FULL PAPERS
Catalytic One-Pot Oxetane to Carbamate Conversions
References
[23] D. J. Darensbourg, A. I. Moncada, Macromolecules
2010, 43, 5996.
[24] J. Rintjema, W. Guo, E. Martin, E. C. Escudero-Adµn,
[1] O. A. Davis, J. A. Bull, Synlett 2015, 26, A–F.
[2] G. Wuitschik, E. M. Carreira, B. Wagner, H. Fischer, I.
Parrilla, F. Schuler, M. Rogers-Evans, K. Müller, J.
Med. Chem. 2010, 53, 3227.
[3] J. A. Burkhard, G. Wuitschik, M. Rogers-Evans, K.
Müller, E. M. Carreira, Angew. Chem. 2010, 122, 9236;
Angew. Chem. Int. Ed. 2010, 49, 9052.
[4] M. D’Auria, R. Racioppi, Molecules 2013, 18, 11384.
[5] D. J. Darensbourg, A. I. Moncada, W. Choi, J. H. Rei-
benspies, J. Am. Chem. Soc. 2008, 130, 6523.
[6] M. Helou, O. Miserque, J.-M. Brusson, J.-F. Carpentier,
S. M. Guillaume, Chem. Eur. J. 2010, 16, 13805.
[7] B. Schulte, C. A. Dannenberg, H. Keul, M. J. Mçller, J.
Polym. Sci. Part A: Polym. Chem. 2013, 51, 1243.
[8] M. McLaughlin, R. Yazaki, T. C. Fessard, E. M. Car-
reira, Org. Lett. 2014, 16, 4070.
[9] P. B. Brady, E. M. Carreira, Org. Lett. 2015, 17, 3350.
[10] K. F. Morgan, I. A. Hollingsworth, J. A. Bull, Chem.
Commun. 2014, 50, 5203.
[11] C. A. Malapit, A. R. Howell, J. Org. Chem. 2015, 80,
8489.
[12] A. F. Stepan, K. Karki, W. S. McDonald, P. H. Dorff,
J. K. Dutra, K. J. DiRico, A. Won, C. Subramanyam,
