Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
Organometallics
Communication
active.10,11 Pleasantly, we found that dimer 3 is active in the
isomerization of the model substrate 1-octen-3-ol (Table 1).
electron monoruthenium active species III formed after
thermal dissociation of the Shvo catalysts. More challenging
organic transformations with related complexes in catalysis is
currently under investigation in our laboratory.
Table 1. Catalytic Isomerization of 1-Octen-3-ol to Octan-3-
a
one Using 3
ASSOCIATED CONTENT
* Supporting Information
■
S
Experimental section, full characterization data for all new
compounds, and crystallographic data. This material is available
a
b
entry
% Ru
solvent
temp
time
yield
1
2
3
4
5
6
1 mol %
THF
THF
THF
THF
THF
H2O
80 °C
80 °C
80 °C
40 °C
rt
5 min
10 min
1 h
>99%
>99%
>99%
>99%
96%
c
0.5 mol %
0.1 mol %
0.5 mol %
0.5 mol %
0.5 mol %
AUTHOR INFORMATION
Corresponding Author
*E-mail: alain.igau@lcc-toulouse.fr.
■
4 h
72 h
Notes
80 °C
40 min
>99%
The authors declare no competing financial interest.
a
Reactions performed under a N2 atmosphere using 2 mmol of 1-
b
octen-3-ol (0.5 M solutions). Yields of octan-3-one determined by
GC. 90% isolated yield after workup.
c
ACKNOWLEDGMENTS
■
M.K. is grateful to MESR for a doctoral fellowship. L.M.-R.,
P.C., and V.C. thank the Spanish MINECO for financial
support (projects CTQ2010-14796 and CTQ2013-40591).
Johnson Matthey is greatly acknowledged for a gift of RuCl3·
xH2O.
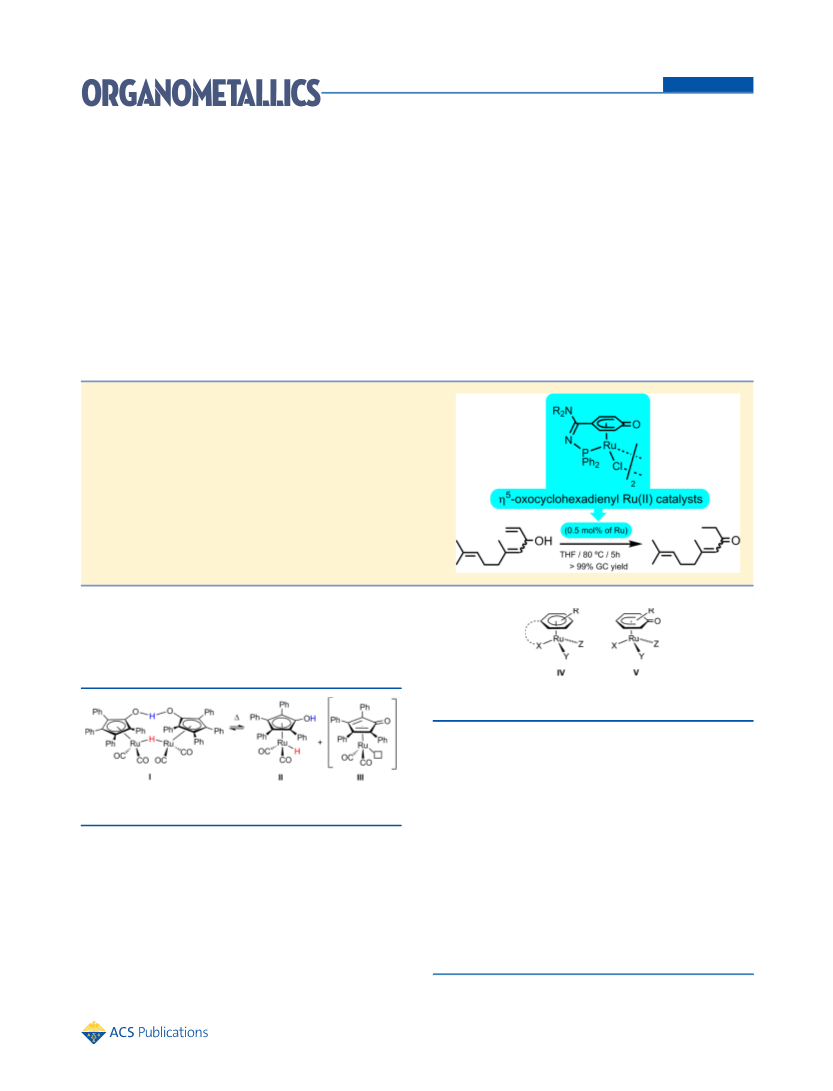
Thus, performing the catalytic reaction in THF at 80 °C with a
metal loading of 1 mol % of Ru (0.5 mol % of 3), selective and
quantitative formation of octan-3-one was observed by GC after
only 5 min of heating (entry 1 in Table 1; TOF = 1200 h−1).12
Note that, contrary to the vast majority of ruthenium catalysts
known for this transformation,10 compound 3 is able to operate
under base-free conditions.13,14 A possible cooperative effect of
the CO unit of the η5-oxocyclohexadienyl ligand or the
