Organic Letters
Letter
Chem., Int. Ed. 2012, 51, 10712. (c) Que, L.; Tolman, W. B. Nature 2008,
Battula, S.; Kushwaha, M.; Gupta, A. P.; Ahmed, Q. N.; Vishwakarma, R.
4
55, 333. (d) Limberg, C. Angew. Chem., Int. Ed. 2003, 42, 5932.
A. Org. Lett. 2014, 16, 1152. For I -mediated reactions, see: (f) Gao, Q.;
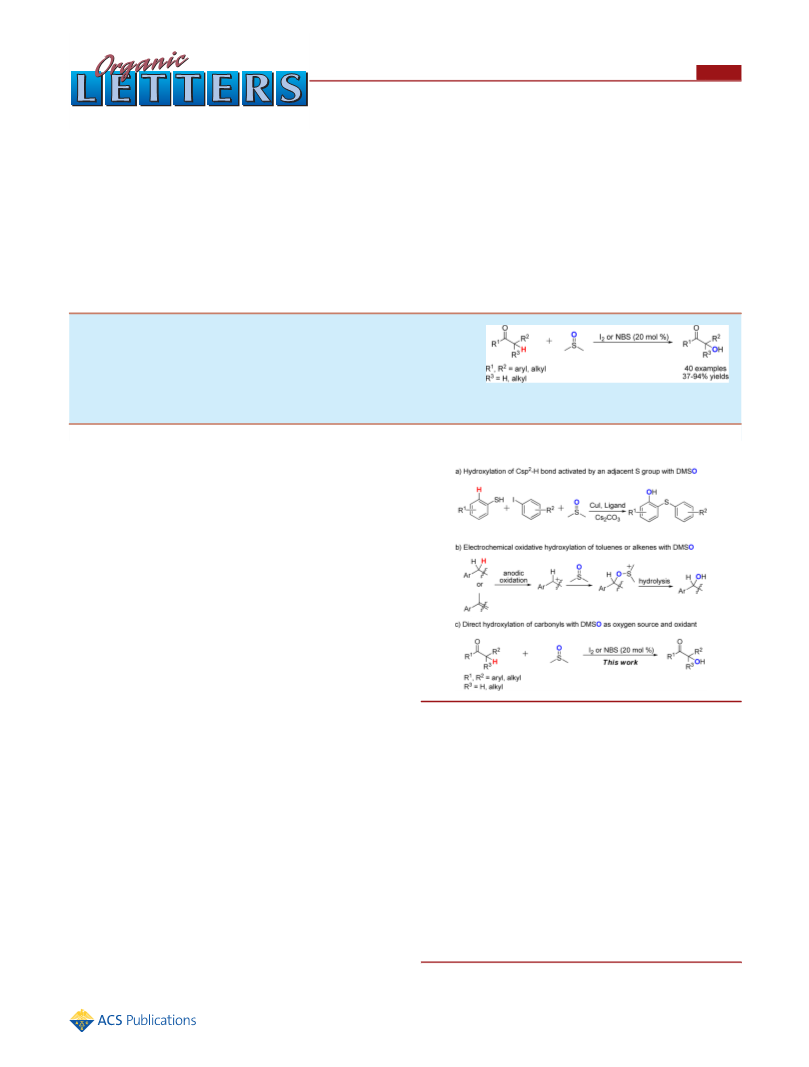
2
2
(
2) For some recent examples of the hydroxylation of Csp −H bonds,
see: (a) Mo, F.; Trzepkowshi, L. J.; Dong, G. Angew. Chem., Int. Ed.
012, 51, 13075. (b) Shan, G.; Yang, X.; Ma, L.; Rao, Y. Angew. Chem.,
Wu, X.; Liu, S.; Wu, A. Org. Lett. 2014, 16, 1732. (g) Yin, G.; Zhou, B.;
Meng, X.; Wu, A.; Pan, Y. Org. Lett. 2006, 8, 2245. (h) Wu, X.; Gao, Q.;
Liu, S.; Wu, A. Org. Lett. 2014, 16, 2888. (i) Wu, X.; Gao, Q.; Lian, M.;
Liu, S.; Wu, A. RSC Adv. 2014, 4, 51180.