I. V. Efremov, C. J. OꢁDonnell, C. E. Nolan, S. L.
Becker, L. R. Pustilnik, B. Sneed, H. Sun, Y. Lu, A. E.
Robshaw, D. Riddell, T. J. OꢁSullivan, E. Sibley, S. Ca-
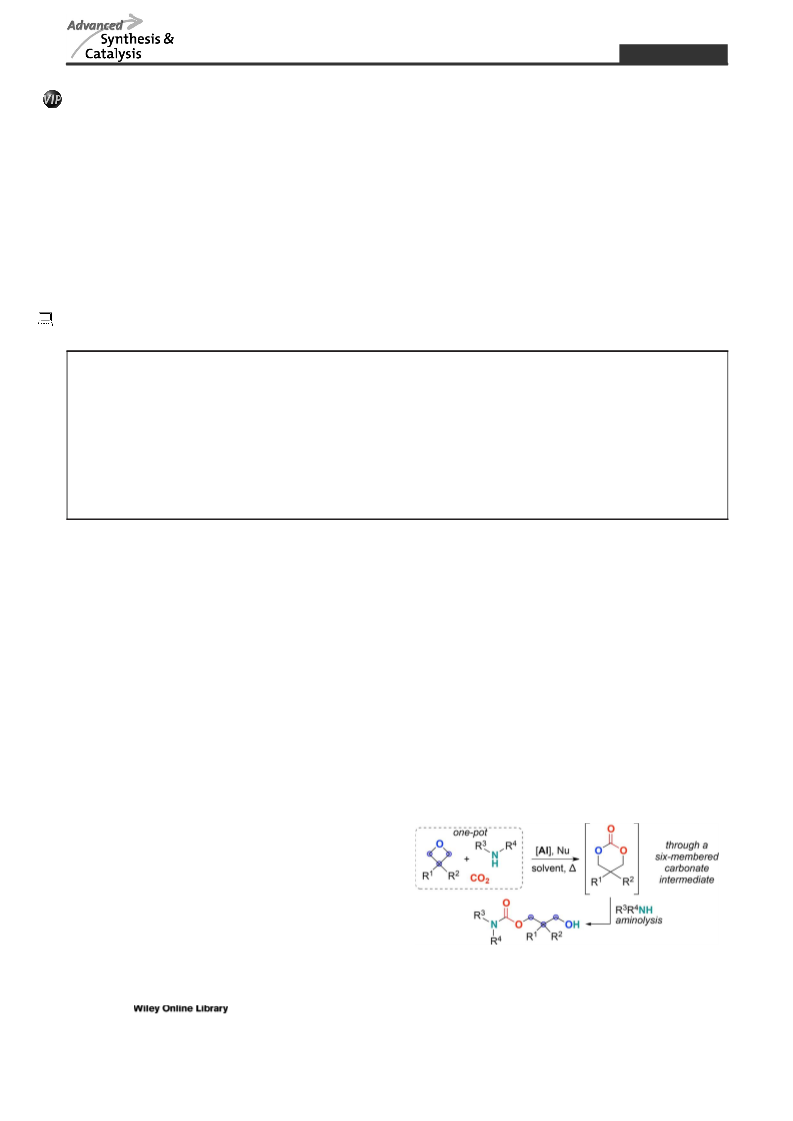
petta, K. Atchison, A. J. Hallgren, E. Miller, A. Wood,
R. S. Obach, J. Med. Chem. 2011, 54, 7772.
[13] M. C. Wani, H. L. Taylor, M. E. Wall, P. Caggon, A. T.
McPhall, J. Am. Chem. Soc. 1971, 93, 2325.
[14] M. Cokoja, C. Bruckmeier, B. Rieger, W. A. Herrmann,
F. E. Kühn, Angew. Chem. 2011, 123, 8662; Angew.
Chem. Int. Ed. 2011, 50, 8510.
A. W. Kleij, Chem. Eur. J. 2015, 21, 10754.
[25] H.-W. Engels, H.-G. Pirkl, R. Albers, R. W. Albach, J.
Krause, A. Hoffmann, H. Casselmann, J. Dormish,
Angew. Chem. 2013, 125, 9596; Angew. Chem. Int. Ed.
2013, 52, 9422.
[26] T. R. Fukuto, Environ. Health Perspect. 1990, 87, 245.
[27] A. K. Ghosh, M. Brindisi, J. Med. Chem. 2015, 58,
2895.
[28] S. Ishii, M. Zhou, Y. Yoshida, H. Noguchi, Synth.
Commun. 1999, 29, 3207.
[29] V. Laserna, G. Fiorani, C. J. Whiteoak, E. Martin, E. C.
Escudero-Adµn, A. W. Kleij, Angew. Chem. 2014, 126,
10584; Angew. Chem. Int. Ed. 2014, 53, 10416.
[30] C. J. Whiteoak, N. Kielland, V. Laserna, E. C. Escu-
dero-Adµn, E. Martin, A. W. Kleij, J. Am. Chem. Soc.
2013, 135, 1228.
[31] L. PeÇa Carrodeguas, J. Gonzµlez-Fabra, F. Castro-
Gómez, C. Bo, A. W. Kleij, Chem. Eur. J. 2015, 21,
6115.
