kathari, S. J. Chem. Sci. 2004, 116, 33; (b)
Sharma, P. K. Indian J. Chem. Soc. 2004, 81, 291; (c)
Kathari, S.; Sharma, P. K.; Banerji, K. K. Indian J. Chem.
Soc. 2003, 80, 431.
References and notes
1. Felton, L. C.; McLaughlin, C. B. J. Org. Chem. 1947, 12,
298–302.
2. Zsolnai, T. Biochem. Pharmacol. 1960, 5, 1–19.
3. For example: UlmannÕs Encyclopedia of Industrial Chem-
istry, 6th ed.; Wiley: Weinheim, 1998.
16. Eia, G.; Hassel, O. Acta Chem. Scand. 1956, 10, 139.
17. Paul, S.; Varinder, G.; Gupta, R. Synth. Commun. 2003,
33, 1917.
4. De la Mare, P. B. Electrophilic Halogenation; Cambridge
University Press: Cambridge, 1976; Chapter 5.
5. Larock, R. C. Comprehensive Organic Transformation;
VCH: New York, 1989; p 315.
6. Dewkar, G. K.; Narina, S. V.; Sudalai, A. Org. Lett. 2003,
5, 4501–4504.
book of Chemistry and Physics, 69th ed.; 1988–1989; c-67;
(c) Edkins, R. P.; Linnell, W. H. Quart. J. Pharm.
Pharmacol. 1936, 9, 75–109; (d) Montagne, P. J. Rec.
Trav. Chim. 1922, 41, 703–721; (e) Joshi, G. G.; Shah, N.
M. J. Indian Chem. Soc. 1954, 31, 220–222.
7. Vyas, P. V.; Bhatt, A. K.; Ramachanraiah, G.; Bedekar,
A. V. Tetrahedron Lett. 2003, 44, 4085–4088.
19. (a) Heravi, M. M.; Ajami, D.; Aghapoor, K.; Ghassem-
zadeh, M. Chem. Commun. 1999, 833; (b) Heravi, M. M.;
Ajami, D.; Aghapoor, K.; Ghassemzadeh, M. Synthesis
1999, 339; (c) Heravi, M. M.; Azemian, P. K.; Oskooie, H.
A.; Ghassemzadeh, M. J. Chem. Res. 2005, 2, 105.
20. (a) Pareek, A.; Kothari, S.; Banerji, K. K. Indian J. Chem.
1996, 35B, 970; (b) Gangwani, H.; Sharma, P. K. J. Chem.
Res. (S) 1999, 1, 854.
21. Pareek, A.; Varshney, S.; Banerji, K. K. React. Kinet.
Catal. Lett. 1997, 60, 127.
22. Choudhary, K.; Suri, D.; Kothari, S.; Banerji, K. K. J.
Phys. Org. Chem. 2000, 13, 283.
8. For recent examples of chlorination and bromination
protocols, see: (a) Smith, K.; Butters, M.; Paget, W. E.;
Goubet, D.; Fromentin, E.; Nay, B. Green Chem. 1999, 1,
83; (b) Barhate, N. B.; Gajare, A. S.; Wakharkar, R. D.;
Bedekar, A. V. Tetrahedron 1999, 55, 11127; (c) Rothe-
berg, G.; Clark, J. H. Org. Proc. Res. Dev. 2000, 4, 270; (d)
Bora, U.; Bose, G.; Chaudhuri, M. K.; Dhar, S. S.;
Gopinath, R.; Khan, A.; Patel, B. K. Org. Lett. 2000, 2,
247; (e) Narender, N.; Mohan, K. V. V. K.; Reddy, R. V.;
Srinivasa, P.; Kulkarni, S. J.; Raghavan, K. V. J. Mol.
Catal. A. 2003, 192, 73.
23. Bailey, R. S.; Drake, G. W.; Hanks, T. W.; Hook, L. L.;
Pennington, T. J. Chem. Soc., Perkin Trans. 2 1997,
2773.
9. (a) Bagheri, M.; Azizi, N.; Saidi, M. R. Can. J. Chem.
2005, 83, 143; (b) Gnaim, J. M.; Sheldon, R. A. Tetra-

 Heravi, Majid M.
Heravi, Majid M.
 Abdolhosseini, Nafiseh
Abdolhosseini, Nafiseh
 Oskooie, Hossein A.
Oskooie, Hossein A.