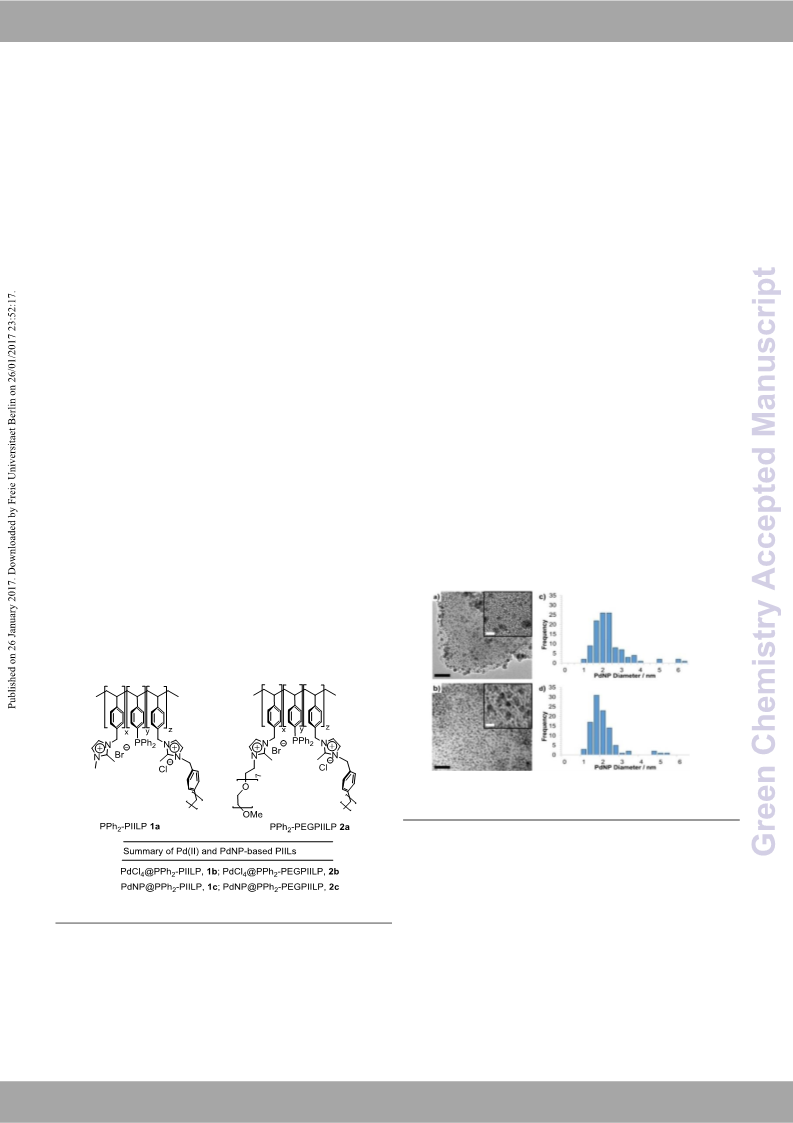
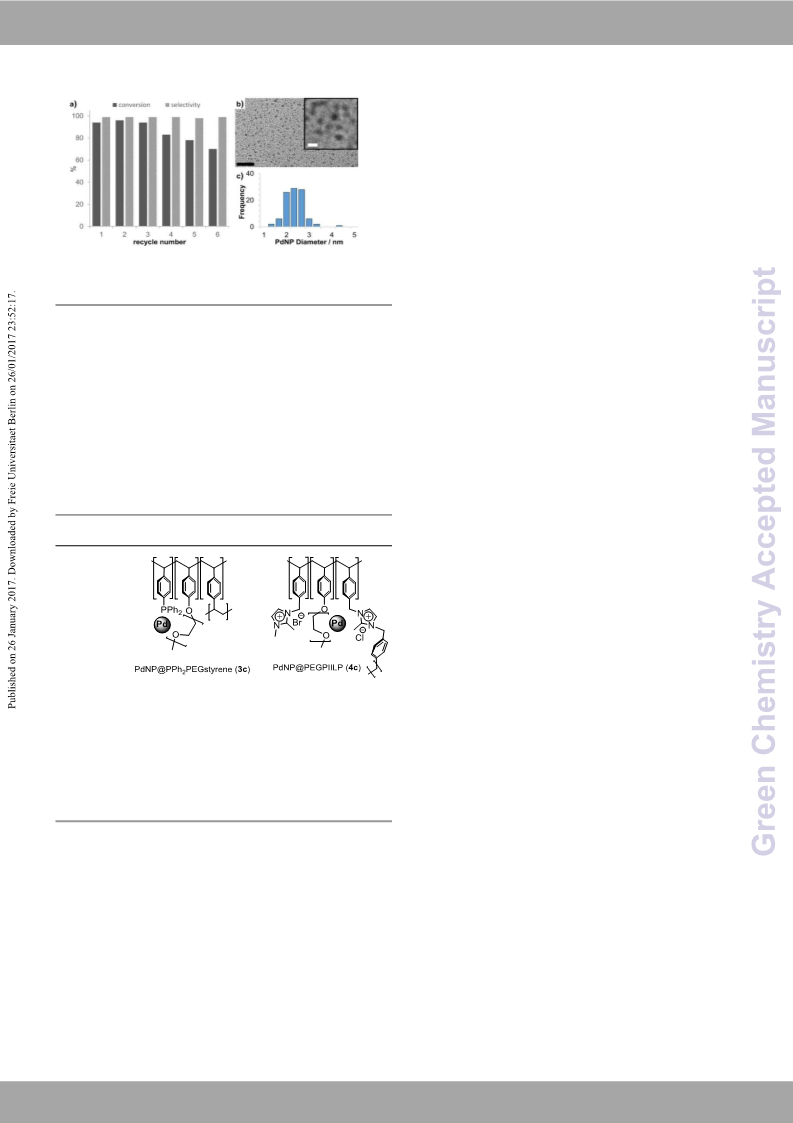
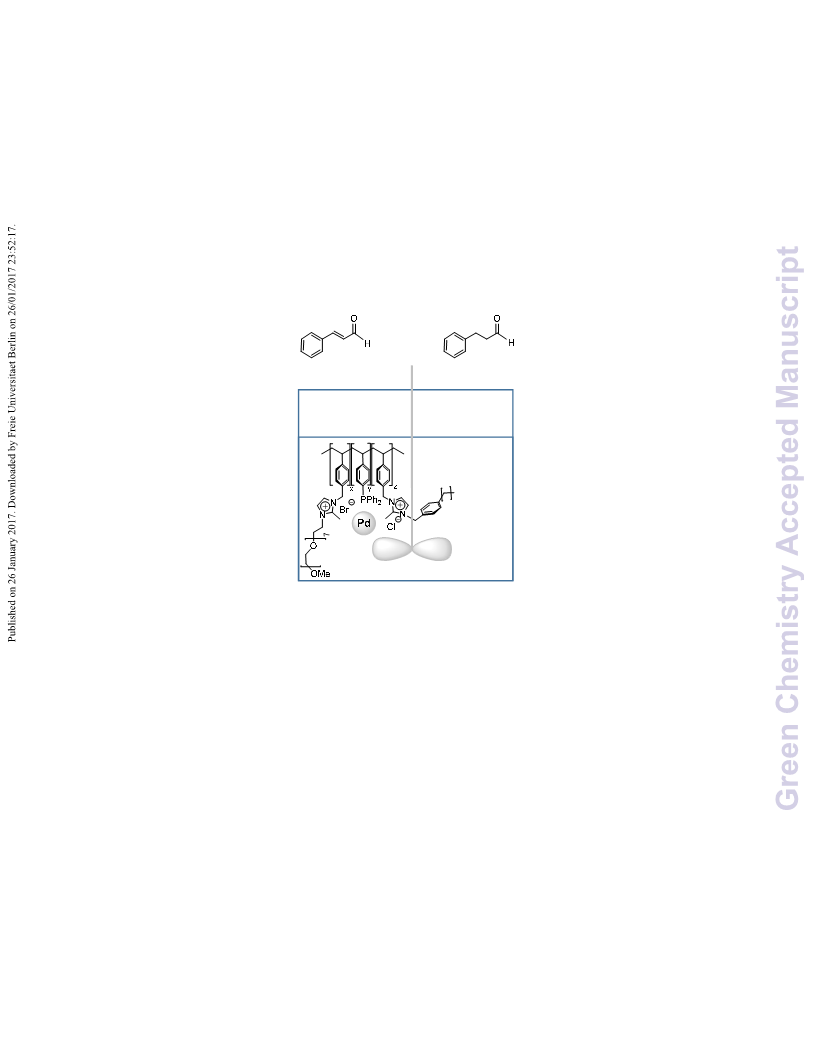
Pleas eG dr eo e nn o Ct ha de jmu si st tmr yargins
Page 6 of 7
COMMUNICATION
Journal Name
1
1
1
2 (a) A. P. Umpierre, P. F. P. Fichtner, S. R. Teixeira and J.
Dupont, Chem. Eur. J.; 2003, , 3263-3269; (b) F. Bellina,
Molecules, 2010, 15, 2211-2245.