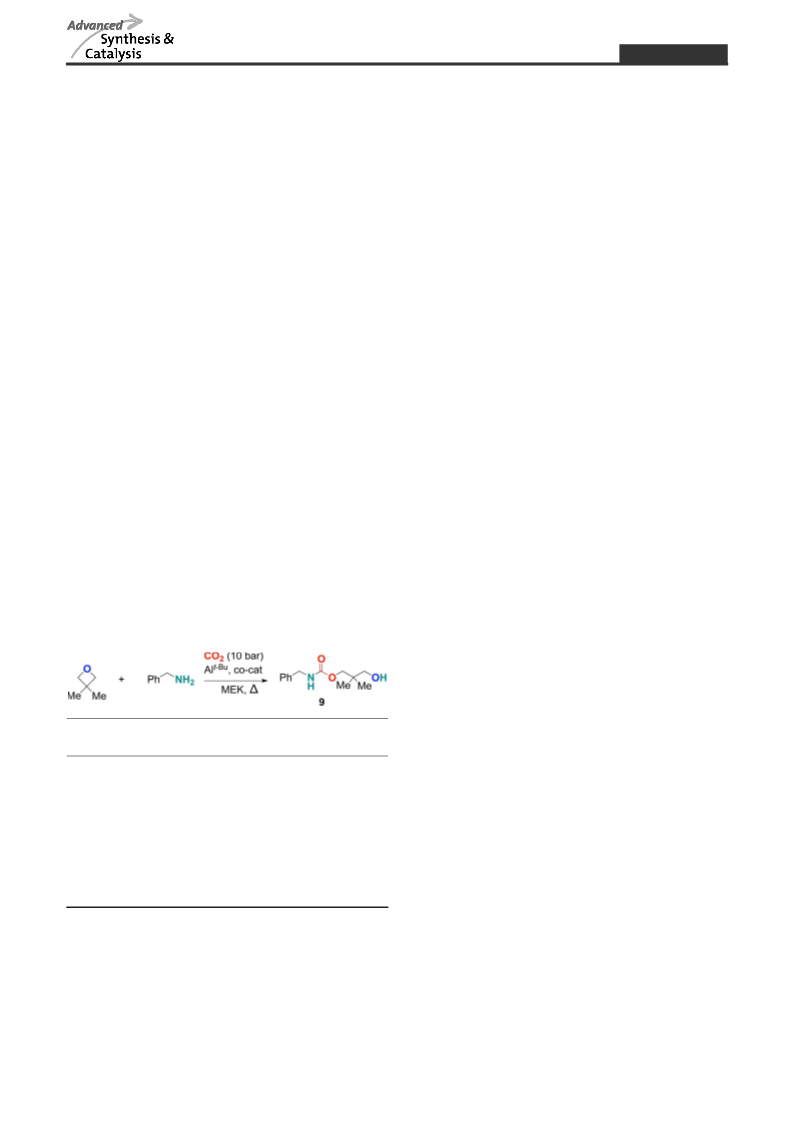
[32] Conditions used in this approach: 2.5 mol% [Alt-Bu],
5.0 mol% TBAB, 758C, 10 bar.
[33] Please note that CO2 and alkylamines may give rise to
carbamate nucleophiles themselves. However, when we
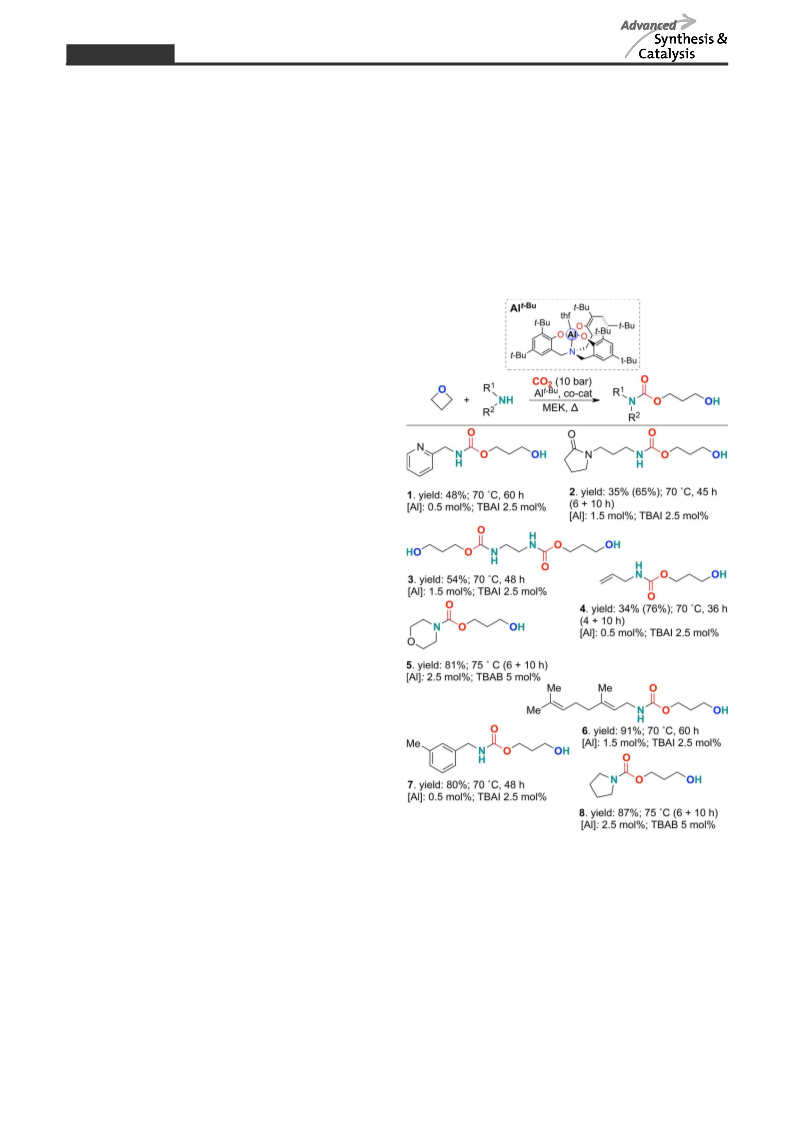
carried out the synthesis of compound 7 (Figure 1)
based on the simplest oxetane in the absence of TBAI,
we found only the amino alcohol product (7% yield by
NMR; mesitylene as internal standard) with traces of
the carbamate product 7. Note that carbamate forma-
tion in the absence of an external nucleophile using
3,3’-dimethyloxirane (Table 1, entries 2 and 3) does not
lead to any product formation. This clearly demon-
strates that carbamate nucleophiles possibly derived
from amines and CO2 are incompetent towards oxetane
