Organic Process Research & Development 2001, 5, 452−455
Scaling Up of Dihydropyridine Ester Synthesis by Using Aqueous Hydrotrope
Solutions in a Continuous Microwave Reactor
Bhushan M. Khadilkar* and Virendra R. Madyar
Department of Chemical Technology, UniVersity of Mumbai, Matunga, Mumbai 400 019, India
Abstract:
carry out reactions under microwave exposure in a continu-
ous flow using CMR. By this we are avoiding the use of
costly and hazardous organic solvent systems by replacing
them with a reusable aqueous hydrotrope solution as a new
generation “green” solvent system.
We report here the scaling up of clinically important dihydro-
pyridine by using a continuous microwave reactor (CMR). We
also report the use of aqueous hydrotrope solution as a cheap,
safe and “green” alternative to organic solvent to carry out
homogeneous reactions under microwave heating. We have
studied different aqueous hydrotrope solutions for the reaction
in batch as well as continuous-flow process.
16
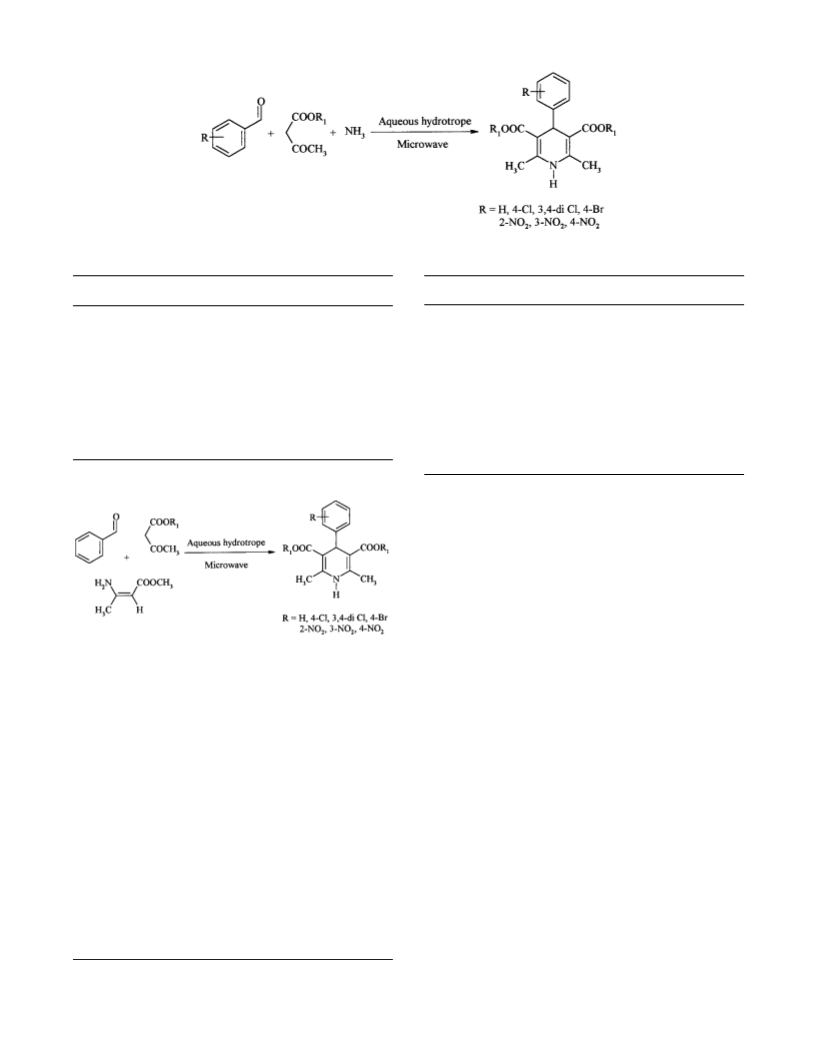
We describe here the Hantzsch ester synthesis using the
hydrotrope solution. The mixture of benzaldehyde, methyl
3-aminocrotonate, and ethyl/methyl acetoacetate was solu-
bilized in the aqueous hydrotrope solution of sodium
p-toluene sulphonate and circulated through the microwave
oven cavity to obtain dihydropyridines in high yield and of
Introduction
1
7-19
The utility of microwave irradiation to carry out organic
reactions has now become a regular feature. This is evident
from the number of reviews1 published on the use of
microwave technology for carrying out organic reactions.
From the future point of view of microwave technology the
question that now needs to be answered is how to carry out
large-scale reactions by employing microwaves and to
understand the nature of the microwave effect. In 1994,
excellent quality. Dihydropyridines
are an important
class of calcium channel blocker and have extensive clinical
use.
