Paper
Dalton Transactions
3
3
CH
(
2 3 2 3
CH , J = 7.5 Hz), 0.88 (t, 3H, CH CH , J = 7.5 Hz), 0.86 Gold complex 11
3
13
1
t, 3H, CH CH , J = 7.5 Hz); C{ H} NMR (CDCl , 101 MHz):
2
3
3
A solution of [AuCl(THT)] (0.041 g, 0.13 mmol) in CH
(
(
temperature for 0.5 h, the reaction mixture was concentrated
to about 2 mL and then n-hexane (20 mL) was added. A white
precipitate formed, which was then separated by filtration and
2
Cl
25 mL) was added to a stirred solution of benzophosphole 8
0.100 g, 0.13 mmol) in CH Cl (25 mL). After stirring at room
2
3
δ = 160.10 (d, arom Cq-O, J(CP) = 10.4 Hz), 158.11 (s, arom
Cq-O), 155.11 (s, arom Cq-O), 15 507 (s, arom Cq-O),
42.98–120.58 (arom. C’s), 77.13 (s, OCH ), 76.92 (s, OCH ),
6.88 (s, OCH
Ar), 30.98 (s, ArCH
.1 Hz), 23.68 (CH CH ), 23.58 (CH CH ), 23.18 (CH CH ),
2 3 2 3 2 3
3.09 (CH
); P{ H} NMR (CDCl
ppm; elemental analysis calcd (%) for C H O P (M
90.97): C 78.96, H 7.01; found (%): C 79.04, H 7.11.
2
2
1
7
2
2
2
), 76.62 (s, OCH
2
), 31.61 (s, ArCH
Ar), 28.55 (d, ArCH
2
Ar), 31.16 (s,
Ar, J(CP) =
3
ArCH
3
2
2
2
2
dried under vacuum to give complex 11 (0.123 g, yield 95%).
2
CH
3
), 11.01 (CH
2
CH
3
), 10.86 (CH
, 162 MHz): δ = 33.4 (s, P(O)Ph)
2
CH
3
), 9.93
1
3 6 4
H NMR (CDCl , 500 MHz): δ = 7.89 (d, 1H, arom. CH, C H ,
J = 6.0 Hz), 7.76 (d, 1H, arom. CH, calixarene, J(PH) =
3
1
1
(CH
2
CH
3
3
3
4
=
5
2
55
5
r
3
1
7
.0 Hz), 7.66–7.61 (m, 3H, arom. CH, C
H, arom. CH, PPh, J = 7.7 Hz), 7.54 (m, 1H, arom. CH, C H ),
.43 (dt, 2H, arom. CH, PPh, J = 7.7 Hz, J(PH) = 2.0 Hz), 7.35
6
H
4
