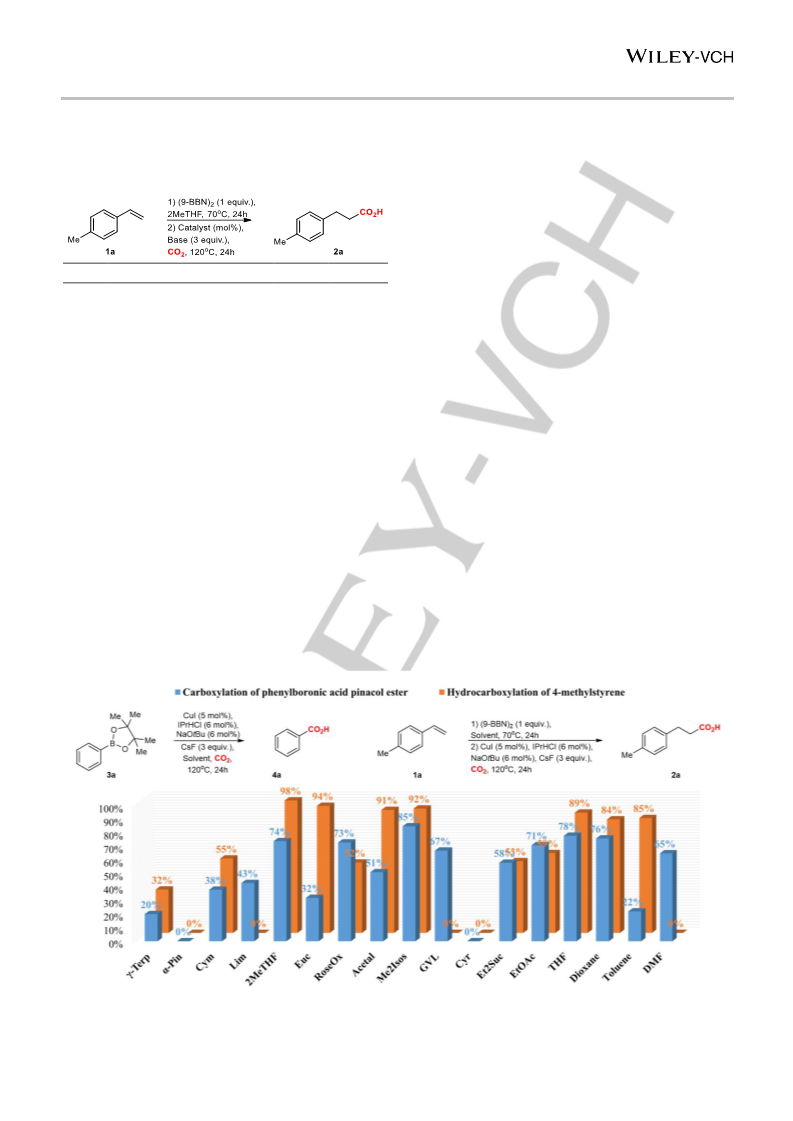
10.1002/cssc.201903224
ChemSusChem
FULL PAPER
type of experiments can be conducted applying exclusively
biomass-derived solvents. The hydrocarboxylation of styrenes
was also examined using different Ni-based catalysts, which
unfortunately were not successful (ESI, Scheme S4, C, D).
Further, we have explored hydrocarboxylation of acetylenes in
biomass-derived solvents (Scheme 4, C-E).[19] We employed
diphenylacetylene 8a to test different catalytic systems based on
Ni, Cu and Fe. Promising results were observed when using the
CuF2/IMesHCl/NaOtBu catalytic system with triethoxysilane as a
reducing agent (Scheme 4, D). The best solvent proved to be
Acetal (61%), whereas the yields of the hydrocarboxylation
product 9a were slightly lower in 2MeTHF (41%) and Euc (43%).
The reaction performed in dioxane (solvent used in the original
work) gave 9a in 57% yield.[19a] Hydrocarboxylation of 8a was also
possible with the Ni(cod)2/CsF catalytic system using Et2Zn as
reductant. In this case the yield of 9a was only 21% in 2MeTHF,
whereas in MeCN (solvent used in the original work) the product
was obtained in 49% yield (Scheme 4, C).[19b] Among Fe-based
catalytic systems moderate yields of 9a were observed with FeCl2
used in combination with 3.4M EtMgBr in 2MeTHF (42% in
2MeTHF, Scheme 4, E). Similar conditions were tested in Et2O
(solvent used in the original work) where 9a was obtained in 14%
yield.[19e]
studies on phenylacetylene 5a indicated that Cs2CO3 alone can
initiate direct C-H carboxylation in 2MeTHF; however, with only
20% isolated yield (Scheme 4, I). The yield was improved up to
31% by switching to GVL. Further improvements were achieved
by using the catalytic system developed for carboxylation of
organoboranes and organoboronates. The best yields of 4o were
observed in 2MeTHF and Acetal (76% and 63% respectively),
while GVL turned to be far less effective with Cu-catalyst (27%).
The best conditions were also tested in THF where 4o was
obtained in 38% yield. Unfortunately, all attempts to accomplish
C-H carboxylation of azoles in biomass-derived solvents failed
(ESI, Scheme S4, A, B).
