8 of 8
SHU ET AL.
[8] a) L. Y. Wang, J. Li, Y. Lv, G. D. Zhao, S. Gao, Appl. Organomet.
Chem. 2012, 26, 37. b) N. Wang, R. Liu, J. Chen, X. Liang, Chem.
Commun. 2005, 42, 5322. c) J. M. Hoover, S. S. Stahl, J. Am.
Chem. Soc. 2011, 133, 16901.
the oxidation of NO to NO2 can be easily carried out in the
presence of molecular oxygen.
4 | CONCLUSIONS
[9] a) P. R. Likhar, R. Arundhathi, S. Ghosh, M. L. Kantam, J. Mol.
Catal. A‐Chem. 2009, 302, 142. b) M. A. Chari, K. Syamasundar,
Synthesis 2005, 5, 708.
In conclusion, a Zr‐based MOF with an organic linker of
2,2′‐bipyridine‐5,5′‐dicarboxylic acid (UiO‐67) was prepared
[10] J. Mao, X. Hu, H. Li, Y. Sun, C. Wang, Z. Chen, Green Chem.
2008, 10, 827.
solvothermally.
A
novel UiO‐67‐FeCl3 catalyst was
synthesized by treating UiO‐67 with FeCl3·6H2O. Then The
UiO‐67‐FeCl3 was utilized as an efficient catalyst for the pro-
motion of aerobic oxidation of alcohols and aerobic benzylic
oxidations. The pyridine moiety of the UiO‐67‐FeCl3 catalyst
also served as an organic base, which allowed the amount of
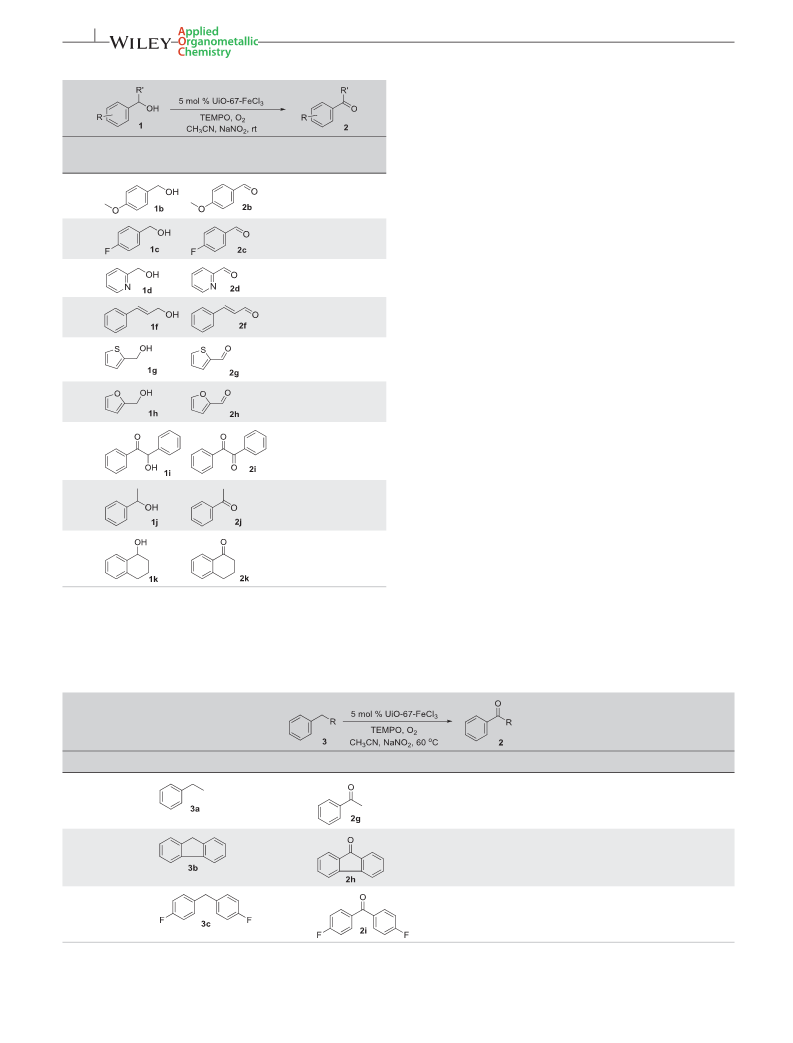
basic additive to be reduced. A variety of alcohols were tested
and converted to their corresponding ketones in good yields.
In addition, similar reaction conditions were extended to the
aerobic oxidation of benzylic carbons to phenomes, using
molecular oxygen as the oxidant. The initial catalytic activity
of the UiO‐67‐FeCl3 catalyst was maintained after five con-
secutive reaction cycles. Hot filtration tests and ICP‐AES
analyses of the solution suggested the extremely low leakage
of the iron content during the reaction process. Further stud-
ies will aim at extending the applications of these MOF cata-
lysts to the oxidation of other substrates.
[11] S. M. Cohen, Chem. Sci. 2010, 1, 32.
[12] a) J. D. Evans, C. J. Sumby, C. J. Doonan, Chem. Soc. Rev. 2014,
43, 5933. b) K. Manna, T. Zhang, W. Lin, J. Am. Chem. Soc.
