Y. Shi et al. / Catalysis Communications 18 (2012) 142–146
145
Table 2
smoothly, resulting in the formation of the corresponding α, β-
unsaturated carbonyl compounds in decent selectivity, although the
complete hydrogenation product (3-phenyl-1-propanol) was ob-
served when using cinnamyl alcohol as a substrate (Table 3, entry
4), possibly arisen from hydrogen transfer over the catalyst [25]. For
cyclohexanol, high conversion (89%) could be obtained at higher re-
action temperature and O2 pressure (Table 3, entry 6). 2-Octanol
was also successfully converted under the similar conditions
(Table 3, entry 7). It was worth noting that the Pd1Au1/LDH catalyst
was also effective for the oxidation of non-activated alcohols, such
as aliphatic primary alcohols, but the corresponding acids were
obtained as the main products. The oxidation 1-hexanol can be nearly
completed within 1.5 h at 373 K (Table 3, entry 8). However, Pd1/LDH
and Au1/LDH catalysts afforded 81% and 30% conversions of 1-
hexanol under the same conditions (Table 3, entries 9 and 10). For
1-octanol, 96% conversion could be obtained by prolonging reaction
time to 6 h, which showed that the non-activated aliphatic primary
alcohols were more reactive than secondary alcohols for the Au–Pd
bimetal nano-catalyst (Table 3, entries 7 and 11). In fact, the similar
results have been observed previously over Pd–Au/TiO2 catalysts
[19]. The oxidation of various alcohols by the mono-metal or bimetal-
lic catalysts was also examined in aqueous phase and the results were
shown in Table S3, which demonstrated that the bimetallic catalyst
still afforded much higher catalytic performance than the corre-
sponding monometallic catalyst.
Oxidation of 1-phenylethyl alcohol to acetophenone by various catalysts.a
Entries
Catalysts
Conv. (%)
Sel. (%)
TOF (h−1
)
1
2
3
4
5
6
7
8b
No
–
–
–
LDH
–
–
–
Pd1/LDH
18
33
35
31
10.5
14
>99
>99
>99
>99
>99
>99
72.4
132.6
140.7
124.6
42.2
