JOURNAL OF
POLYMER SCIENCE
NOTE
WWW.POLYMERCHEMISTRY.ORG
isolated solid by vacuum (10 Torr) at room temperature for
half an hour, before use for the next catalytic run.
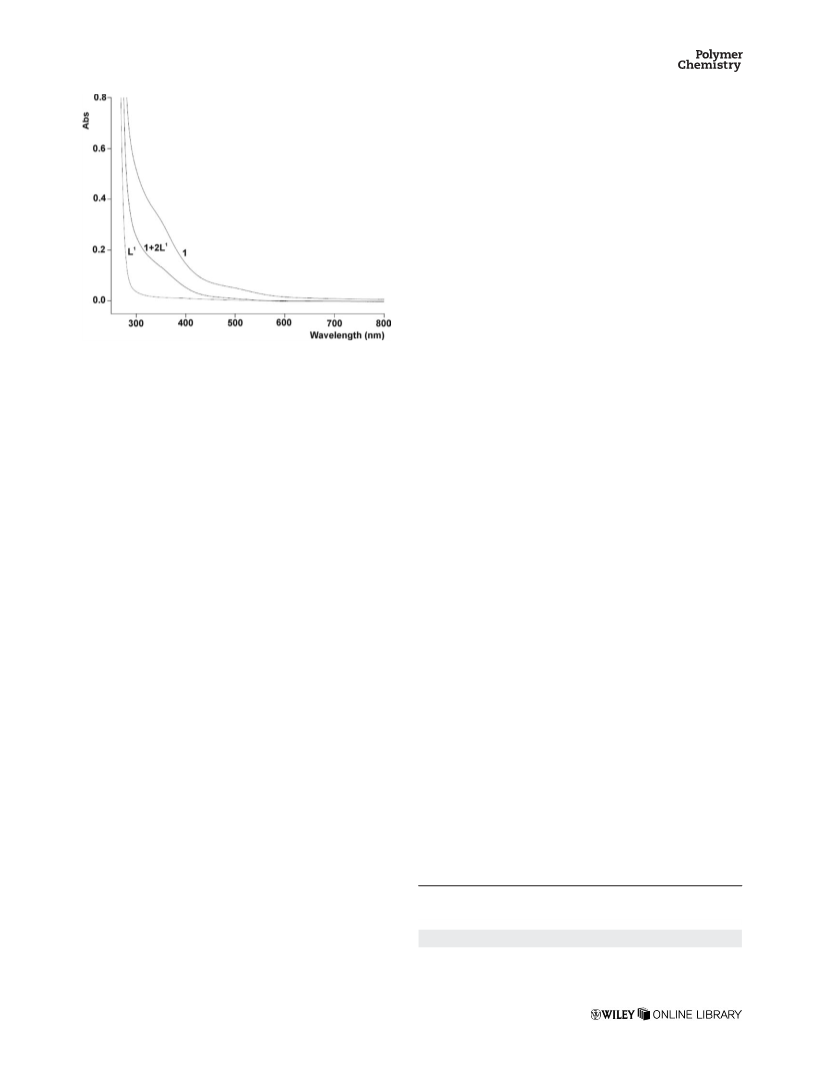
tem, 1þ2L1 can be recovered from the reaction solution by a
simple precipitation step and reused for consecutive catalytic
runs.
Instruments and Characterization
UV–vis spectra were recorded on a Varian-Cary 4000 UV–vis
spectrophotometer, using HPLC-grade CHCl3 solutions with a
concentration of 3 mg/mL. The measurement was performed
in a quartz cuvette with a 10 mm optical path length.
ACKNOWLEDGMENT
The present research was performed in the framework of the
PRIN-2008 project (no. 200898KCKY) entitled: Inorganic nano-
hybrides based on bio-polyesters from renewable resources,
which was financed by the Ministero dell ‘Istruzione, dell’ Uni-
(GPC)-analyses were performed with a GPC system, equipped
with a Waters Binary HPLC 1525 pump; a manual injector
with a six-way valve and a 200 lL loop; three in-series con-
nected Shodex KF-802.5; KF-803 and KF-804 columns (length:
300 mm each; inner diameter: 8.0 mm; 24,500 theoretical
plates; exclusion limit for PS: up to 400,000 g/mol); a RI de-
tector (Waters, mod. 2414). HPLC-grade THF with a water
content of maximal 0.1 vol % was used as eluent at a constant
ꢀ
versita e della Ricerca.
REFERENCES AND NOTES
1 Stahl, S. S. Angew. Chem. Int. Ed. Engl. 2004, 43, 3400–3420.
2 Nishimura, T.; Onoue, T.; Ohe, K.; Uemura, S. J. Org. Chem.
1999, 64, 6750–6755.
ꢀ
flux of 1 mL/min, keeping the columns at 30 C by a thermo-
3 Komano, T.; Iwasawa, T.; Tokunaga, M.; Obora, Y.; Tsuji, Y.
Org. Lett. 2005, 7, 4677–4679.
stat. The GPC system was calibrated using polystyrene (PS) as
standard. It is important to note at this point, that Mn values
for PLA-based materials determined by PS-based GPC-calibra-
tion curves are overestimated, due to the different hydrody-
namic volume of PLA and PS of the same molar mass.
4 Bettucci, L.; Bianchini, C.; Filippi, J.; Lavacchi, A.; Ober-
hauser, W. Eur. J. Inorg. Chem. 2011, 1797–1805.
