Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
N. B. Barhate et al. / Tetrahedron Letters 42 (2002) 6031–6033
6033
trimethoxybenzaldehyde
and
3-nitrobenzaldehyde
References
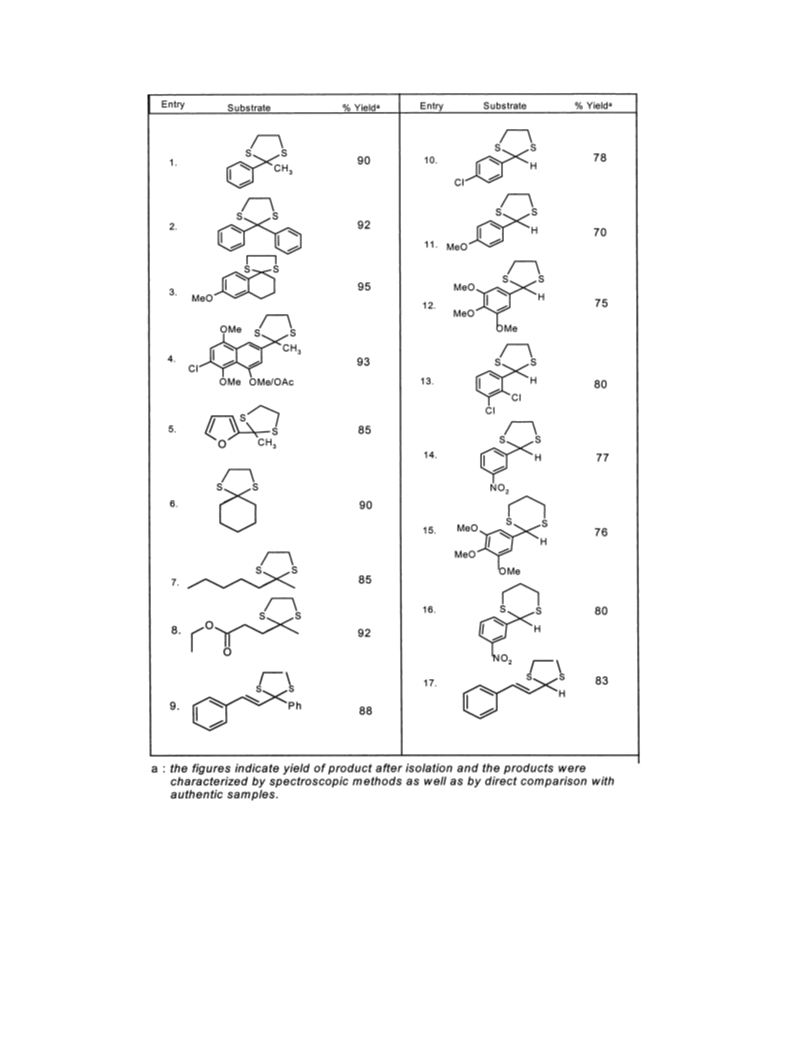
(entries 15 and 16) on treatment with TBHP in
methanol also furnished the cleavage products in good
yields, indicating the generality of the reagent for dithi-
olanes as well as dithianes.
1. Greene, T. W.; Wuts, P. G. M. Protecting Groups in
Organic Synthesis, 3rd ed.; Wiley: New York, 1999; pp.
329–344.
2. Smith, M. B. Organic Synthesis; McGraw-Hill: New
York, 1994; Chapter 7.
3. Kocienski, P. J. Protecting Groups; Thieme: Stuttgart,
1994.
4. (a) Saleur, D.; Bouillon, J.-P.; Portella, C. Tetrahedron
Lett. 1999, 40, 1885–1886; (b) Veyrat, M.; Fantin, L.;
Desmoulins, S.; Petitjean, A.; Mazzanti, M.; Ramasseul,
R.; Marchon, J. C.; Bau, R. Bull. Soc. Chim. Fr. 1997,
134, 703–711; (c) Alonso, E.; Ramon, D. J.; Yus, M. An.
Quim. Int. Ed. 1998, 94, 56–61.