-8
Results and Discussion
We have used for the first time an aromatic hydrotrope
solution system such as 50% sodium p-toluene sulphonate
aqueous solution (NaPTSA), 40% sodium cumene sulphonate
aqueous solution (NaCuS), and 20% sodium p-xylene
sulphonate (NaXS) aqueous solution to carry out Hantzsch
ester synthesis to give 4-aryl-1,4-dihydropyridines under
microwave exposure.
We studied two routes for Hantzsch ester synthesis of
4-aryl-1,4-dihydropyridines under microwave exposure. Ini-
tially each route was studied and optimized for a batch
process in 50% NaPTSA under microwave exposure (Figure
9
Strauss and co-workers demonstrated the use of a continuous
microwave reactor for scaling up organic reactions in
homogeneous conditions. However, later on, the reports were
very scanty. Now attention is focused on the aspect of scaling
up of reactions using microwave technology. The research
groups of Kabza et al., Loupy et al., E. Esveld et al.,
and Hamelin et al.13 have published promising results.
Keeping this in mind we have developed a continuous
microwave reactor (CMR) for carrying out reactions on a
larger scale using a domestic microwave oven. By using this
reactor, about 500 mL of the reaction mixture can be
processed within a short time. We are developing new
10
11
12
1
). We also studied the reaction in 40% NaCuS and 20%
NaXS hydrotrope solution. We found that the yields of DHPs
were higher in 50% NaPTSA than in the other two hydro-
trope solution.
technology by making use of an aqueous hydrotrope solu-
as an homogeneous reaction medium, which is a
reliable, cheap, and safe alternative to organic solvents to
Development and Application of Continuous Microwave
Reactor
tion1
4,15
The promising results obtained from the batch reaction
for DHP’s synthesis encouraged us to develop a continuous
microwave reactor (CMR) to carry out DHP synthesis
reaction at a larger scale. We have used 50% NaPTSA
hydrotrope solution to carry out reaction under CMR.
(
(
(
(
1) Caddick, S. Tetrahedron 1995, 51(38), 10403.
2) Strauss, C. R.; Trainor, R. W. Aust. J. Chem. 1995, 48, 1665.
3) Galema, S. A. Chem. Soc. ReV. 1997, 26, 233.
4) Loupy, A.; Petit, A.; Hamelin, J.; Texier-Boullet, F.; Jacquault, P.; Mathe,
D. Synthesis 1998, 1213.
(
(
(
(
(
5) Adamo, F.; Alberto, B. Pure Appl. Chem. 1999, 71(4), 573.
6) Cresswell, S. L.; Haswell, S. J. Chem Ind. (London) 1999, (16), 621.
7) Varma, R. S. Green Chem. 1999, 1(1), 43.
8) Strauss, C. R. Aust. J. Chem. 1999, 52, 83.
9) Cablewski, T.; Faux, A. F.; Straus, C. R. J. Org. Chem. 1994, 59, 3408.
Description of the Reactor
An omega-shaped circular glass reactor was constructed
for CMR study. The omega shaped reactor was found to be
(
(
(
10) Kabza, K. G.; Chapados, B. R.; Gestwick, J. E.; McGrath, J. L. J. Org.
Chem. 2000, 65, 1210.
(15) Khadilkar, B. M.; Sadvilkar, V. G.; Gaikar, V. G. J. Chem. Technol.
Biotechnol. 1995, 63, 33.
(16) Natale, N. R. Chem. InnoV. 2000 (Nov.), 23.
(17) Janis, R. A.; Triggle D. J. J. Med. Chem. 1983, 26, 775.
(18) Leov, B.; Goodman, M. M.; Snader, K. M.; Tedeschi, R.; Macko E. J.
Med. Chem. 1974, 19, 956.
11) Cleophax, J.; Liagre, M.; Loupy, A.; Petit, A. Org. Process Res. DeV. 2000,
4
, 498.
12) Esveld, E.; Chemat, F.; vanHaveren, J. Chem. Eng. Technol. 2000, 23(3),
97.
2
(
(
13) Bperio, B.; Dozias, M.; Hamelin, J. Org. Process. Res. DeV. 1998, 2, 428.
14) Khadilkar, B. M.; Gaikar, V. G.; Chitnavis, A. A. Tetrahedron Lett. 1995,
(19) Coburn, R. A.; Wierzba, M.; Suto, M. J.; Solo, A. J.; Triggle, A. M.; Triggle,
D. J. J. Med. Chem. 1988, 31, 2103.
3
6 (44), 8083.
4
52
•
Vol. 5, No. 4, 2001 / Organic Process Research & Development
10.1021/op010026q CCC: $20.00 © 2001 American Chemical Society and The Royal Society of Chemistry
Published on Web 06/21/2001

 Khadilkar, Bhushan M.
Khadilkar, Bhushan M.
 Madyar, Virendra R.
Madyar, Virendra R.