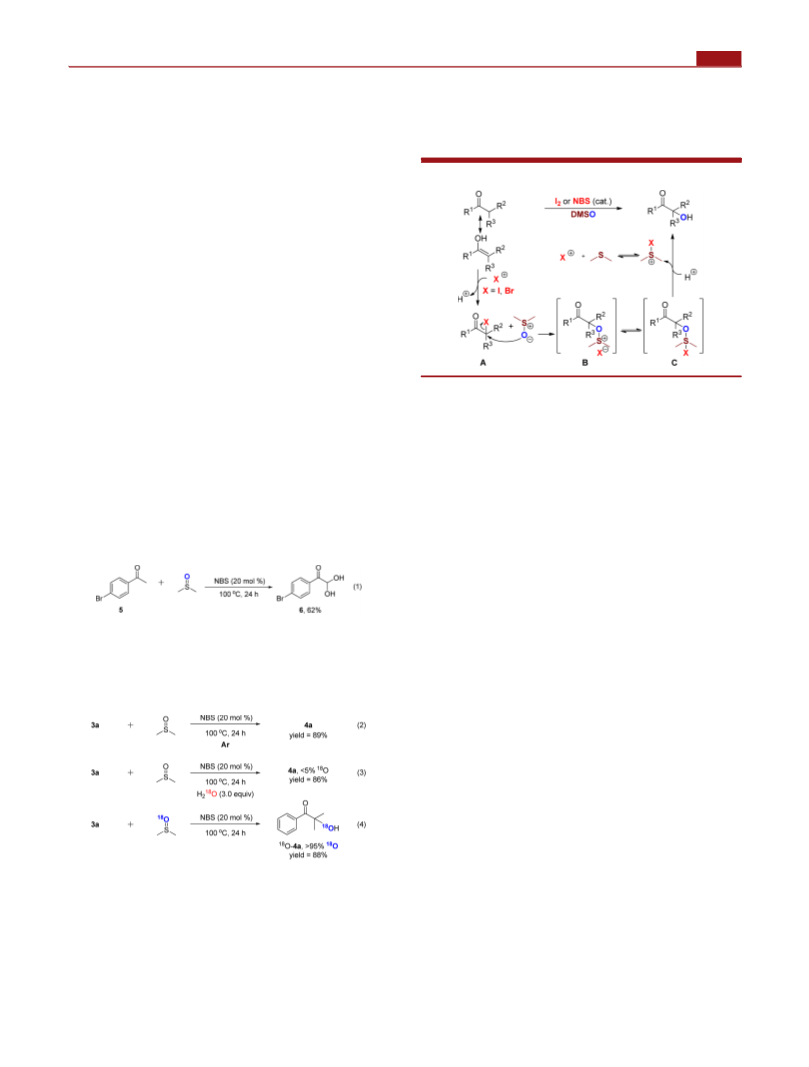
(16) There is only one DMSO-oxidized Kornblum reaction with the
formation of hydroxyl product in limited cases, in which the OH group
2
Int. Ed. 2012, 51, 13070. (c) Yan, Y.; Feng, P.; Zheng, Q.-Z.; Liang, Y.-F.;
Lu, J.-F.; Cui, Y.; Jiao, N. Angew. Chem., Int. Ed. 2013, 52, 5827.
(
d) Yuan, C.; Liang, Y.; Hernandez, T.; Berriochoa, A.; Houk, K. N.;
Siegel, D. Nature 2013, 499, 192. (e) Ohkubo, K.; Fujimoto, A.;
Fukuzumi, S. J. Am. Chem. Soc. 2013, 135, 5368. (f) Serrano-Plana, J.;
Garcia-Bosch, I.; Miyake, R.; Costas, M.; Company, A. Angew. Chem., Int.
