
Catalysis Letters p. 987 - 995 (2017)
Update date:2022-08-10
Topics:
 Liu, Guoxiu
Liu, Guoxiu
 Liu, Shiwei
Liu, Shiwei
 Liu, Siyuan
Liu, Siyuan
 Yu, Shitao
Yu, Shitao
 Li, Lu
Li, Lu
 Liu, Fusheng
Liu, Fusheng
 Xie, Congxia
Xie, Congxia
 Song, Xiuyan
Song, Xiuyan
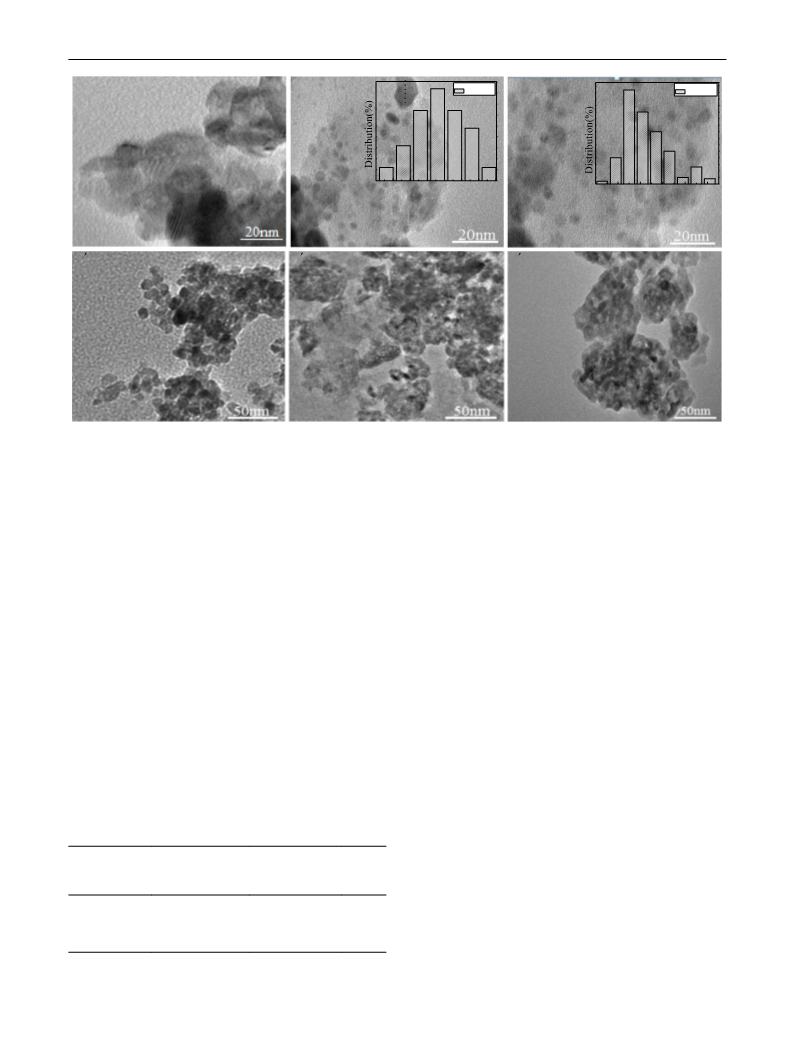
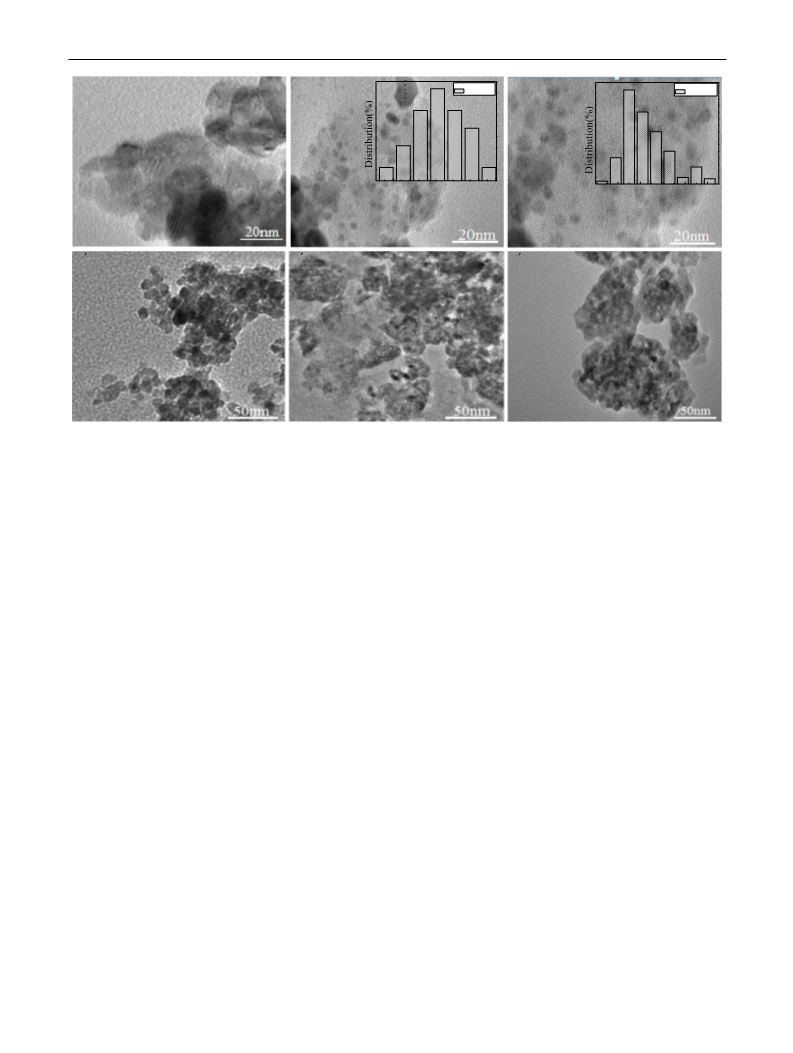
Abstract: The catalytic hydrogenation of 2-ethylhexenal was investigated over Pd supported on ZrO2, CeO2, Al2O3, MCM-41, MAS-7 and SBA-15. The activities and the selectivities of the catalysts were strongly affected by the nature of the support. Pd/ZrO2 had an excellent catalytic performance for the hydrogenation. The superior dispersion of Pd on the support ZrO2, and the stable structure of active components on ZrO2 as well as the synergistic effect of the bifunctional metal-support interaction enhanced the catalytic performance of Pd/ZrO2. The conversion of 2-ethylhexenal and the selectivity for 2-ethylhexanol were 100 and 99.1% respectively when the reaction was carried out at 240 °C for 7?h. The product was