5 Iwasawa, T.; Tokunaga, M.; Obora, Y.; Tsuji, Y. J. Am. Chem.
Soc. 2004, 126, 6554–6555.
6 Ye, X.; Johnson, M. D.; Diao, T.; Yates, M. H.; Stahl, S. S.
Green Chem. 2010, 12, 1180–1186.
XRPD spectra were acquired on a X’Pert PRO (PANalytical)
powder diffractometer in a 2H range from 5 to 80ꢀ with a
step size of 0.1050ꢀ, using Cu Ka radiation (1.541874 Å).
7 Kakiuchi, N.; Maeda, Y.; Nishimura, T.; Uemura, S. J. Org.
Chem. 2001, 66, 6620–6625.
(GC)-analyses were performed with a Shimadzu 2010 gas
chromatograph equipped with a flame ionization detector
and a 30 m (0.25 mm i.d., 0.25 lm film thickness) VF-
WAXms capillary column, using n-decane as internal stand-
ard. (GC-MS)-analyses were performed with a Shimadzu QP
5000 apparatus, equipped with a 30 m (0.32 mm i.d., 0.50
lm film thickness) CP-WAX 52 CB WCOT-fused silica column.
8 Giachi, G.; Frediani, M.; Oberhauser W.; Passaglia, E.
J. Polym. Sci. Part A: Polym. Chem. 2011, 49, 4708–4713.
9 Bergbreiter, D. E.; Tian, J.; Hongfa, C. Chem. Rev. 2009, 109,
530–582.
10 Lu, J.; Toy, P. H. Chem. Rev. 2009, 109, 815–838.
11 Benaglia, M.; Puglisi, A.; Cozzi, F. Chem. Rev. 2003, 103,
3401–3429.
12 Dickerson, T. J.; Reed, N. N.; Janda, K. D. Chem. Rev. 2002,
102, 3325–3344.
(ICP-MS)-analyses were performed on a Thermo ICP-MS
XSeries 2.
13 Bergbreiter, D. E. Chem. Rev. 2002, 102, 3345–3384.
14 Osburn, P. L.; Bergbreiter, D. E. Prog. Polym. Sci. 2001, 26,
2015–2081.
CONCLUSIONS
The macroligands L1 and L2, bearing an end-capping 4-pyri-
dinemethylene unit, were obtained by a Sn(Oct)2-catalyzed
ROP reaction of l-lactide and e-caprolactone, respectively.
Both macroligands coordinated trans to each other to
Pd(OAc)2 yielding the corresponding homoleptic macrocom-
plexes 1 and 2. Unlike L2, L1 showed, under real aerobic oxi-
dative catalytic conditions, high stability against polymer
chain degradation. From a screening of the catalytic perform-
ance of 1 and 2 versus a reference system [i.e., trans-
[Pd(OAc)2(4-EtPy)2] (4-EtPy ¼ 4-ethylpyridine)] emerged
that: (i) 1 is superior to 2 concerning substrate conversion,
which might be rationalized by a polymer chain flexibility-
driven substrate migration toward the palladium center; (ii)
the addition of two molequivalents of L1 to 1-catalyzed reac-
tions (i.e., 1þ2L1) led to a catalytic system that showed a
superior stability against Pd-black formation compared to
the analogous reference system, due to an efficient polymer
chain-assisted separation of the metal centers from each
other, avoiding thus agglomeration of Pd-centers in the
course of the catalytic reaction; (iii) unlike the reference sys-
15 Bergbreiter, D. E. J. Polym. Sci. Part. A: Polym. Chem. 2001,
39, 2351–2363.
16 Toy, P. H.; Janda, K. D. Acc. Chem. Res. 2000, 33, 546–554.
17 Wentworth, P. J.; Janda, K. D. Chem. Commun. 1999,
1917–1924.
18 Bergbreiter, D. E. Catalysis Today 1998, 42, 389–297.
19 Ferreira, P.; Hayes, W.; Phillips, E.; Rippon, D.; Tsang, S. C.
Green Chem. 2004, 6, 310–312.
20 Dijksman, A.; Arends, I. W. C. E.; Sheldon, R. A. Chem.
Commun. 2000, 271–272.
21 Chung, C. W. Y.; Toy, P.
H J. Comb. Chem. 2007, 9,
115–120.
22 Pozzi, G.; Cavazzini, M.; Quici, S.; Benaglia, M.; Dell’ Anna,
G. Org. Lett. 2004, 6, 441–443.
23 Tanyeli, C.; Gumusꢀ, A. Tetrahedron Lett. 2003, 44,
¨
¨
1639–1642.
24 Hamamoto, H.; Suzuki, Y.; Yamada, Y. M. A.; Tabata, H.;
Takahashi, H.; Ikegami, S. Angew. Chem. Int. Ed. Engl. 2005,
44, 4536–4538.
25 Nlate, S.; Astruc, D.; Neumann, R. Adv. Synth. Catal. 2004,
346, 1445–1448.
6
JOURNAL OF POLYMER SCIENCE PART A: POLYMER CHEMISTRY 2012, 000, 000–000

 Giachi, Guido
Giachi, Guido
 Frediani, Marco
Frediani, Marco
 Oberhauser, Werner
Oberhauser, Werner
 Passaglia, Elisa
Passaglia, Elisa