56.3
Pd1Au0.67/LDH
Pd1Au1/LDH
Pd1Au2/LDH
Au1/LDH
Pd1/LDH+Au1/LDH
a
Reaction conditions: 0.1 g catalyst, 3 ml H2O, substrate/metal=67:1(molar ratio),
353 K, 10 min, O2 atmosphere.
b
The amounts of Pd and Au used in the mixture were the same as those in the
Pd1Au1/LDH catalyst.
alcohol cannot be oxidized in the absence of catalyst or sole LDH as a
catalyst (Table 2, entries 1 and 2). Additionally, the different supports
were found to have a significant influence on the activity of bimetallic
Au–Pd catalysts (Table S2, entries 1–4), which revealed that the base
sites of the LDH and the Au–Pd nanoparticles showed a strong syner-
gistic effect for the oxidation of alcohols [6].
3.3. Reaction kinetics
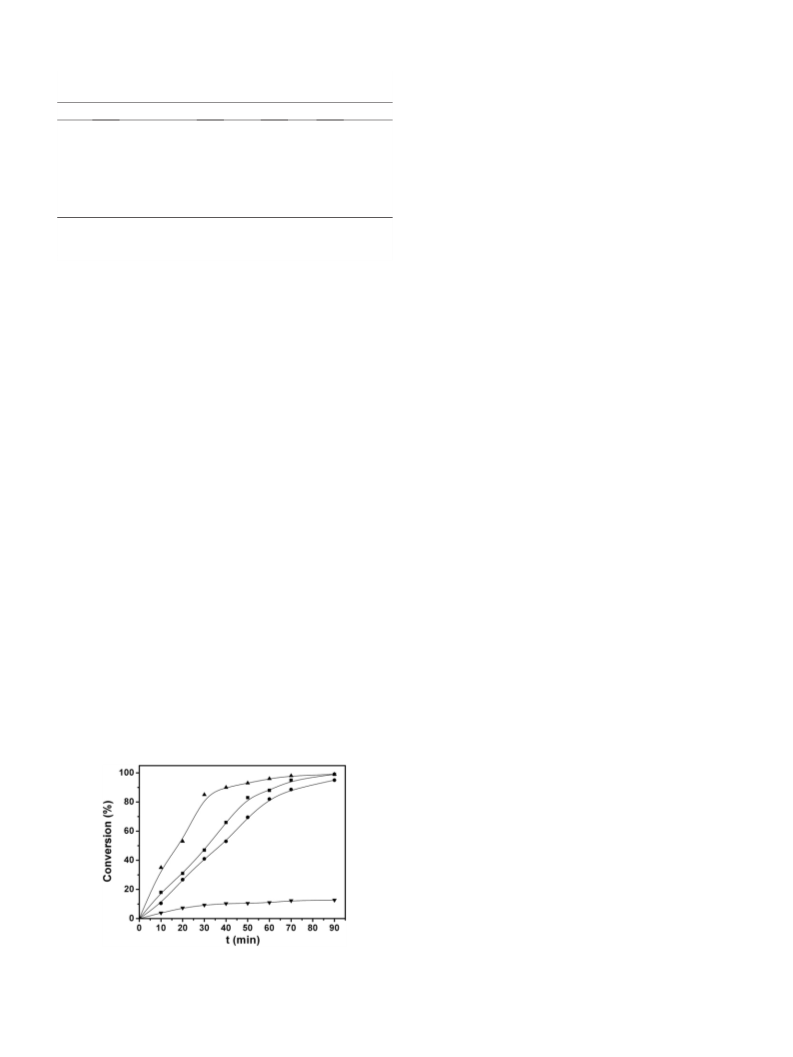
The kinetic curves of Pd1Au1/LDH, Pd1/LDH, Au1/LDH and com-
mercial Pd/C catalysts for oxidation of 1-phenylethyl alcohol were
presented in Fig. 4. At the beginning of 30 min, the catalytic activity
of Pd1Au1/LDH was significantly higher than Pd1/LDH and Au1/LDH.
The Pd1Au1/LDH catalyst gave 85% conversion towards acetophenone
after only 30 min. However, the Au1/LDH and Pd1/LDH catalysts
afforded around 30–45% conversions for the same reaction time. For
the commercial Pd/C, the conversion was only 12% even after reaction
over 1 h, which indicated clearly that the activity of Pd/C catalyst was
lower than other catalysts under the same conditions.
The recyclability of the Pd1Au1/LDH catalyst was also examined for
the oxidation of 1-phenylethyl alcohol. It has been demonstrated the
catalyst exhibited particularly good stability. For example, the catalyt-
ic system gave 95% conversion after the sixth run (Table 3, entry 2).
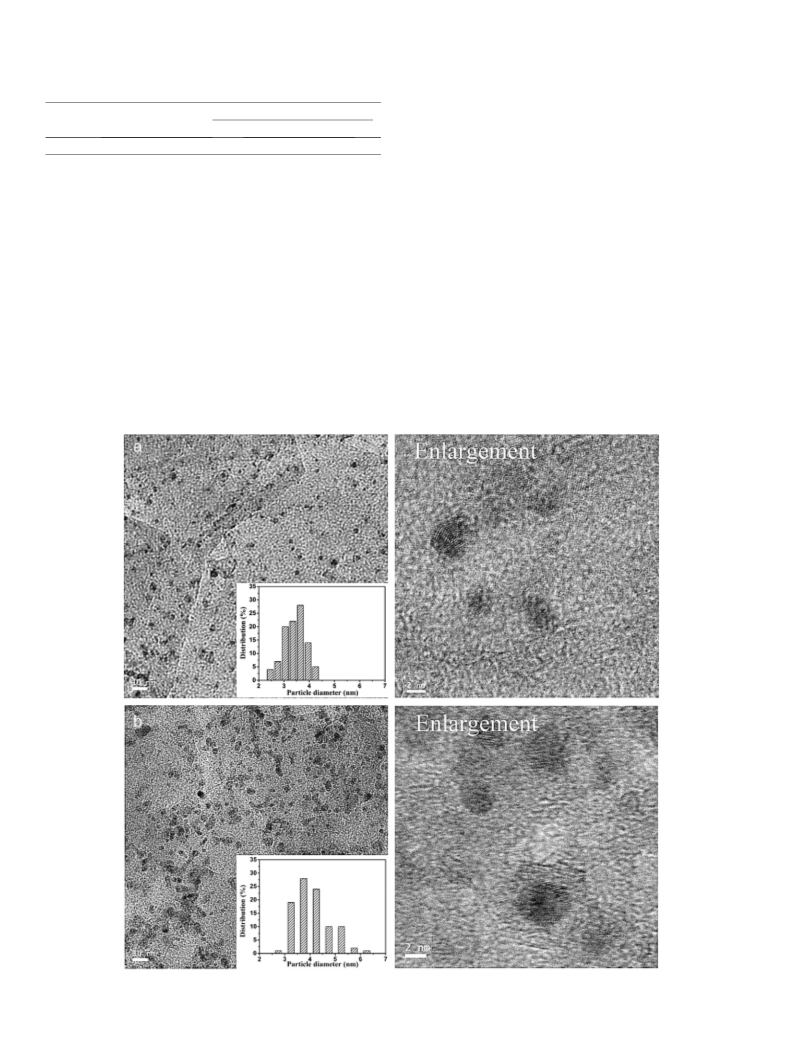
Notwithstanding the average size of the bimetal nanoparticles in-
creased slightly after the sixth run, its catalytic activity was kept con-
stant, possibly because the size of most particles was still less than
5 nm [26].
