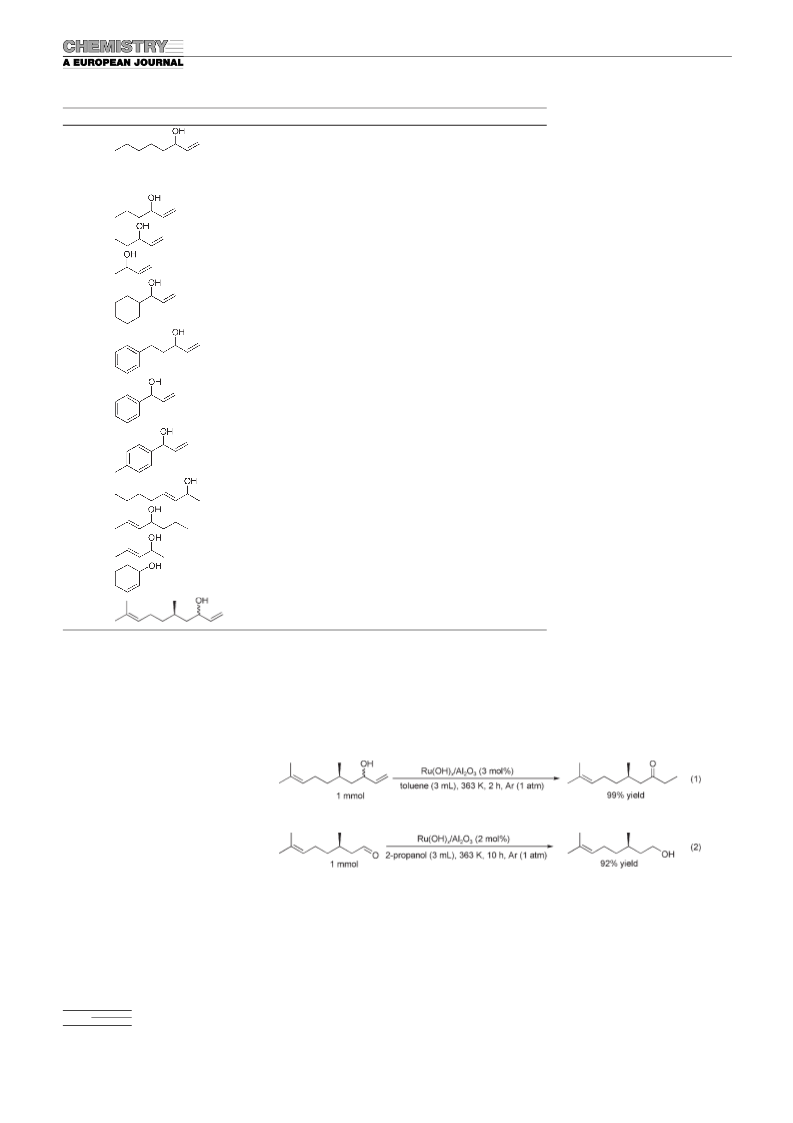N. Mizuno et al.
On the basis of the above results, we propose a possible re-
action mechanism for the present Ru(OH)x/Al2O3-catalyzed
reduction of allylic alcohols (Scheme 1). Initially, the catalyt-
saturated alcohols. Furthermore, the catalyst/product separa-
tion can easily be carried out and the Ru(OH)x/Al2O3 is re-
cyclable.
Experimental Section
General: NMR spectra were recorded
on a JEOL JNM-EX-270 instrument.
1H and 13C NMR spectra were mea-
sured at 270 and 67.8 MHz, respective-
ly, in [D1]chloroform with TMS as an
internal standard. 2H NMR spectra
were measured at 41.25 MHz using
[D6]benzene as an external standard.
GC analyses were performed on a Shi-
madzu GC-2014 instrument using
a
flame ionization detector equipped
with a DB-WAX capillary column (in-
ternal diameter=0.25 mm, length=
30 m). Mass spectra were recorded on
a Shimadzu GCMS-QP2010 equipped
with a TC-5HT capillary column (in-
ternal diameter=0.25 mm, length=
Scheme 1. A possible reaction mechanism for Ru(OH)x/Al2O3-catalyzed reduction of allylic alcohols in the
presence of 2-propanol.