Ed. 2014, 53, 9608. (g) Yang, F.; Rauch, K.; Kettelhoit, K.; Ackermann,
L. Angew. Chem., Int. Ed. 2014, 53, 11285. (h) Li, X.; Liu, Y.-H.; Gu, W.-
was produced by hydrolysis with H
2
O; see: Baranac-Stojanovic, M.;
́
Markovic, R.; Stojanovic, M. Tetrahedron 2011, 67, 8000.
́
́
(17) Xu, R.; Wan, J.-P.; Mao, H.; Pan, Y. J. Am. Chem. Soc. 2010, 132,
15531.
(18) (a) Ashikari, Y.; Nokami, T.; Yoshida, J. Org. Lett. 2012, 14, 938.
(b) Ashikari, Y.; Shimizu, A.; Nokami, T.; Yoshida, J. J. Am. Chem. Soc.
2013, 135, 16070. (c) Ashikari, Y.; Nokami, T.; Yoshida, J. J. Am. Chem.
Soc. 2011, 133, 11840.
J.; Li, B.; Chen, F.-J.; Shi, B.-F. Org. Lett. 2014, 16, 3904.
(
3
3) For reviews on Csp −H hydroxylation: (a) Newhouse, T.; Baran,
P. S. Angew. Chem., Int. Ed. 2011, 50, 3362. (b) White, M. C. Science
012, 335, 807.
(
19) (a) Decker, C. Macromol. Rapid Commun. 2002, 23, 1067.
b) Dietliker, K.; Murer, P.; Husler, R.; Jung, T. PCT Int. Appl. US
0100104979, 2010. (c) Fouassier, J. P.; Allonas, X.; Lalevee, J.; Dietlin,
2
(
3
(
2
̈
4) For some recent examples of the hydroxylation of Csp −H bonds,
́
see: (a) Simmons, E. M.; Hartwig, J. F. Nature 2012, 483, 70. (b) Ren,
Z.; Mo, F.; Dong, G. J. Am. Chem. Soc. 2012, 134, 16991. (c) Prat, I.;
C. In Photochemistry and Photophysics of Polymer Materials; Wiley:
Hoboken, NJ, 2010; pp 351.
Gom
́
ez, L.; Canta, M.; Ribas, X.; Costas, M. Chem.Eur. J. 2013, 19,
908. (d) Gormisky, P. E.; White, M. C. J. Am. Chem. Soc. 2013, 135,
4052. (e) Sun, X.; Lee, H.; Lee, S.; Tan, K. L. Nat. Chem. 2013, 5, 790.
(
20) (a) Hu
b) Meneguzzo, E.; Norcini, G.; Bassi, G. L. PCT Int. Appl. US
0110065962, 2011.
21) (a) Majetich, G.; Hicks, R.; Reister, S. J. Org. Chem. 1997, 62,
321. (b) Mislow, K.; Simmons, T.; Melillo, J.; Ternay, A. J. Am. Chem.
Soc. 1964, 86, 1452 and references cited therein.
̈
sler, R.; Fuchs, A. PCT Int. Appl. WO 2004009651, 2004.
1
1
(
2
(
4
(
f) Ghavtadze, N.; Melkonyan, F. S.; Gulevich, A. V.; Huang, C.;
Gevorgyan, V. Nat. Chem. 2014, 6, 122. (g) Adams, A. M.; Du Bois, J.
Chem. Sci. 2014, 5, 656. (h) Flender, C.; Adams, A. M.; Roizen, J. L.;
McNeill, E.; Du Bois, J. Chem. Sci. 2014, 5, 3309.
(
5) Chuang, G. J.; Wang, W.; Lee, E.; Ritter, T. J. Am. Chem. Soc. 2011,
1
33, 1760.
(
6) Liang, Y.-F.; Jiao, N. Angew. Chem., Int. Ed. 2014, 53, 548.
(
7) A frequently used method for the preparation of α-
hydroxycarbonyls involves the hydroxylation of preformed enolates or
enol ethers with diverse oxidants, such as peroxyacids, hypervalent
iodines, metal oxides, N-sulfonyloxaziridines, and molecular oxygen. For
a review, see: Chen, B.-C.; Zhou, P.; Davis, F. A.; Ciganek, E. Organic
Reactions; Overman, L. E., Ed.; John Wiley & Sons, Inc.: New York,
2
(
003; Vol. 62, p 1.