easily separated from the catalyst and the catalyst was of good reusability when it was repeated six times. In addition, the aggregation of Pd nanoparticles and the coking of ZrO2 the catalysts were the main cause for the catalyst deactivation. Graphical Abstract: [Figure not available: see fulltext.]
View More






Dalian Join King Biochemical Tech. Co., Ltd.
Contact:0411 39216206
Address:814 First State Blvd
Anhui Redstar Pharmaceutical Corp., Ltd
Contact:+86-563-5120837
Address:Jingxian Industrial Development Zone, Anhui , China
WEIFANG RICHEM INTERNATIONAL LTD
Contact:86-536-2222176
Address:weifang,shandong
website:http://www.china-sinoway.com
Contact:+86-592-5853819
Address:16/F,Huicheng Comm,Complex,No839 XiaHe Rd, Xiamen,China
Jiangsu Jiuri Chemical Co.,Ltd.
Contact:+86-519-82118868
Address:Tianwang Town, Jurong City, Jiangsu Province, China
Doi:10.1039/c6ra07555j
(2016)Doi:10.1007/s10562-013-1094-3
(2013)Doi:10.1016/j.jssc.2010.03.016
(2010)Doi:10.1039/c39880000823
(1988)Doi:10.1039/c7dt04340f
(2018)Doi:10.1016/S0039-128X(84)80036-7
(1984)