The effect of metal leaching on the reaction has also been investi-
gated. After the oxidation reaction of 1-phenylethyl alcohol was car-
ried out for 1.5 h and then the reaction mixture was cooled to room
temperature, the organic phase was extracted with diethyl ether
three times and diethyl ether was further removed under vacuum.
Then aqueous phase was filtrated and removed the catalyst. The
new substrate 1-phenylethyl alcohol was added to the recovered
aqueous phase (filtrate) and was oxidized under the same reaction
conditions. It was found that the oxidation of 1-phenylethyl alcohol
did not occur with the filtrate. The elemental analysis showed that
only about 0.7 ppm of Pd and neglectable Au were leached after reac-
tion under the conditions used in this work. This implied that the
leaching Pd cannot contribute to the oxidation of 1-phenylethyl alco-
hol in the present catalytic system.
3.4. The scope and recyclability of the Pd1Au1/LDH catalyst
In next step, the scope of the substrates was also investigated, in
which the various alcohols were chosen as substrates. As shown in
Table 3, the Pd1Au1/LDH catalyst has been examined for the aerobic
oxidation of various alcohols using molecular oxygen as oxidant in
aqueous phase. The present catalyst was very active for the oxidation
of 1-phenylethanol and benzyl alcohol to acetophenone and benzal-
dehyde. Upon reacting for 1.5 h at 353 K, 1-phenylethanol and benzyl
alcohol were completely converted in excellent selectivity (Table 3,
entries 1 and 3). Oxidation of allylic alcohols such as cinnamyl alcohol
(Table 3, entry 4) and geraniol (Table 3, entry 5) also proceeded
4. Conclusions
The aerobic oxidation of alcohols in water has been investigated
using Au and Pd supported on basic Layered Double Hydroxide. The
Au–Pd bimetallic catalyst showed much higher catalytic activity
than the corresponding monometallic catalysts because of the elec-
tronic interaction between Au and Pd. Furthermore, the present cata-
lyst showed an efficient catalytic activity for the oxidation of various
alcohols, especially for that of the non-activated aliphatic primary al-
cohols, presumably due to synergetic interaction between Au and Pd.
Obviously, this catalyst system is green, simple, easily separable, and
recyclable.
Acknowledgments
The authors are grateful for the support from the National Natural
Science Foundation of China (No. 21073058), and Research Fund for
the Doctoral Program of Higher Education of China (20100074110014).
Fig. 4. Time variation of 1-phenylethyl alcohol conversion to acetophenone with
Pd1Au1/LDH(▲), Pd1/LDH(■), Au1/LDH(●) and Pd/C(▼). Reaction conditions: 3 ml
H2O, substrate/metal=67:1(molar ratio), 353 K, O2 atmosphere.

 Shi, Yu
Shi, Yu
 Yang, Hanmin
Yang, Hanmin
 Zhao, Xiuge
Zhao, Xiuge
 Cao, Ting
Cao, Ting
 Chen, Jizhong
Chen, Jizhong
 Zhu, Wenwen
Zhu, Wenwen
 Yu, Yinyin
Yu, Yinyin
 Hou, Zhenshan
Hou, Zhenshan