and PPh), 7.58 (t,
7
3
6
4
3
4
3
4
(
6 4
dt, 1H, arom. CH, C H , J = 7.2 Hz, J(PH) = 3.5 Hz), 7.14 (d,
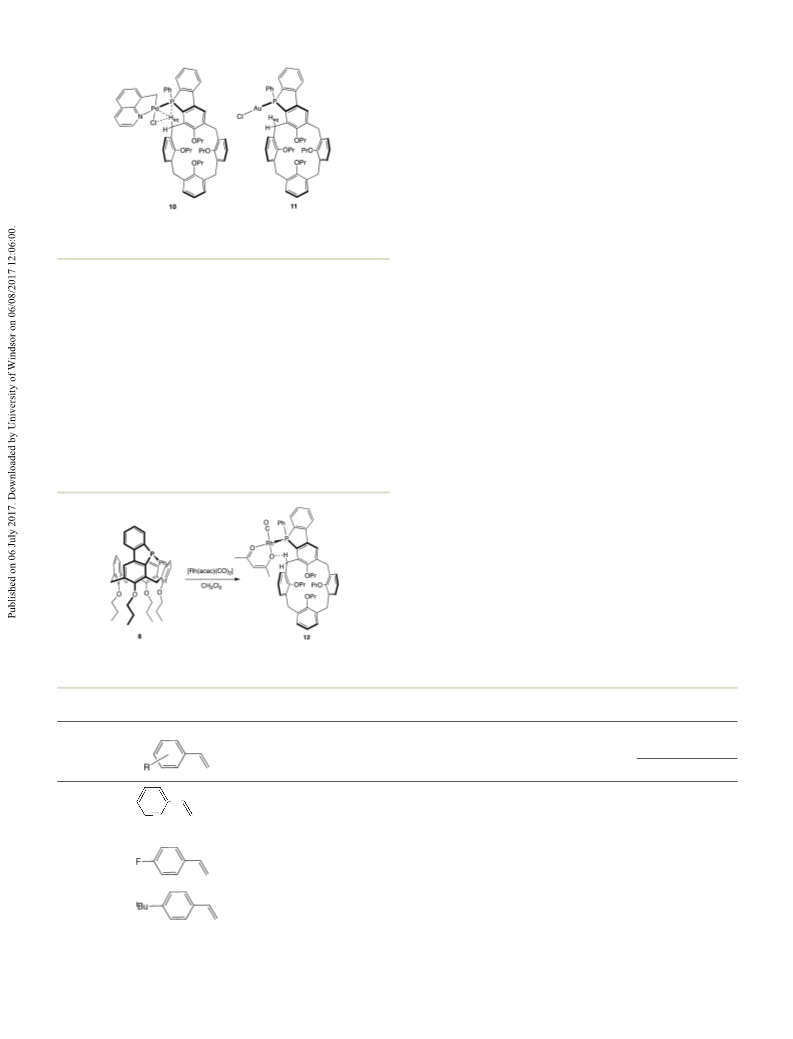
Palladium complex 10
3
1
H, arom. CH, calixarene, J = 7.0 Hz), 7.12 (d, 1H, arom. CH,
A solution of [Pd(8-mq)Cl]
2
(0.018 g, 0.03 mmol) in CH
2
Cl
2
3
3
calixarene, J = 7.0 Hz), 6.93 (t, 1H, arom. CH, calixarene, J =
(
0
10 mL) was added to a stirred solution of 8 (0.049 g,
.06 mmol) in CH Cl (10 mL). After stirring at room tempera-
ture for 0.5 h, the reaction mixture was concentrated to about
mL and n-hexane (20 mL) was added. The yellow precipitate
formed was separated by filtration and dried under vacuum
0.050 g, yield 73%). H NMR (CDCl
H, arom. CH of 8-mq, J = 3.5 Hz, J = 1.0 Hz), 8.14 (dd, 1H,
arom. CH of 8-mq, J = 8.5 Hz, J = 1.0 Hz), 7.90 (t, 1H, arom.
3
7
1
.5 Hz), 6.70 (d, 1H, arom. CH, calixarene, J = 7.0 Hz), 6.26 (t,
2
2
3
H, arom. CH, calixarene, J = 7.5 Hz), 6.25 (t, 1H, arom. CH,
3
3
calixarene, J = 7.5 Hz), 6.14 (d, 1H, arom. CH, calixarene, J =
2
3
6
1
.2 Hz), 6.12 (d, 1H, arom. CH, calixarene, J = 6.2 Hz), 6.08 (d,
H, arom. CH, calixarene, J = 7.5 Hz), 4.58 and 3.35 (AB spin
3
1
(
1
3
5
, 500 MHz): δ = 9.53 (tt,
2
system, 2H, ArCH Ar, J = 14.0 Hz), 4.45 and 3.17 (AB spin
2
3
2
2
system, 2H, ArCH Ar, J = 13.0 Hz), 4.40 and 3.15 (AB spin
3
4
2
system, 2H, ArCH Ar, J = 13.5 Hz), 4.23 and 3.66 (AB spin
system, 2H, ArCH Ar, J = 14.0 Hz), 4.11–4.02 (m, 2H, OCH ),
2
3
CH, C
Pd-8-mq), 7.77 (d, 1H, arom. CH, calixarene, J(PH) = 2.0 Hz),
.52–7.47 (m, 2H, arom. CH, PPh), 7.38–7.32 (m, 6H, arom.