5. Hirano, M.; Ukawa, K.; Yakabe, S.; Clark, J. H.; Mori-
moto, T. Synthesis 1997, 858–860.
6. Hirano, M.; Ukawa, K.; Yakabe, H.; Morimoto, T. Org.
Prep. Proceed. Int. 1997, 29, 480–484.
7. Armstrong, A.; Jones, L. H.; Barsanti, P. A. Tetrahedron
Lett. 1998, 39, 3337–3340.
The dithioacetals were prepared by reported proce-
dures.19 In a typical procedure the dithioacetal (10
mmol) was dissolved in methanol (10 ml) and TBHP
(20 mmol, 2 equiv.) was added to the reaction mixture
which was refluxed until completion of reaction. After
completion (4–6 h as monitored by TLC), methanol
was removed under reduced pressure. The residue was
taken up in ethyl acetate, washed with water, dried over
sodium sulfate and concentrated to afford the crude
product which was purified by column chromatography
(silica gel; petroleum ether: EtOAc) to furnish the pure
product in yields as shown in Table 1. All compounds
in Table 1 were characterized by direct comparison
with authentic samples as well as IR and NMR
spectroscopy.
8. Ballini, R.; Petrini, M. Synthesis 1990, 336.
9. (a) Stork, G.; Zhao, K. Tetrahedron. Lett. 1989, 30, 287;
(b) Liu, H. J.; Wiszniewski, V. Tetrahedron Lett. 1988,
29, 5471.
10. Vakalopoulos, A.; Hoffmann, H. M. R. Org. Lett. 2001,
3, 2185–2188.
11. Mondal, E.; Sahu, P. R.; Bose, G.; Khan, T. Tetrahedron
Lett 2002, 43, 2843 and references cited therein.
12. Saleur, D.; Bouillon, J.-P.; Portella, C. Tetrahedron Lett.
2000, 41, 321–324.
13. Bouillon, J.-P.; Saleur, D.; Portella, C. Synthesis 2000,
843–849.
14. Kolb, H. C.; Ley, S. V.; Slawin, A. M. Z.; Williams, D.
J. J. Chem. Soc., Perkin Trans. 1 1992, 2735.
15. Barhate, N. B.; Gajare, A. S.; Wakharkar, R. D.;
Bedekar, A. V. Tetrahedron Lett. 1998, 39, 6349.
16. Barhate, N. B.; Gajare, A. S.; Wakharkar, R. D.;
Bedekar, A. V. Tetrahedron 1999, 55, 11127.
17. Barhate, N. B.; Gajare, A. S.; Sudalai, A.; Wakharkar,
R. D. Tetrahedron Lett. 1997, 38, 653.
In summary, we have devised a simple and convenient
method for the demasking of 1,3-dithiolanes and dithi-
anes under neutral conditions in good to excellent yield.
The advantages of this protocol are that it provides an
economically viable, non-hazardous and efficient
methodology using a readily available reagent. Applica-
tion of commercially available TBHP (aq. 70%) offers
experimentally simple conditions wherein no special
precautions (inert atmosphere etc.) are necessary. On
environmental grounds the use of methanol as the
solvent provides an added advantage as it is preferred
over chlorinated solvents such as dichloromethane and
carbon tetrachloride, which are conventionally used for
heterogeneous catalysis. The deprotection conditions
being neutral and chemoselective are general for 1,3-
dithiane and thiolanes of cyclic, acyclic and aliphatic
aromatic, aldehydes and ketones as demonstrated in
this communication.
18. Barhate, N. B.; Sasidharan, M.; Sudalai, A.; Wakharkar,
R. D. Tetrahedron Lett. 1996, 37, 2067.
Acknowledgements
19. Vogel’s Textbook of Practical Organic Chemistry, 5th ed.;
Longman: Harlow, 1989; p. 787.
N.B.B., P.D.S. and V.A.M. are thankful to the CSIR
for their research fellowships.

 Barhate, Nivrutti B
Barhate, Nivrutti B
 Shinde, Popat D
Shinde, Popat D
 Mahajan, Vishal A
Mahajan, Vishal A
 Wakharkar, Radhika D
Wakharkar, Radhika D