30 m). Reagents and substrates
(except for 5a–8a and 13a–15a) were
obtained commercially from Tokyo
Kasei, Aldrich, and Fluka (reagent
grade) and used without further purifi-
ically active ruthenium monohydride species is formed by
the reaction of ruthenium hydroxide species with an alcohol
(allylic alcohol or 2-propanol) (Scheme 1a).[8] The 1,4-addi-
tion of the hydride species to the a,b-unsaturated carbonyl
compound[13] formed by the reaction of the ruthenium hy-
droxide species with an allylic alcohol yielding the s-enolate
species is followed by intermolecular ligand exchange with
an alcohol (allylic alcohol or 2-propanol) to give the corre-
sponding saturated carbonyl compound as an intermediate
(Scheme 1b).[9a,14] Then the MPV-type reduction of the inter-
mediate carbonyl compound proceeds to give the corre-
sponding saturated alcohol (Scheme 1c). It was confirmed
that Al2O3 and Al2O3 treated with an aqueous NaOH solu-
tion do not catalyze the MPV-type reduction of 1c under
the present conditions, suggesting that the catalytic activity
for the MPV-type reduction originates from ruthenium spe-
cies.[15]
Whereas the formation rate of the final saturated alcohol
in the reduction of 1a in 2-deuterio-2-propanol was much
lower than that in 2-propanol (entry 1 in Table 1 versus
entry 3 in Table 3), the rate of the initial isomerization step
in 2-deuterio-2-propanol (R=22.6mmminÀ1) was fairly simi-
lar to that in 2-propanol (26.1mmminÀ1). This suggests that
the MPV-type reduction (Scheme 1c) is the rate-determining
step for this transformation.[16]
cation. 2-Propanol (Kanto) and 2-deuterio-2-propanol (Cambridge Iso-
tope Laboratory) were particularly carefully purified (degassed) before
use.[17] Al2O3 (KHS-24, BET surface area 160 m2 gÀ1), TiO2 (ST-01,
316 m2 gÀ1), SiO2 (CARiACT Q-10, 273 m2 gÀ1), Fe3O4 (Cat. No. 310069,
6.8 m2 gÀ1), and ZnO (Cat. No. 37002-95, 2.9 m2 gÀ1) were obtained from
Sumitomo Chemical, Ishihara Sangyo Kaisya, Fuji Silysia, Aldrich, and
Nacalai Tesque, respectively. Supported metal catalysts (5 wt%) were
supplied by NE Chemcat. Ru–HAP (ruthenium on HAP, 9.1 wt%) was
purchased from Wako. Compounds 5a–8a and 13a were synthesized by
Grignard reaction of the corresponding aldehydes with vinylmagnesium
bromide.[9g] Compound 14a was synthesized by oxidation of 1a[18] fol-
lowed by reduction with lithium aluminum deuteride.[19] Compound 15a
was synthesized according to the literature procedures.[8c,12] See the Sup-
porting Information for the synthetic procedures for the allylic alcohols
5a–8a, and 13a and the deuterated compounds 14a and 15a.
Preparation of supported ruthenium hydroxide catalysts: The supported
ruthenium hydroxide catalysts were prepared by the procedure reported
previously.[8] Al2O3 powder (2.0 g) calcined at 823 K for 3 h was stirred
vigorously with an aqueous solution (60 mL) of RuCl3 (8.3mm) at room
temperature. After 15 min, the pH of the solution was adjusted slowly to
13.2 by addition of an aqueous NaOH solution (1.0m) and the resulting
slurry was stirred for 24 h. The solid was filtered off, washed with a large
amount of water, and dried in vacuo; yield of Ru(OH)x/Al2O3 2.1 g (dark
green powder, ruthenium content 2.0–2.1 wt%). The XRD pattern of
Ru(OH)x/Al2O3 was the same as that of the parent Al2O3 support and no
signals from Ru metal (clusters) and RuO2 were observed. Particles of
Ru metal (clusters) and RuO2 were not detected by TEM. The binding
energies of Ru 3d5/2 and Ru 3p3/2 of Ru(OH)x/Al2O3 (XPS) were detected
at 281.8 (full width at half maximum (FWHM) 2.4 eV) and 463.5 eV
(FWHM 4.7 eV), respectively, showing that the oxidation state of the
ruthenium species in Ru(OH)x/Al2O3 is +3.[20] The IR spectrum showed
a very broad n(OH) band in the range 3000–3700 cmÀ1. These facts sug-
gest that ruthenium(III) hydroxide is highly dispersed on Al2O3.
A
Conclusion
Ru(OH)x/Al2O3-catalyzed reduction: All operations for the reactions
were performed in a glove box under Ar. Ru(OH)x/Al2O3 (2 mol% Ru),
1a (1 mmol), and 2-propanol (3 mL) were placed successively in a Pyrex
glass vial . The reaction mixture was stirred vigorously with a Teflon-
coated magnetic stir bar (800 rpm) at 363 K (bath temperature) under Ar
Ru(OH)x/Al2O3 can act as a heterogeneous catalyst for the
reduction of allylic alcohols. Thus various terminal and in-
ternal allylic alcohols can be converted to the corresponding
4108
ꢁ 2008 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Chem. Eur. J. 2008, 14, 4104 – 4109

 Kim, Jung Won
Kim, Jung Won
 Koike, Takeshi
Koike, Takeshi
 Kotani, Miyuki
Kotani, Miyuki
 Yamaguchi, Kazuya
Yamaguchi, Kazuya
 Mizuno, Noritaka
Mizuno, Noritaka