CH, C and CH of 8-mq), 7.26–7.24 (m, 1H, arom. CH of
6
H
4
, J = 8.2 Hz), 7.86–7.80 (m, 3H, arom. CH, PPh and
2
2
2
4
3
4
.00 (t, 2H, OCH
2
, J = 8.2 Hz), 3.74–3.67 (m, 2H, OCH
2
), 3.60
7
3
(
t, 2H, OCH2, J = 6.7 Hz), 1.99–1.85 (m, 6H, CH CH ),
2 3
6
H
4
3
1
1
7
.82–1.75 (m, 2H, CH CH ), 1.12 (t, 3H, CH CH , J = 7.5 Hz),
2 3 2 3
3
4
8
1
1
1
-mq), 7.09 (dd, 1H, arom. CH, calixarene, J = 7.5 Hz, J =
.5 Hz), 7.02 (dd, 1H, arom. CH, calixarene, J = 7.5 Hz, J =
.5 Hz), 6.87 (t, 1H, arom. CH, calixarene, J = 7.5 Hz), 6.75 (d,
H, arom. CH, calixarene, J = 7.5 Hz), 6.14–6.13 (m, 2H, arom.
CH, calixarene), 6.02–6.01 (m, 1H, arom. CH, calixarene), 5.63
d, 1H, arom. CH, calixarene, J = 7.5 Hz), 4.99 (t, 1H, arom.
3
3
.03 (t, 3H, CH
2 3 2 3
CH , J = 7.2 Hz), 0.87 (t, 3H, CH CH , J =
3
4
3
13
1
.5 Hz), 0.84 (t, 3H, CH CH , J = 7.5 Hz); C{ H} NMR
2
3
3
3
(
CDCl , 125 MHz): δ = 157.95 (d, arom Cq-O, J(CP) = 11.7 Hz),
3
3
157.15 (s, arom Cq-O), 154.21 (s, arom Cq-O), 153.73 (s, arom
Cq-O), 142.41–120.37 (arom. C’s), 76.09 (s, OCH ), 75.92 (s,
2
3
(
OCH ), 75.89 (s, OCH ), 75.71 (s, OCH ), 30.75 (s, ArCH Ar),
2
2
2
2
3
CH, calixarene, J = 7.5 Hz), 4.60 and 3.35 (AB spin system, 2H,
3
0.12 (s, ArCH
2
2 2
Ar), 30.05 (s, ArCH Ar), 28.71 (d, ArCH Ar,
2
ArCH
ArCH
2
2
Ar, J = 13.5 Hz), 4.52 and 4.35 (AB spin system, 2H,
3
J(CP) = 7.1 Hz), 22.66 (CH CH ), 22.56 (CH CH ), 22.12
2
3
2
3
2
Ar, J = 14.0 Hz), 4.43 and 3.13 (AB spin system, 2H,
(
CH CH ), 22.07 (CH CH ), 10.00 (CH CH ), 9.84 (CH CH ),
2 3 2 3 2 3 2 3
2
ArCH Ar, J = 13.5 Hz), 4.33 and 2.98 (AB spin system, 2H,
31
1
2
8.95 (CH
2
CH
3
), 8.92 (CH
2
CH
3
);
3
P{ H} NMR (CDCl ,
2
ArCH
2
2
Ar, J = 13.5 Hz), 4.18–4.08 (m, 2H, OCH
2
), 4.03–3.93 (m,
162 MHz): δ = −22.8 (s, PPh) ppm; MS (ESI): m/z = 1029.31
H, OCH
2
), 3.73–3.66 (m, 2H, OCH ), 3.63–3.59 (m, 1H,
2
+
[M + Na] expected isotopic profile; elemental analysis calcd
OCH ), 3.48–3.44 (m, 1H, OCH ), 3.12 and 2.42 (ABX spin
system with X = P, 2H, CH
J(BX) = 7.5 Hz), 2.02–1.85 (m, 6H, CH
2
2
(
%) for C52 55AuClO P (M = 1007.39): C 62.00, H 5.50;
H
4
r
2
3
2
Pd, J(AB) = 14.5 Hz, J(AX) = 0 Hz,
CH ), 1.80–1.73 (m, 2H,
found (%): C 62.05, H 5.53.