8) (a) Raduchel, B. Synthesis 1980, 292. (b) Edward, M. G.;
Kenworthy, M. N.; Kitson, R. R. A.; Scott, M. S.; Taylor, R. J. K. Angew.
Chem., Int. Ed. 2008, 47, 1935. (c) Minotti, G.; Menna, P.; Salvatorelli,
E.; Cairo, G.; Gianni, L. Pharmacol. Rev. 2004, 56, 185. (d) Davis, F. A.;
Chen, B.-C. Chem. Rev. 1992, 92, 919.
(
9) (a) Kang, S.; Han, J.; Lee, E. S.; Choi, E. B.; Lee, H.-K. Org. Lett.
2
010, 12, 4184. (b) Ooi, T.; Uraguchi, D.; Morikawa, J.; Maruoka, K.
Org. Lett. 2000, 2, 2015. (c) Shindo, M.; Yoshimura, Y.; Hayashi, M.;
Soejima, H.; Yoshikawa, T.; Matsumoto, K.; Shishido, K. Org. Lett. 2007,
9, 1963. (d) Lee, H.-K.; Kang, S.; Choi, E. B. J. Org. Chem. 2012, 77,
5
454. (e) Liu, X.-L.; Wang, Q.-Y.; Sheng, S.-R.; Xu, C.; Cai, M.-Z. Synth.
Commun. 2008, 38, 3338.
10) For reviews, see: (a) Epstein, W. W.; Sweat, F. W. Chem. Rev.
967, 67, 247. (b) Martin, H. D.; Weise, A.; Niclas, H.-J. Angew. Chem.,
Int. Ed. 1967, 6, 318. (c) Mancuso, A. J.; Swern, D. Synthesis 1981, 165.
d) Tidwell, T. T. Synthesis 1990, 857. (e) Kilenyi, S. N. Oxidation of
(
1
(
Carbon−Halogen Bonds. In Comprehensive Organic Synthesos; Trost, B.
M., Fleming, I., Ed.; Pergamon: Oxford, 1991.
(11) Omura, K.; Sharma, A. K.; Swern, D. J. Org. Chem. 1976, 41, 957.
(12) Pfitzner, K. E.; Moffatt, J. G. J. Am. Chem. Soc. 1963, 85, 3027.
(13) Corey, E. J.; Chaykovsky, M. J. Am. Chem. Soc. 1962, 84, 867.
(14) Kornblum, N.; Powers, J. W.; Anderson, G. J.; Jones, W. J.; Larson,
H. O.; Levand, O.; Weaver, W. M. J. Am. Chem. Soc. 1957, 79, 6562.
15) For the developed Kornblum reaction with nonbromo-
(
substituted substrates, see: (a) Floyd, M. B.; Du, M. T.; Fabio, P. F.;
Jacob, L. A.; Johnson, B. D. J. Org. Chem. 1985, 50, 5022. (b) Schipper,
E.; Cinnamon, M.; Rascher, L.; Chiang, Y. H.; Oroshnik, W. Tetrahedron
Lett. 1968, 9, 6201. (c) Collis, A. J.; Foster, M. L.; Halley, F.; Maslen, C.;
McLay, I. M.; Page, K. M.; Redford, E. J.; Souness, J. E.; Wilsher, N. E.
Bioorg. Med. Chem. Lett. 2001, 11, 693. (d) McKenna, J. M.; Halley, F.;
Souness, J. E.; McLay, I. M.; Pickett, S. D.; Collis, A. J.; Page, K. M.;
Ahmed, I. J. Med. Chem. 2002, 45, 2173. (e) Mupparapu, N.; Khan, S.;
D
Org. Lett. XXXX, XXX, XXX−XXX

 Liang, Yu-Feng
Liang, Yu-Feng
 Wu, Kai
Wu, Kai
 Song, Song
Song, Song
 Li, Xinyao
Li, Xinyao
 Huang, Xiaoqiang
Huang, Xiaoqiang
 Jiao, Ning
Jiao, Ning