ring-opening under the reaction conditions used.
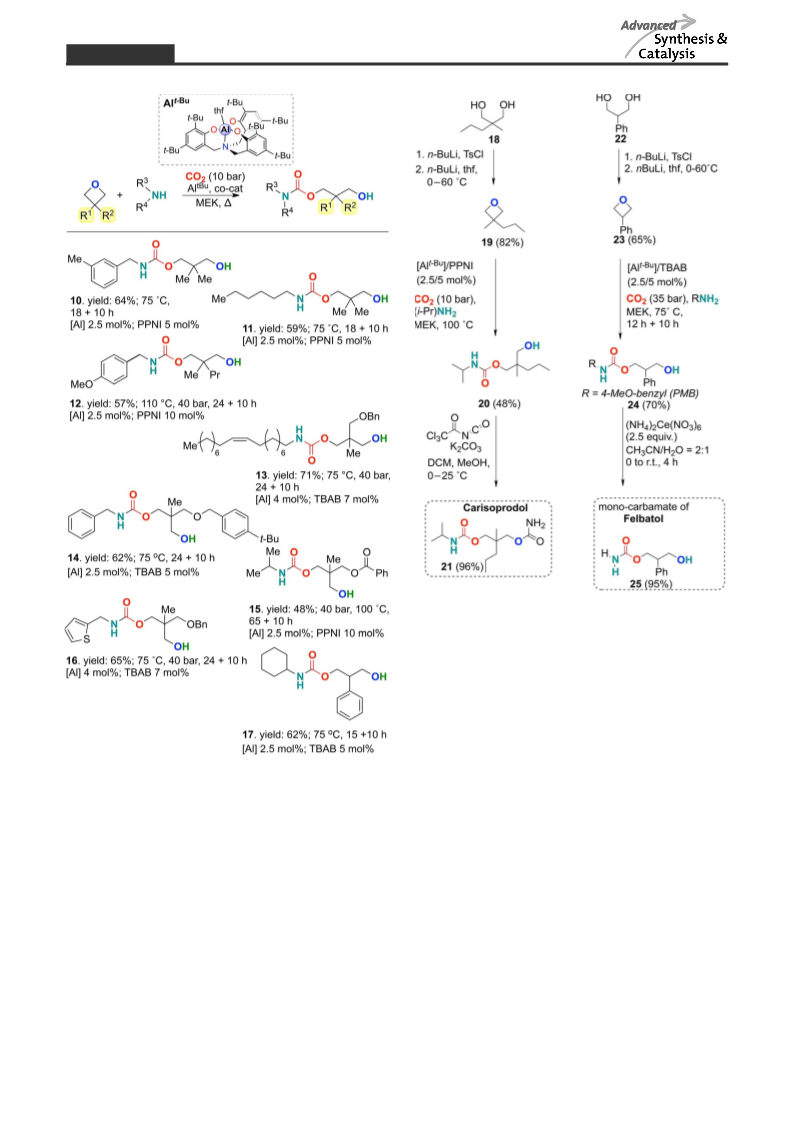
[34] Note that Darensbourg et al. also observed that 3,3’-
substituted oxetanes show much slower kinetics in their
coupling with CO2, see: D. J. Darensbourg, A. I. Mon-
cada, S.-H. Wei, Macromolecules 2011, 44, 2568.
[35] P. Picard, D. Leclercq, J.-P. Bats, J. Moulines, Synthesis
1981, 550.
[15] M. Aresta, A. Dibenedetto, A. Angelini, Chem. Rev.
2014, 114, 1709.
[16] C. J. Whiteoak, N. Kielland, A. W. Kleij, Adv. Synth.
Catal. 2013, 355, 2115.
[17] M. Peters, B. Kçhler, W. Kuckshinrichs, W. Leitner, P.
Markewitz, T. E. Müller, ChemSusChem 2011, 4, 1216.
[18] C. Martín, G. Fiorani, A. W. Kleij, ACS Catal. 2015, 5,
1353.
[19] M. Taherimehr, P. P. Pescarmona, J. Appl. Polym. Sci.
2014, 131, 41141.
[36] P. A. Hedin, R. C. Gueldner, A. C. Thompson, Anal.