3
2
3
3
CH CH ), 1.13 (t, 3H, CH CH , J = 7.2 Hz), 1.02 (t, 3H,
2
3
2
3
3
3
Rhodium complex 12
CH
2
CH
3
, J = 7.5 Hz), 0.89 (t, 3H, CH
2
CH
3
, J = 7.7 Hz), 0.85 (t,
3
13
1
3
1
1
(
7
H, CH
59.74 (d, arom Cq-O, J(CP) = 11.1 Hz), 158.33 (s, arom Cq-O), (15 mL) was added to a stirred solution of benzophosphole 8
55.27 (s, arom Cq-O), 154.80 (s, arom Cq-O), 152.07–120.73 (0.120 g, 0.15 mmol) in CH Cl (15 mL). After stirring at room
), 76.90 (s, OCH ), 76.85 (s, OCH ), temperature for 0.5 h, the solution was concentrated to about
2
CH
3
, J = 7.5 Hz); C{ H} NMR (CDCl , 125 MHz): δ = A solution of [Rh(acac)(CO) ] (0.040 g, 0.15 mmol) in CH Cl
3
2
2
2
3
2
2
arom. C’s), 77.03 (s, OCH
2
2
2
2
6.71 (s, OCH ), 33.14 (d, Pd–CH , J(CP) = 3.0 Hz), 31.82 (s, 2 mL and MeOH (5 mL) was added. A yellow precipitate
2
2
ArCH
ArCH
2
2
Ar), 31.15 (s, ArCH
Ar, J(CP) = 6.5 Hz), 23.71 (CH
2
2
Ar), 30.92 (s, ArCH Ar), 30.58 (d, formed, which was separated by filtration and then dried
3
1
2
CH
3
), 23.68 (CH
2
CH
3
), under vacuum (0.081 g, yield 52%). H NMR (C
6
D
6
, 500 MHz):
2
3.14 (CH CH ), 23.07 (CH CH ), 11.05 (CH CH ), 10.91 δ = 8.00–7.95 (m, 2H, arom. CH, PPh), 7.81 (t, 1H, arom. CH,
2 3 2 3 2 3
3
1
1
3
3
(
(
CH
CDCl
2
CH
3
), 10.10 (CH
2
CH
3
), 9.95 (CH
2
CH
3
);
P{ H} NMR
6 4
C H , J = 8.0 Hz), 7.71 (d, 1H, arom. CH, calixarene, J =
4
3
, 162 MHz): δ = 25.73 (s, PPh) ppm; MS (ESI): m/z = 7.5 Hz), 7.68 (d, 1H, arom. CH, calixarene, J(PH) = 1.5 Hz),
+
3
1
022.3564 [M − Cl] expected isotopic profile.
7.62 (d, 1H, arom. CH, calixarene, J = 7.5 Hz), 7.20–7.16 (m,
9842 | Dalton Trans., 2017, 46, 9833–9845
This journal is © The Royal Society of Chemistry 2017

 Elaieb, Fethi
Elaieb, Fethi
 Sémeril, David
Sémeril, David
 Matt, Dominique
Matt, Dominique
 Pfeffer, Michel
Pfeffer, Michel
 Bouit, Pierre-Antoine
Bouit, Pierre-Antoine
 Hissler, Muriel
Hissler, Muriel
 Gourlaouen, Christophe
Gourlaouen, Christophe
 Harrowfield, Jack
Harrowfield, Jack