2014, 136, 6566. c) A. Dhakshinamoorthy, A. M. Asiri, H. Garcia,
Chem. – Eur. J. 2016, 22, 8012. d) A. Dhakshinamoorthy,
M. Alvaro, H. Garcia, Catal. Sci. Technol. 2011, 1, 856.
[13] a) C. J. Doonan, W. Morris, H. Furukawa, O. M. Yaghi, J. Am.
Chem. Soc. 2009, 131, 9492. b) J. Canivet, S. Aguado,
Y. Schuurman, D. Farrusseng, J. Am. Chem. Soc. 2013, 135, 4195.
[14] a) H. Duan, M. Li, G. Zhang, J. R. Gallagher, Z. Huang, Y. Sun,
Z. Luo, H. Chen, J. T. Miller, R. Zou, A. Lei, Y. Zhao, ACS Catal.
2015, 5, 3752. b) S. Øien, G. Agostini, S. Svelle, E. Borfecchia,
K. A. Lomachenko, L. Mino, E. Gallo, S. Bordiga, U. Olsbye,
K. P. Lillerud, C. Lamberti, Chem. Mater. 2015, 27, 1042.
c) P. Neves, A. C. Gomes, T. R. Amarante, F. A. A. Paz,
M. Pillinger, I. S. Gonçalves, A. A. Valente, Microporous Mesopo-
rous Mater. 2015, 202, 106. d) P. Valvekens, E. D. Bloch,
J. R. Long, R. Ameloot, D. E. De Vos, Catal. Today 2015, 246,
55. e) K. Leus, Y. Y. Liu, M. Meledina, S. Turner, G. Van Tendeloo,
P. Van Der Voort, J. Catal. 2014, 316, 201. f) Y. Y. Liu, R. Decadt,
T. Bogaerts, K. Hemelsoet, A. M. Kaczmarek, D. Poelman,
M. Waroquier, V. Van Speybroeck, R. Van Deun, P. Van der Voort,
J. Phys. Chem. C 2013, 117, 11302. g) A. E. Platero‐Prats,
A. B. Gómez, L. Samain, X. Zou, B. Martín‐Matute, Chem. – Eur.
J. 2015, 21, 861.
ACKNOWLEDGEMENTS
We thank the Beijing Municipal Natural Science Foundation
(Grant No. 2172037), the National Natural Science Founda-
tion of China (No. 51503016) and Fundamental Research
Funds for the Central Universities (Grant No. FRF‐TP‐16‐
004A3) for financial support. Xin Shu also thanks BUCT
Fund for Disciplines Construction and Development
(No. XK1529) for financial support.
[15] K. Chen, P. Zhang, Y. Wang, H. Li, Green Chem. 2014, 16, 2344.
[16] H. Feia, S. M. Cohen, Chem. Commun. 2014, 50, 4810.
[17] T. Toyao, K. Miyahara, M. Fujiwaki, T. H. Kim, S. Dohshi,
Y. Horiuchi, M. Matsuoka, J. Phys. Chem. C 2015, 119, 8131.
[18] Y. Zhang, Z. X. Zhang, T. B. Li, X. G. Liu, B. S. Xu, Spectrochim.
Acta A 2008, 70, 1060.
REFERENCES
[1] Q. Cao, L. M. Dornan, L. Rogan, N. L. Hughes, M. J. Muldoon,
Chem. Commun. 2014, 50, 4524.
[2] C. Parmeggiani, F. Cardona, Green Chem. 2014, 14, 547.
SUPPORTING INFORMATION
[3] a) A. Xie, X. Zhou, L. Feng, X. Hu, W. Dong, Tetrahedron 2014,
70, 3514. b) N. Gunasekaran, P. Jerome, S. W. Ng, E. R. T. Tierink,
R. Karvembu, J. Mol. Catal. A‐Chem. 2012, 353, 156.
Additional Supporting Information may be found online in
the supporting information tab for this article.
[4] Y. Qi, Y. Luan, J. Yu, X. Peng, G. Wang, Chem. – Eur. J. 2015, 21,
1589.
How to cite this article: Shu X, Yu Y, Jiang Y,
Luan Y, Ramella D. Direct synthesis of Fe(III)
immobilized Zr‐based metal–organic framework for
aerobic oxidation reaction. Appl Organometal Chem.
[5] S. E. Allen, R. R. Walvoord, R. Padilla‐Salinas, M. C. Kozlowski,
Chem. Rev. 2013, 113, 6234.
[6] M. Stratakis, H. Garcia, Chem. Rev. 2012, 112, 4469.
[7] a) J. M. Hoover, B. L. Ryland, S. S. Stahl, ACS Catal. 2013, 3,
2599. b) M. F. Semmelhack, C. R. Schmid, D. A. Cortes,
C. S. Chou, J. Am. Chem. Soc. 1984, 106, 3374.

 Shu, Xin
Shu, Xin
 Yu, Ying
Yu, Ying
 Jiang, Yi
Jiang, Yi
 Luan, Yi
Luan, Yi
 Ramella, Daniele
Ramella, Daniele