pendant NiPr2 group, which could facilitate the generation of
the more coordinating oxo-allyl anion by deprotonation of the
allylic alcohol, may explain this fact. As shown in entries 2 and
3, lower metal loadings were tolerated without a drastic increase
in the reaction times. A wide range of temperatures and
solvents in which 3 is able to operate is tolerated (full details in
the Supporting Information). For example, at 40 °C, complete
conversion of 1-octen-3-ol into octan-3-one was reached within
4 h using a metal loading of 0.5 mol % (entry 4). At rt, a longer
time is required to attain a good conversion (entry 5).15
Especially noteworthy is the possibility of using water as solvent
(entry 6).16 Other allylic alcohols were subjected to the action
of complex 3, and efficient and selective formation of the
desired carbonyl compounds was in all cases observed (full
details in the Supporting Information). As a representative
example, the chemoselective and quantitative isomerization of
the citral-derived alcohol 4 (1:1 mixture of E/Z isomers) into
the ketone 5 is shown in Scheme 2.17
REFERENCES
■
(1) (a) Ruthenium in Organic Synthesis; Murahashi, S.-I., Ed.; Wiley-
VCH: Weinheim, 2004. (b) Ruthenium Catalysts and Fine Chemistry;
Bruneau, C.; Dixneuf, P. H., Eds.; Springer: Berlin, 2004.
(2) Blum, Y.; Czarkie, D.; Rahamin, Y.; Shvo, Y. Organometallics
1985, 4, 1459−1461.
(3) (a) Warner, M. C.; Casey, C. P.; Backvall, J.-E. Top. Organomet.
̈
Chem. 2011, 37, 85−125. (b) Conley, B. L.; Pennington-Boggio, M.
K.; Boz, E.; Williams, T. J. Chem. Rev. 2010, 110, 2294−2312.
(c) Karvembu, R.; Prabhakaran, R.; Natarajan, K. Coord. Chem. Rev.
2005, 249, 911−918.
(4) (a) Therrien, B.; Ward, T. R. Angew. Chem., Int. Ed. 1999, 38,
405−408. (b) Faller, J. W.; D’Alliessi, D. G. Organometallics 2003, 22,
2749−2757. (c) Cetinkaya, B.; Demir, S.; Ozdemir, I.; Toupet, L.;
Sem
́
eril, D.; Bruneau, C.; Dixneuf, P. H. Chem.Eur. J. 2003, 9,
́
2323−2330. (d) Cadierno, V.; Díez, J.; García-Alvarez, J.; Gimeno, J.
