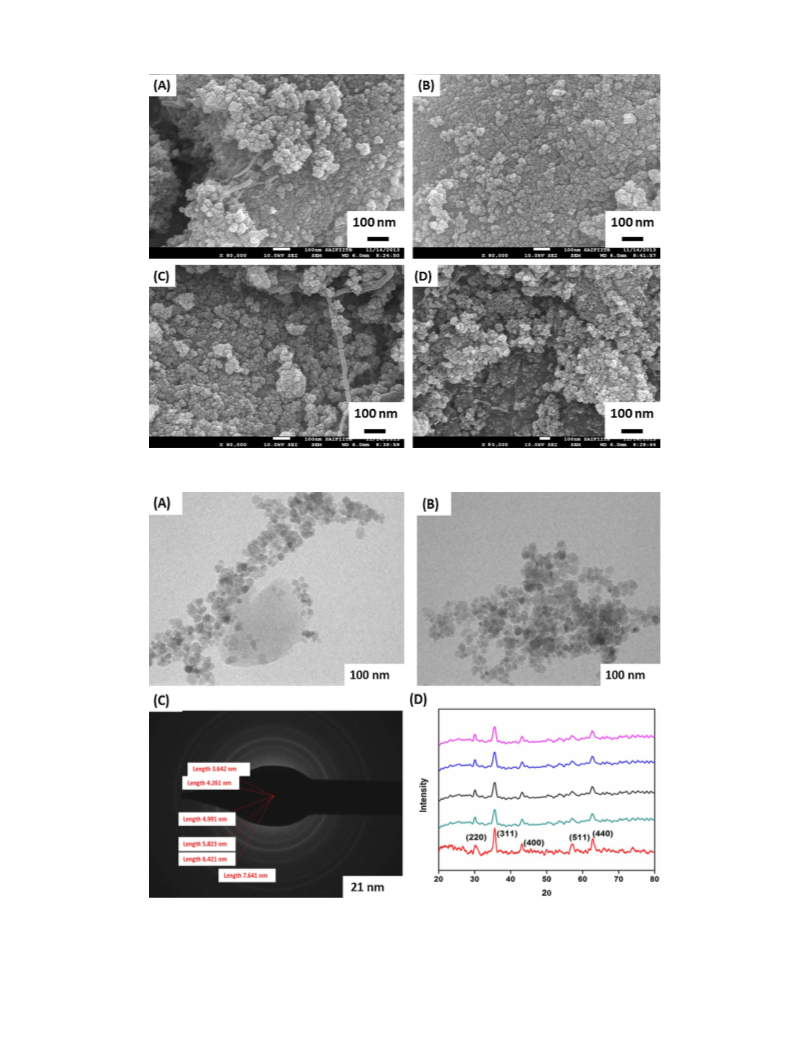
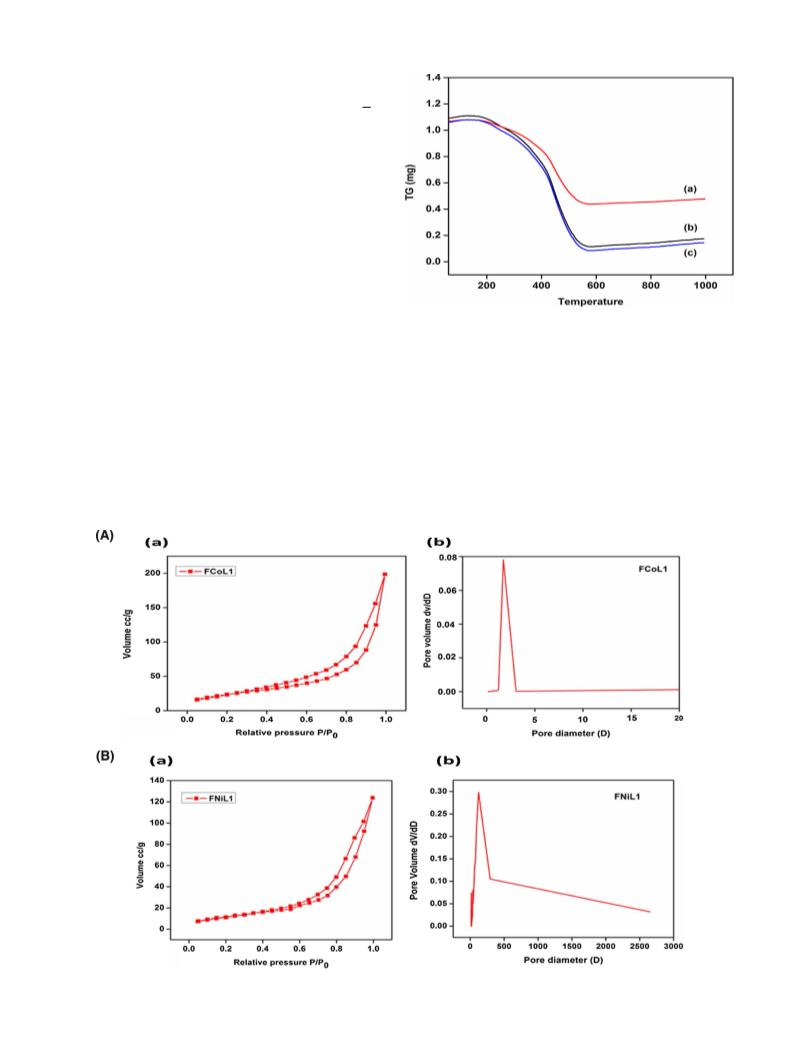
Journal of Molecular Catalysis A: Chemical 409 (2015) 42–49
Journal of Molecular Catalysis A: Chemical
journal homepage: www.elsevier.com/locate/molcata
Immobilized magnetic nano catalyst for oxidation of alcohol
a
b
b
a,∗
Pooja B. Bhat , Ravindra Rajarao , Veena Sahajwalla , Badekai Ramachandra Bhat
a
Catalysis and Materials Chemistry Laboratory, Department of Chemistry, National Institute of Technology Karnataka, Surathkal, Srinivasa Nagar 575025,
India
b
Centre for Sustainable Materials Research and Technology (SMaRT@UNSW), School of Materials Science and Engineering, UNSW Australia, Sydney, NSW
052, Australia
2
a r t i c l e i n f o
a b s t r a c t
Article history:
Received 14 April 2015
Received in revised form 20 July 2015
Accepted 12 August 2015
Available online 17 August 2015
Covalent attachment of Schiff base on magnetic nanoparticles yielded good selectivity for oxidation of
alcohols. The ferromagnetic interaction in the complex added comprehensive advantage in enhancing the
catalytic activity of the nanocatalyst. A greener approach for alcohol oxidation was achieved in solventless
method with good yield (>78%). Leaching experiments confirmed a strong interaction between magnetic
support and complex. The catalyst showed significant conversion even after 5 catalytic runs.
©
2015 Elsevier B.V. All rights reserved.
Keywords:
Covalent attachment
Schiff base
Magnetic
Oxidation of alcohols
1
. Introduction
within the framework enhancing magnetic and catalytic activity
of the immobilized catalyst.
Metal(II) complexes coordinating with organic base ligands
Schiff base ligand framework forms stable complex depending
on the flexibility of the linker group [9]. Schiff base ligands possess
the ability to chelate many metal ions and form stable complexes.
Researchers have proved that Co(II) and Ni(II) complexes binds with
molecular oxygen and the resultant dioxygen–Co and Ni complexes
exhibits efficient oxidation reaction [10]. The immobilized supports
have shown better control on efficiency of the catalyst due to the
microenvironment provided by the support. Hence, immobilized
metal complexes have found its advantage in selective oxidation of
alcohols with hydrogen peroxide oxidant without any organic sol-
vent, phase transfer catalyst or additive. The redox potential of Co
and Ni metal ions influences the decomposition of hydrogen per-