For isolation and purification of the obtained carboxylic acids we
mainly used acid-base extraction techniques. Analysis of different
renewable solvents for extraction showed that Et2O, which is
readily available from ethanol, but is not popular in industry due
to volatility and flammability, can be replaced by renewable
2MeTHF, Acetal, diethoxymethane and dimethoxymethane
without any noticeable drop in yields. Column chromatography,
based on the use of mixtures of heptane/EtOAc and
Et2O/pentane/HCO2H, was only necessary for the purification of
the products of decarboxylative hydrocarboxylation of
phenylacetylene (Scheme 4, A).
Next, we examined other carboxylative transformations. Excellent
results were observed for the Cu-catalyzed borocarboxylation of
styrenes (Scheme 4, F).[20] Particularly, it was found that the
catalytic system based on CuCl and ICyHCl (1,3-
dicyclohexylimidazolium chloride), originally developed by Popp
et al.,[20b] operates well in biomass-derived ethers, initiating
efficient borocarboxylation of 1a. In this case the best solvent was
Euc (85%), but good yields of borocarboxylation product 10a were
also observed in 2MeTHF (81%), while Acetal (44%) was far less
effective. For comparison, the borocarboxylation of 1a performed
in THF (solvent used in the original work) gave 10a in 78%
yield.[20b]
We also explored the carbocarboxylation of olefins, which is
known to proceed under influence of a wide range of catalysts
based on both early and late transition metals.[21] Screening of
several catalysts derived from Zr and Ti as well as reducing
agents showed that carbocarboxylation of 4-methylstyrene 1a can
be performed in biomass-derived solvents (Scheme 4, G). The
best results were observed using Cp2ZrCl2 (zirconocene
dichloride) as catalyst precursor combined with EtMgBr in Acetal
(24%). Application of other ethers as solvents did not improve the
yield of 11a. Using THF under elsewise identical conditions gave
11a in comparable 28% yield.[21c]
Conclusion
We have shown that a variety of CO2-based carboxylations can
be performed in biomass-derived solvents, including a range of
previously unknown solvents. The studied media included polar
aprotic biomass-derived ethers (2MeTHF, Acetal, Me2Isos, Cyr,
Euc, RoseOx) and esters (GVL, Et2Suc, EtOAc) as well as
nonpolar aprotic unsaturated terpenes and their derivatives (γ-
Terp, α-Pin, Lim, Cym). Initial studies on Cu-catalyzed
carboxylation of in situ generated organoboranes and -boronates
revealed that most of the biosolvents are suitable for carboxylative
transformations, with biomass-derived ethers showing the best
efficiency. Our methodology was successfully applied to
organoboranes generated from styrenes and internal alkenes, as
well as for carboxylation of aryl-, alkenyl-, alkynyl- and
benzylboronic acid pinacol esters. On basis of the latter, we have
synthesized the commercial drugs Fenoprofen and Flurbiprofen.
Biomass-derived
solvents
were
further
applied
for
hydrocarboxylation of acetylenes and styrenes, using catalysts
based on Cu, Ni or Fe. We observed from moderate to good yields
and excellent regioselectivities. Very good results were obtained
for the Cu-catalyzed borocarboxylation of styrenes and C-H
carboxylation of phenylacetylene. Biomass-derived ethers can
also be used for the Cu-catalyzed carboxylation of
triethoxyphenylsilane and the Zr-catalyzed carbocarboxylation of
styrenes. Most of the reactions were examined in traditional
organic solvents as a comparison. These studies revealed that
there is no advantage in using traditional solvents for the reactions
described here. In most of the cases, the yields obtained in
traditional solvents were comparable with those in biosolvents,
while in other cases, biomass-derived solvents, as a matter of fact,
performed better. Biomass-derived ethers showed the best
performance, with 2MeTHF generally being superior; however, it
is not a universal solvent. In several cases, excellent results were
instead observed when using Me2Isos, Acetal, RoseOx or Euc
solvents. We believe that the biomass-derived solvents
Similar to organoboronates, the carboxylation of organosilicon
reagents can be performed in biomass-derived solvents (Scheme
4,
H).[22]
The
best
results
were
observed
with
triethoxyphenylsilane 12a using Cu-based catalysts. Particularly,
it was found that biomass-derived ethers are not the best solvents
for this reaction (2MeTHF 16%, Acetal 0%). The best yields of
benzoic acid 4a were observed when using the esters GVL (42%)
and Et2Suc (36%) as solvent, CuBr as catalyst precursor and CsF
as a base. In this case, the yield of 4a could be notably improved
when running the reaction in DMA (62%). It should be noted that
the reaction does not work without Cu-catalyst.
Eventually,
we
examined
TM-catalyzed
direct
C-H
carboxylations.[23,24] So far, direct C-H carboxylations were
performed on azoles possessing an acidic C-H bond, arenes with
appropriate directing groups[23] and terminal acetylenes.[24] Our
6
This article is protected by copyright. All rights reserved.

 Gevorgyan, Ashot
Gevorgyan, Ashot
 Hopmann, Kathrin H.
Hopmann, Kathrin H.
 Bayer, Annette
Bayer, Annette