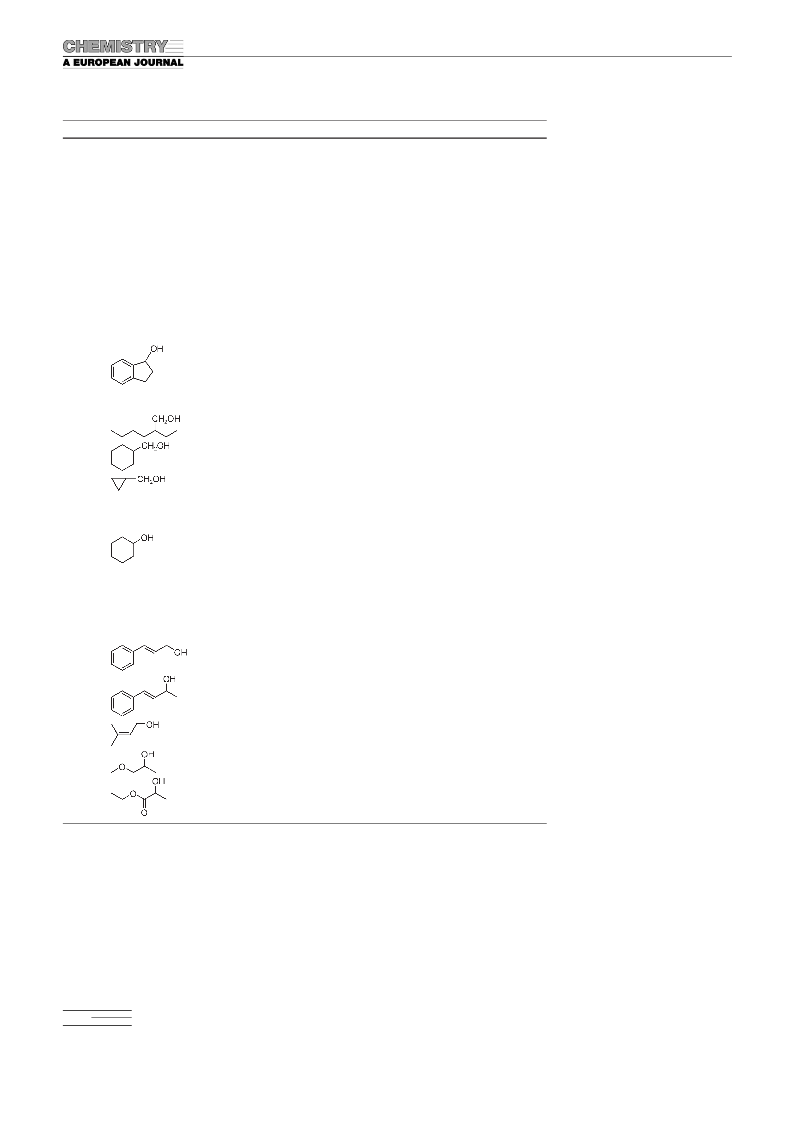TEMPO/HCl/NaNO2-Catalyzed Oxidation of Alcohols
FULL PAPER
TEMPO and NaNO2 in catalytically oxidizing benzyl alco-
hol to benzaldehyde. The observations could be rationalized
in terms of the positive halogen species being able to oxidize
TEMPO/TEMPOH to the oxoammonium cation (TEMPO+).
However, we were surprised to find that a few less oxidative
halogen-containing compounds, such as HCl and HBr, in
combination with TEMPO and NaNO2, were also effective
in promoting the catalysis. Of particular interest was the
finding that hydrochloric acid, a very inexpensive and readi-
ly available inorganic acid, cooperated exquisitely with
NaNO2/TEMPO in facilitating aerobic oxidation of the sub-
strate to the corresponding aldehyde under mild conditions
(entry 10). In the light of this result, we also screened some
other commonly used acids. Unfortunately, the majority of
these acids produced only a low conversion of the substrate;
only a few acids, for example H2SO4, gave a moderate con-
version. However, we observed a critical fact from these re-
sults, namely that HCl and HBr, two hydrohalogenide acids,
yielded extremely high substrate conversions and selectivi-
ties. Thus, we presumed that acidic reaction conditions and
the existence of chlorine or bromine, whether positive, nega-
tive, or neutral, were crucial to obtaining the overall catalyt-
ic activity of the TEMPO/NaNO2 catalyst. The role of the
acid was assumed to be donation of H+ to NaNO2 to gener-
ate NO/NO2. The effect of the halide anions on the catalysis
may stem from their reaction with NO2 to generate oxidiz-
ing species such as NOX (X = Cl, Br),[25] which are known
to oxidize TEMPO/TEMPOH to TEMPO+.[26,27] To verify
the importance of the chloride or bromide anions in the cat-
alysis, we carried out the oxidation of benzyl alcohol under
standard reaction conditions with a variety of acids in the
presence of NaCl or the quaternary ammonium chloride
BnNMe3Cl (Table 1, entries 22–27). The results demonstrat-
ed that ClÀ greatly promoted the oxidative conversion of
benzyl alcohol to benzaldehyde compared to the reactions
without ClÀ (Table 1, entries 18–21). Interestingly, the use of
NaNO3 in place of NaNO2 led to a very slow reaction over
10 h, with complete conversion with about 100% selectivity
being achieved by prolonging the reaction time to 24 h
(entry 15). These preliminary studies revealed that acidic
conditions and chloride or bromide anions had beneficial
impacts on the catalytic oxidation. Importantly, the results
clearly demonstrated that oxidizing positive-halogen-con-
taining compounds were not essential to drive the TEMPO/
NaNO2-based catalytic system in the aerobic alcohol oxida-
tion. Being composed of a proton and chloride anion, hydro-
chloric acid was thus considered to be a promising co-cata-
lyst in combination with TEMPO and NaNO2 for the mild
and highly selective oxidation of a broad range of alcohols.
