
Journal of the Brazilian Chemical Society p. 1509 - 1516 (2010)
Update date:2022-08-30
Topics:
 Vieira, Gizelle A. B.
Vieira, Gizelle A. B.
 De Freitas Araujo, Daniel M.
De Freitas Araujo, Daniel M.
 Lemos, Telma L. G.
Lemos, Telma L. G.
 De Mattos, Marcos Carlos
De Mattos, Marcos Carlos
 Da Conceic?a?o F. De Oliveira, Maria
Da Conceic?a?o F. De Oliveira, Maria
 Melo, Va?nia M. M.
Melo, Va?nia M. M.
 De Gonzalo, Gonzalo
De Gonzalo, Gonzalo
 Gotor-Ferna?ndez, Vicente
Gotor-Ferna?ndez, Vicente
 Gotor, Vicente
Gotor, Vicente
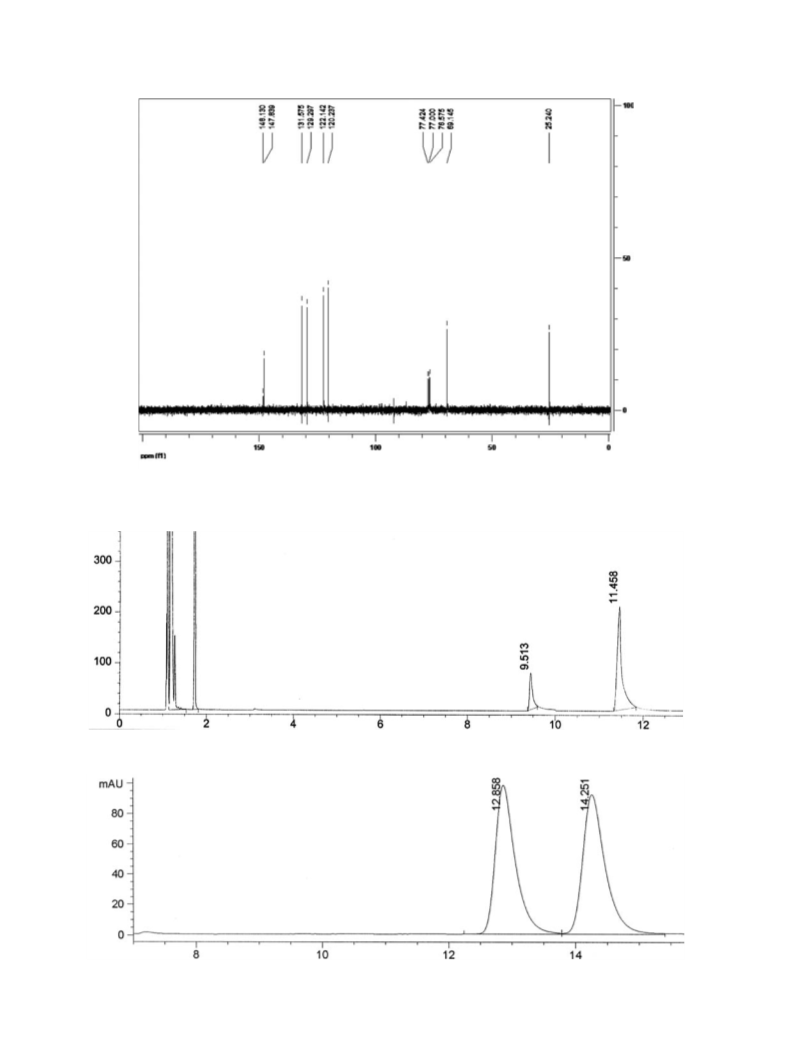
The reactivity and stereoselectivity showed by a new strain of Candida tropicalis in the reduction of prochiral ketones have been compared with the ones previously attained in our laboratory using microorganisms from the Brazilian biodiversity. In this manner, Candida tropicalis has demonstrated its versatility as stereoselective agent in the bioreduction of a series of aromatic ketones. These prochiral compounds were converted into their corresponding optically alcohols with moderate to excellent stereopreference depending on the substrate structure. Among ketones tested, nitroacetophenones were enzymatically reduced to enantiopure (S)-alcohol with complete conversion.
View More
























Tangshan Moneide Trading Co., Ltd.
Contact:+86-315-8309571
Address:2-7-420 Jidong Building Materials Commercial Center, Tangshan, Hebei, 064000 China
Engineering Research Center of Pesticide, Heilongjiang Province
Contact:+86-451-86609001
Address:No.74 of Xuefu Road, Nangang District,
Wuxi Morality Chemical Co., Ltd
Contact:
Address:B/7F, 321th WuYun Rd, Wanda Plaza, Wuxi City, 214174, China
A.M FOOD CHEMICAL CO., LIMITED
Contact:86-531-87100375
Address:20Floor,Bblock,1Building,pharma-valley,Jinan,China
Shenyang NovPharm Technology Co., Ltd.
Contact:.+86-24-24165786
Address:Room 306, Hongjin Mansion, No. 36-1, Wanliutang Rd., Shenhe District, Shenyang, Liaoning, P.R.C.
Doi:10.1021/jacs.0c05804
(2020)Doi:10.1016/0022-2852(68)90023-4
(1968)Doi:10.1002/pola.24692
(2011)Doi:10.1126/science.133.3470.2067
(1961)Doi:10.1080/15421406.2014.905087
(2015)Doi:10.1007/BF00629774
(1991)