oxide by reducing its energy of activation and assists in enhancing
the catalytic activity of the Schiff bases [11].
Immobilization of Schiff complexes by covalent attachment
has proven to be the most effective method for anchoring the
metal complexes. Hu et al. have recently reported that Schiff
base molybdenum(VI) complexes immobilized onto zirconium
poly(styrene-phenylvinylphosphonate)-phosphate showed com-
parable or even higher conversion and chemical selectivity for
epoxidation of olefins compared with other heterogeneous cata-
lysts [12]. The catalysts were easily recovered by simple filtration
and were reused with little loss of activity. The potential use of the
catalyst for oxidation was reported by Jiang et al. with increased
catalytic activity by immobilizing Co salophen complex on
montmorillonite [13]. Researchers have explored that higher cat-
alytic activity is observed for oxidation of alcohols to carbonyls
including Schiff bases are well-known to interact with molecular
oxygen to form dioxygen adducts and have extensively been used
as efficient and selective homogeneous catalysts for a variety of oxi-
dation reactions in recent years [1,2]. However, the main drawback
of these homogeneous catalysts is the necessity of their separation
from the reaction mixture at the end of the reaction. Immobiliza-
tion of Schiff base complexes to the magnetic nanoparticle support
via covalent attachment provides facile separation of the catalyst
during the reaction by external magnet [3]. The magnetic support
offers high catalytic efficiency and better recycling without having
the inherent problems of leaching of complex/ligand during the
reaction.
In recent years, aromatic linkers have been extensively used
to immobilize ligands on nanoparticle via click reaction [4,5]. The
magnetic superexchange observed in the aromatic linkers such as
m-phenylene, p-phenylene provides stable ferromagnetic interac-
tions between immobilized ligands and the support [6,7]. Recent
experimental and theoretical studies on dinuclear metal(II) com-
plexes have shown higher amount of ferromagnetic interaction
with bisbidentate bridging ligands bearing m-phenylene linkers
[
∗ Corresponding author. Fax: +91 824 2474033.
1
381-1169/© 2015 Elsevier B.V. All rights reserved.

 Bhat, Pooja B.
Bhat, Pooja B.
 Rajarao, Ravindra
Rajarao, Ravindra
 Sahajwalla, Veena
Sahajwalla, Veena
 Bhat, Badekai Ramachandra
Bhat, Badekai Ramachandra