During the screening of a variety of reaction conditions,
which included varying the amount of catalyst and the sol-
vent used, we found that the catalyst system TEMPO/HCl/
NaNO2 could be applied in a wide range of solvents, such as
ClCH2CH2Cl, EtOAc, CH3CN, HOAc, and PhF, for efficient
conversions of benzyl alcohol in air at atmospheric pressure
and ambient temperature (without dioxygen or air bub-
bling).[28]
TEMPO/HCl/NaNO2-catalyzed aerobic oxidation of alcohol
substrates: Having established the optimal conditions for
the transition-metal-free catalysis, we next examined the
range of alcohols to which this catalytic aerobic oxidation
[Eq. (1)] could be applied. As revealed in Table 2, all ben-
zylic alcohols were converted into the corresponding benzal-
dehydes in high isolated yields (entries 1–13). The rates of
these benzylic alcohol oxidations were not significantly af-
fected by the electronic properties of the substituents on the
benzene ring, an observation that was seemingly at variance
with previous studies.[7c,11a,c] Similarly, activated secondary
aryl alcohols, such as 1-phenylethanol, 1-phenyl-1-propanol,
1-indanol, and benzhydrol, could be smoothly oxidized to
the corresponding acetophenone, propiophenone, 1-inda-
none, and benzophenone, respectively, in near quantitative
isolated yields (entries 15–18). We were pleased to find that
primary aliphatic alcohols, which remain difficult recalci-
trant substrates in many aerobic oxidation protocols,[4c,11]
were oxidized to the expected aldehydes with high conver-
sions and selectivities. For example, complete oxidations of
1-octanol, 2-ethyl hexanol, cyclohexyl carbinol, and cyclo-
propyl carbinol could be accomplished at ambient tempera-
ture and pressure, directly exploiting dioxygen from the air
without bubbling (entries 19–22). 2-Phenylethanol could
also be oxidized to the corresponding aldehyde with high se-
lectivity (entry 23). Secondary aliphatic alcohols could be
completely converted to the corresponding ketones in high
isolated yields under the standard mild conditions (en-
tries 24 and 25). In the case of cyclohexanol, slower oxida-
tion requiring 24 h even with method D (entry 26) was ob-
served, possibly as a result of its notable steric effect.[29] The
use of HBr in place of HCl led to completion of this reac-
tion with 100% selectivity in 14 h (entry 26). Of particular
interest is the fact that 4-(methylthio)benzyl alcohol and 2-
thiophene methanol (entries 27 and 28), which are usually
regarded as difficult substrates in most aerobic oxidations
involving transition metals due to their strong coordinating
ability, were also very smoothly converted into the corre-
sponding aldehydes in high yield. In entries 29 and 30, the
oxidation reaction was applied to pairs of substrates, both of
which bore groups susceptible to oxidation, namely hydroxy
and sulfide groups. To our delight, the oxidation system
highly selectively oxidized the hydroxy groups to carbonyl
groups, whereas the sulfide groups remained intact. More-
over, sulfide groups, which are not compatible with transi-
tion-metal-catalyzed oxidation methodology, did not have a
deleterious effect on the present oxidation reactions. In the
case of 3-pyridylmethanol, slow oxidation was observed and
the reaction was incomplete even after 24 h using method C
(entry 31). We assume that the HCl was partially neutralized
by the basic pyridine. Allylic alcohols such as cinnamyl alco-
Chem. Eur. J. 2008, 14, 2679 – 2685
ꢀ 2008 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
2681

 Wang, Xinliang
Wang, Xinliang
 Liu, Renhua
Liu, Renhua
 Jin, Yu
Jin, Yu
 Liang, Xinmiao
Liang, Xinmiao