Chem. Commun. 2004, 1820−1821. (e) Ito, M.; Endo, Y.; Ikariya, T.
Organometallics 2008, 27, 6053−6055. (f) Arquier, D.; Vendier, L.;
Miqueu, K.; Sotiropoulos, J.-M.; Bastin, S.; Igau, A. Organometallics
2009, 28, 4945−4957. (g) Parekh, V.; Ramsden, J. A.; Wills, M. Catal.
Sci. Technol. 2012, 2, 406−411.
(5) (a) Hayes, A. M.; Morris, D. J.; Clarkson, G. J.; Wills, M. J. Am.
Chem. Soc. 2005, 127, 7318−7319. (b) Cheung, F. K.; Hayes, A. M.;
Hannedouche, J.; Yim, A. S. Y.; Wills, M. J. Org. Chem. 2005, 70,
3188−3197.
Scheme 2. Isomerization of a Citral-Derived Allylic Alcohol
(6) Here are some examples of half-sandwich η5-oxocyclohexadienyl
ruthenium complexes: (a) Cole-Hamilton, D. J.; Young, R. J.;
Wilkinson, G. J. Chem. Soc., Dalton Trans. 1976, 1995−2001.
(b) Obeso Rosete, R.; Cole-Hamilton, D. J.; Wilkinson, G. J. Chem.
Soc., Dalton Trans. 1979, 1618−1623. (c) Snelgrove, J. L.; Conrad, J.
C.; Yap, G. P. A.; Fogg, D. E. Inorg. Chim. Acta 2003, 345, 268−278.
(d) Abdur-Rashid, K.; Fedorkiw, T.; Lough, A. J.; Morris, R. H.
Organometallics 2004, 23, 86−94. (e) Snelgrove, J. L.; Conrad, J. C.;
Eelman, M. D.; Moriarty, M. M.; Yap, G. P. A.; Fogg, D. E.
Organometallics 2005, 24, 103−109. (f) Ferrando-Miguel, G.; Wu, P.;
Huffman, J. C.; Caulton, K. G. New J. Chem. 2005, 29, 193−204.
(g) MacInnis, M. C.; McDonald, R.; Ferguson, M. J.; Tobisch, S.;
Turculet, L. J. Am. Chem. Soc. 2011, 133, 13622−13633.
In conclusion, we developed a straightforward synthesis of
original tethered piano-stool η5-oxocyclohexadienyl Ru(II)
complexes and provided pioneering evidence for catalytic
activity of a metal complex incorporating an η5-oxocyclohex-
adienyl ligand. The dimeric Ru(II) complex presented herein is
effective in the base-free redox isomerization of allylic alcohols
and able to operate under mild conditions.18 It is noteworthy
that the monomeric ruthenium fragment of the η5-oxocyclo-
hexadienyl-Ru(II) dimer 3 is structurally related to the 16-
(7) Here are some examples of sandwich complexes incorporating η5-
oxocyclohexadienyl ligands. See for ruthenium complexes: (a) Loren,
S. D.; Campion, B. K.; Heyn, R. H.; Don Tilley, T.; Bursten, B. E.;
Luth, K. W. J. Am. Chem. Soc. 1989, 111, 4712−4718. (b) Koelle, U.;
C
dx.doi.org/10.1021/om501073q | Organometallics XXXX, XXX, XXX−XXX

 Kechaou-Perrot, Manel
Kechaou-Perrot, Manel
 Vendier, Laure
Vendier, Laure
 Bastin, Stphanie
Bastin, Stphanie
 Sotiropoulos, Jean-Marc
Sotiropoulos, Jean-Marc
 Miqueu, Karinne
Miqueu, Karinne
 Menndez-Rodrguez, Luca
Menndez-Rodrguez, Luca
 Crochet, Pascale
Crochet, Pascale
 Cadierno, Victorio
Cadierno, Victorio
 Igau, Alain
Igau, Alain