3 (a) A. Zhang and H. Cui, Langmuir, 2009, 25, 2604-2612; (b)
Z. Wang, Q. Zhang, D. Kuehner, A. Ivaska and L. Niu, Green
Chem. 2008, 10, 907-909.
9
120; (g) W. Zhu, H. Yang, Y. Yu, L. Hua, H. Li, B. Feng and Z.
DOI: 10.1039/C6GC03528K
Hou, Phys. Chem. Chem. Phys., 2011, 13, 13492-13500; (h) N.
Iranpoor, S. Rahimi and F. Panahi, RSC Adv., 2016, 6, 3084-
3090; (i) N. Iranpoor, S. Rahimi and F. Panahi, RSC Adv., 2015,
, 69559-49567; (j) B. L. Albuquerque, A. Denicourt-Nowicki,
5
4 (a) D. B. Zhao, Z. F. Fei, T. J. Geldbach, R. Scopelliti, and P. J.
Dyson, J. Am. Chem. Soc., 2004, 126, 15876-15882. (b) C.
Chiappe, D. Pieraccini, D. Zhao and Z. Fei and P. J. Dyson,
Adv. Synth. Catal., 2006, 348, 68-74; (c) Z. Fei, D. Zhao, D.
Pieraccini, W. H. Ang, T. J. Geldbach, R. Scopelliti, C. Chiappe
and P. J. Dyson, Organometallics, 2007, 26, 1588-1598; (d) X.
Yang, Z. Fei, D. Zhao, W. H. Ang, Y. Li and P. J. Dyson, Inorg.
Chem., 2008, 47, 3292-3297; (e) M. H. G. Prechtl, J. D.;
Scholten and J. Dupont, J. Mol. Cat. A: Chem., 2009, 313, 74-
C. Meriadec, J. B. Domingos and A. Roucoux, J. Catal. 2016,
340, 144-153. (k) C. Deraedt and D. Astruc, Acc. Chem. Res.,
2014, 47, 494-503. For examples of aqueous phase catalysis
with surfactant-free palladium nanoparticles see: (l) M.
Hyotanishi, Y. Isomura, H. Yamamoto, H. Kawasaki and Y.
Obora, Chem. Commun., 2011, 47, 5750-5752; (m) M.
Nasrollahzadeh, S. Mohammad Sajadi, M. Maham, A. Ehsani,
RSC Adv., 2015,
5, 2562-2567; (n) A. Aijaz, Q.-L. Zhu, N.
Tsumori, T. Akita, Q. Xu, Chem. Commun., 2015, 51, 2577-
2580. For examples of aqueous phase catalysis by polymer-
stabilised palladium nanoparticles see: (o) D. Damodara, R.
Arundhathi, T. Venkata Ramesh Babu, M. K. Legan, H. J.
7
8.
5 (a) H. Itoh, K. Maka and Y. Chujo, J. Am. Chem. Soc., 2004,
26, 3026-3027; (b) N. Kocharova, J. Leiro, J. Lukkari, M.
1
1
1
Heinonen, T. Skala, F. Sutara, M. Skoda and M. Vondracek,
Langmuir, 2008, 24, 3235-3242.
Kumpaty and P. R. Likhar, RSC Adv., 2014, 4, 22567-22574;
(p) Y. Lee, S. Shabbir, S. Lee, H. Ahn and H. Rhee, Green
Chem., 2015, 17, 3579-3583. For examples of aqueous phase
catalysis by palladium nanoparticles stabilised in metal
organic frameworks see: (q) F. Zhang, S. Zheng, Q. Xiao, Y.
Zhong, W. Zhu, A. Lin and M. S. El-Shall, Green Chem., 2016,
18, 2900-2908.
6 (a) B. Léger, A. Denicourt-Nowicki, A. Roucoux and H. Olivier-
Bourbigou, Adv. Synth. Catal., 2008, 350, 153-159; (b) B.
Léger, A. Denicourt-Nowicki, H. Olivier-Bourbigou and A.
Roucoux, Inorg. Chem., 2007, 47
, 9090-9096; (c) A.
Denicourt-Nowicki, B. Léger and A. Roucoux, Phys. Chem.
Chem. Phys., 2011, 13, 13510-13517; (d) R. R. Dykeman, N. 22 Astruc and co-worker have recently reported a similar
Yan, R. Scopelliti and P. J. Dyson, Inorg. Chem., 2011, 50
17-719.
,
approach for the preparation of transition metal
nanoparticles for highly efficient aqueous phase catalysis. C.
Wang, R. Ciganda, L. Salmon, D. Gregurec, J. Irigoyen, S.
