G Model
CATTOD-10546; No. of Pages11
ARTICLE IN PRESS
2
S.C. Patankar, G.D. Yadav / Catalysis Today xxx (2017) xxx–xxx
Nomenclature
kacid
kmetal
W
Intrinsic constant pertaining to acid sites
−1
−1
A
n-Butanal
(cm6 mol−1 min
g
cat
)
B
C
D
2-Ethylhexenal
2-Ethylhexenol
Hydrogen
Intrinsic const−a1nt pertaining to metal sites
−1
(cm6 mol−1 min gcat
)
)
Catalyst loading (g cm−3
E
F
G
H
2-Ethyl-1-hexanol
Butanoic acid
Butyl butanoate
Butanol
carbon chain length on the efficacy of the catalyst and to extend
its utility in another industrially important reaction.
I
n-Propanal
J
K
L
M
N
2-Methylpentenal
2-Methylpentenol
2-Methylpentanol
Propanoic acid
Propyl propanoate
Propanol
2. Experimental
2.1. Chemicals
Magnesium nitrate hexahydrate, aluminium nitrate nonahy-
drate, glycerol and copper(II) nitrate trihydrate were procured
from S.D. Fine Chemicals Pvt. Ltd, Mumbai, India. n-Butanal and n-
propanalwere procured from M/s. Thermo Fischer Scientific (India)
Pvt. Ltd, Mumbai. Pure samples of 2-ethyl-1-hexanol and 2-methyl-
1-pentanol were procured from Sigma Aldrich Chemicals Pvt. Ltd,
Mumbai.
O
Sb
Sm
Sa
CSm
CSb
CSa
K(A,I)m
Base site
Metal site
Acid site
Concentration of metal site (mol cm−3
)
Concentration of base site (mol cm−3
)
)
Concentration of acid site (mol cm−3
Adsorption constant of n-butanal and n-propanal on
metal site (cm3 mol−1
Adsorption constant of n-butanal and n-propanal on
base site (cm3 mol−1
Adsorption constant of n-butanal and n-propanal on
acid site (cm3 mol−1
Adsorption constant of 2-ethylhexenal and 2-
methylpentenal on metal site (cm3 mol−1
Adsorption constant of 2-ethylhexenal and 2-
methylpentenal on base site (cm3 mol−1
Adsorption constant of hydrogen on metal site
(cm3 mol−1
K(C,K)m Adsorption constant of 2-ethylhexenol and 2-
methylpentenol on metal site (cm3 mol−1
Adsorption constant of 2-ethylhexanol and 2-
methylpentanol on metal site (cm3 mol−1
Adsorption constant of butanoic acid and propanoic
acid on acid site (cm3 mol−1
Adsorption constant of butyl butanoate and propyl
propanoate on acid site (cm3 mol−1
K(H,O)m Adsorption constant of butanal and propanal on
metal site (cm3 mol−1
Adsorption constant of water on acid site
(cm3 mol−1
Rate of consumption of n-butanal (mol cm−3 min−1
Rate constant of aldol condensation reaction
(cm3 mol−1 min−1
Rate constant of hydrogenation reaction of alkenal
to alkenol (cm3 mol−1 min−1
Rate constant of hydrogenation reaction of alkenol
to aklanol (cm3 mol−1 min−1
Rate constant of acid hydrolysis reaction
2.2. Catalyst synthesis
)
K(A,I)b
K(A,I)a
K(B,J)m
K(B,J)b
KD
Saturated aqueous solution of magnesium nitrate hexahydrate
made such that the molar ratio of Mg:Al was 3. Copper (II) nitrate
trihydrate (1.17 mmol) was then added to the mixture such that
the loading of copper on hydrotalcite was 5% (w/w). Glycerol
(0.025 mol) was used as a fuel and mixed with the nitrate solu-
tion [13]. The slurry was heated at 80 ◦C for 30 min. The thick slurry
was later kept in preheated muffle furnace at 500 ◦C and allowed
to combust. The voluminous material formed post combustion was
used as a catalyst and designated as Cu/MMO.