Chem. 1970, 42, 403.
[37] Y. Ichikawa, M. Osada, I. I. Ohtani, M. Isobe, J. Chem.
Soc. Perkin Trans. 1 1997, 1449.
[38] P. Kocovsky, Tetrahedron Lett. 1986, 27, 5521.
[39] H. Adams, J. C. Anderson, S. Peace, A. M. K. Pennell,
J. Org. Chem. 1998, 63, 9932.
[20] X.-B. Lu, D. J. Darensbourg, Chem. Soc. Rev. 2012, 41,
1462.
[21] B. R. Buckley, A. P. Patel, K. G. Upul Wijayantha, Eur.
J. Org. Chem. 2015, 474.
[22] D. J. Darensbourg, A. Horn Jr, A. I. Moncada, Green
Chem. 2010, 12, 1376.
[40] F. M. Berger, B. J. Ludwig, U.S. Patent 2,937,119, 1960;
Chem. Abstr. 1960, 54, 18367.
Adv. Synth. Catal. 2016, 358, 1602 – 1607
ꢀ 2016 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
1607

 Guo, Wusheng
Guo, Wusheng
 Laserna, Victor
Laserna, Victor
 Rintjema, Jeroen
Rintjema, Jeroen
 Kleij, Arjan W.
Kleij, Arjan W.