7
1
1
7 (a) N. Yan, X. Yang, Z. Fei, Y. Li, Y. Kou and P. J. Dyson,
Organometallics, 2009, 28, 937-939; (b) X. Yuan, N. Yan, S. A.
Katsyuba, E. E. Svereva, Y. Kou and P. J. Dyson, Phys. Chem.
Chem. Phys., 2012, 14, 6026-6033.
Moya, J. Ruiz and D. Astruc, Angew. Chem. Int. Ed. 2016, 55
,
3091-3095.
23 (a) E. Bus, R. Prins and J. A. van Bokhoven, Catal. Commun.,
8 (a) B. C. Leal, C. S. Consorti, G. Machado and J. Dupont, Catal.
2007, 8, 1397-1402; (b) P. Virtanen, J. Mikkola, T. Salmi, Ind.
Sci. Technol. 2015,
5
, 903-909; (b) K. L. Luska and A. Moores,
Eng. Chem. Res., 2009, 48, 10335-10342; (c) P. Maki-Arvela,
Xiao and Z.-T. Liu, Ind. Eng. Chem. Res., 2012, 51, 11112-
11121.
Adv. Synth. Catal., 2011, 353
Bahadorikhalili, L. Ma’mani, H. Mahdavi and A. Shafiee, RSC
Adv., 2015, , 71297-71305.
9 (a) S. G. Kwon, G. Krylova, A. Sumer, M. M. Schwartz, E. E.
,
3167-3177; (c) S.
5
1
Bunel, C. L. Marshall, S. Chattopadhyay, B. Lee, J. Jellinek and 24 (a) P. H. Z. Ribeiro, E. Y. Matsubara, J. M. Rosolen, P. M.
E. V. Shevchenko, Nano Lett. 2012, 12, 5382-5388; (b) Z.
Donate and R. Gunnella, J. Mol. Cat. A: Chem., 2015, 410, 34-
40; (b) H. Gu, X. Xu, A. Chen, P. Ao and X. Yan, Catal.
Commun., 2013, 41, 65-69.
Guo, C. Xiao, R. V. Maligal-Ganesh, L. Zhou, T. W. Goh, X. Li,
D. Tesfagaber, A. Thiel and W. Huang, ACS Catal., 2014, 4,
1
340-1348; (c) K. R. Kahsar, D. K. Schwartz and J. W. Medlin, 25 A. S. Nagpure, L. Gurrala, P. Gogoi and S. V. Chilukuri, RSC
J. Am. Chem. Soc., 2014, 136, 520-526. Adv., 2016, , 44333-44340.
0 I. Cano, A. M. Chapman, A. Urakawa and P. W. N. M. van 26 Y. Zhao, M. Lui, B. Fan, Y. Chen, W. Lv, N. Lu and R, Li, Catal.
Leeuwen, J. Am. Chem. Soc., 2014, 136, 2520-2528. Commun., 2014, 57, 119-123.
1 For examples of aqueous phase catalysis by surfactant- 27 K. Anderson, P. Goodrich, C. Hardacre and D. W. Rooney,
stabilised palladium nanoparticles see: (a) B. S. Souza, E. C. Green Chem., 2003, , 448-453.
Leopoldino, D. W. Tondo, J. Dupont and F. Nome, Langmuir, 28 S. Bhogeswararao and D. Srinivas, J. Catal. 2012, 285, 31-40.
6
2
2
5
2
012, 28, 833-840; (b) G. La Sorella, P. Canton, G. Strukul and 29 R. Herbios, S. Noël, B. Léger, S. Tilloy, S. Menuel, A. Addad, B.
A. Scarso, ChemmCatChem, 2014,
6
, 1575-1578; (c) J. Feng,
Martel, A. Ponchel, and E. Monflier, Green Chem. 2015, 17,
2444-2454.
S. Handa, F, Gallou and B. H. Lipshutz, Angew. Chem. Int. Ed.,
2
016, 55, 8979-8983; (d) S. Handa, Y. Wang, F. Gallou and B. 30 M. von Arx, T. Mallat, A. Baiker, J. Mol. Cat. A: Chem. 1999,
H. Lipshutz, Science, 2015, 349, 1087-1091; (e) B. H. Lipshutz
and S. Ghorai, AldrichimicaActa, 2008, 41, 59-72; (f) J.-H. Noh
148, 275-283.
6
| J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Doherty
Doherty
 Knight
Knight
 Backhouse
Backhouse
 Abood
Abood
 Alshaikh
Alshaikh
 Fairlamb
Fairlamb
 Bourne
Bourne
 Chamberlain
Chamberlain
 Stones
Stones