)
)
)
)
)
)
2.3. Catalyst characterization
K(E,L)m
K(F,M)a
K(G,N)a
)
The synthesized catalyst was characterized per se and after use
to understand the structure activity relationship. Temperature pro-
grammed desorption (TPD) was used with CO2 and NH3 as probe
the catalyst (AutoChem II 2910, Micromeretics, USA). Twenty mg
catalyst sample was heated to 500 ◦C in quartz tube under flow
of helium to remove traces of moisture, air and any other organic
compounds in case of used catalyst [21–23]. 10% (w/w) Ammonia in
helium was then adsorbed on the catalyst surface. The physisorbed
gas was desorbed with flow of helium. The chemisorbed gas was
desorbed using a temperature program and measured using a ther-
for CO2-TPD experiments with use of 10% (w/w) carbon dioxide
in helium. Temperature programmed reduction (TPR) was studied
to understand the strength of metal sites. Catalyst sample (20 mg)
was heated to 500 ◦C under 5% O2 in helium so that the catalyst was
completely oxidised [24]. The traces of O2 were removed by flow of
helium and the catalyst sample cooled to room temperature. 10%
(w/w) hydrogen in argon was then passed over the catalyst by heat-
ing the catalyst to 500 ◦C. The hydrogen uptake was measured using
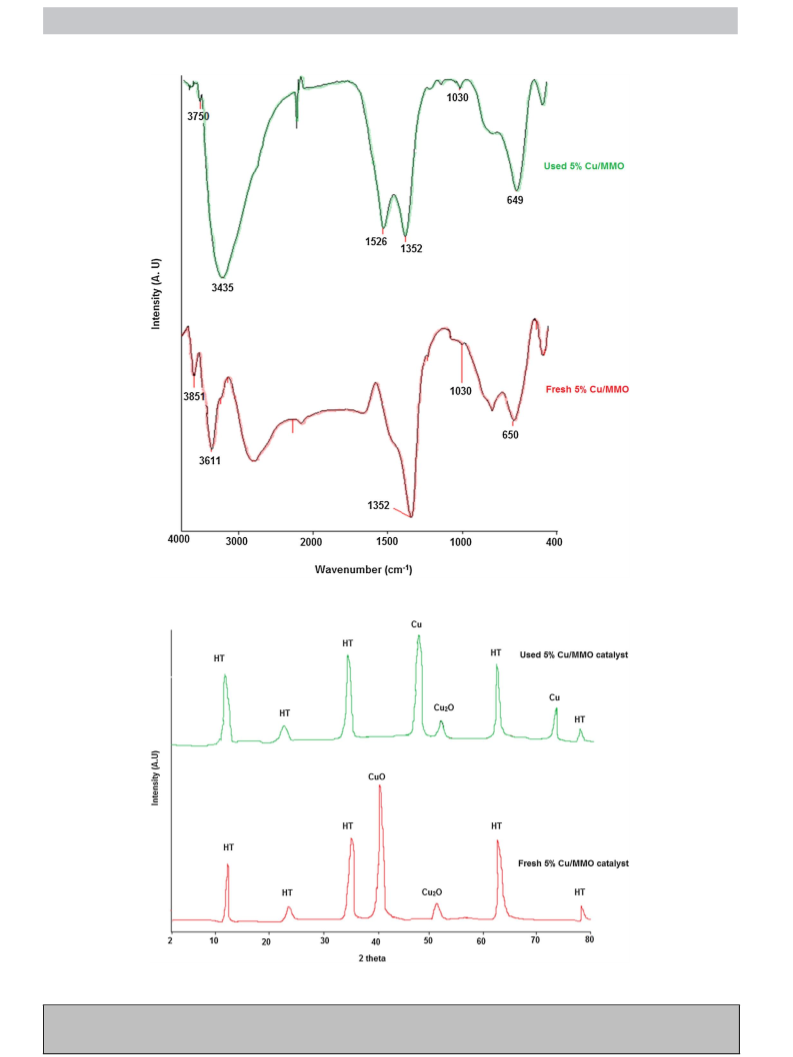
a TCD. FTIR spectra was obtained from the catalyst sample pressed
in KBr pellet to understand the bending and stretching vibrations
(Perkin Elmer, 1000-PC). The analysis was done in the fingerprint
region from wavenumber of 400 –4000 cm−1 at a resolution of
2 cm−1. Powder X-ray Diffraction (XRD) was used to understand
the crystallinity of the catalyst (Bruker D8 Advance, USA). Analysis
was done using Cu k␣ radiations with beam current of 40 kV and
)
)
)
KX
)
rA
k1,7
)
)
k2,8
)
k3,9
)
k4,10
k5,11
k6,12
kbase
(cm3 mol−1 min−1
Rate constant
(cm3 mol−1 min−1
)
of
esterification
reaction
)
Rate constant of hydrogenation reaction of aldehyde
(cm3 mol−1 min−1
Intrinsic constant pertaining to base sites
)
−1
−1
)
(cm6 mol−1 min
g
cat
Please cite this article in press as: S.C. Patankar, G.D. Yadav, Cascade engineered synthesis of 2-ethyl-1-hexanol from n-butanal and
2-methyl-1-pentanol from n-propanal using combustion synthesized Cu/Mg/Al mixed metal oxide trifunctional catalyst, Catal. Today

 Patankar, Saurabh C.
Patankar, Saurabh C.
 Yadav, Ganapati D.
Yadav, Ganapati D.