T. Baran and M. Nasrollahzadeh
Inorganic Chemistry Communications 119 (2020) 108117
high catalytic activity and wide functional group tolerance in catalytic
reactions [11–15]. However, major disadvantages in the synthesis of Pd
NPs are the aggregation and reusability problems. These disadvantages
can be overcome by designing novel supports, which strongly interact
with metals.
GC–MS was used for the characterization of benzonitriles using an
Agilent GC-7890 A-MS 5975 instrument.
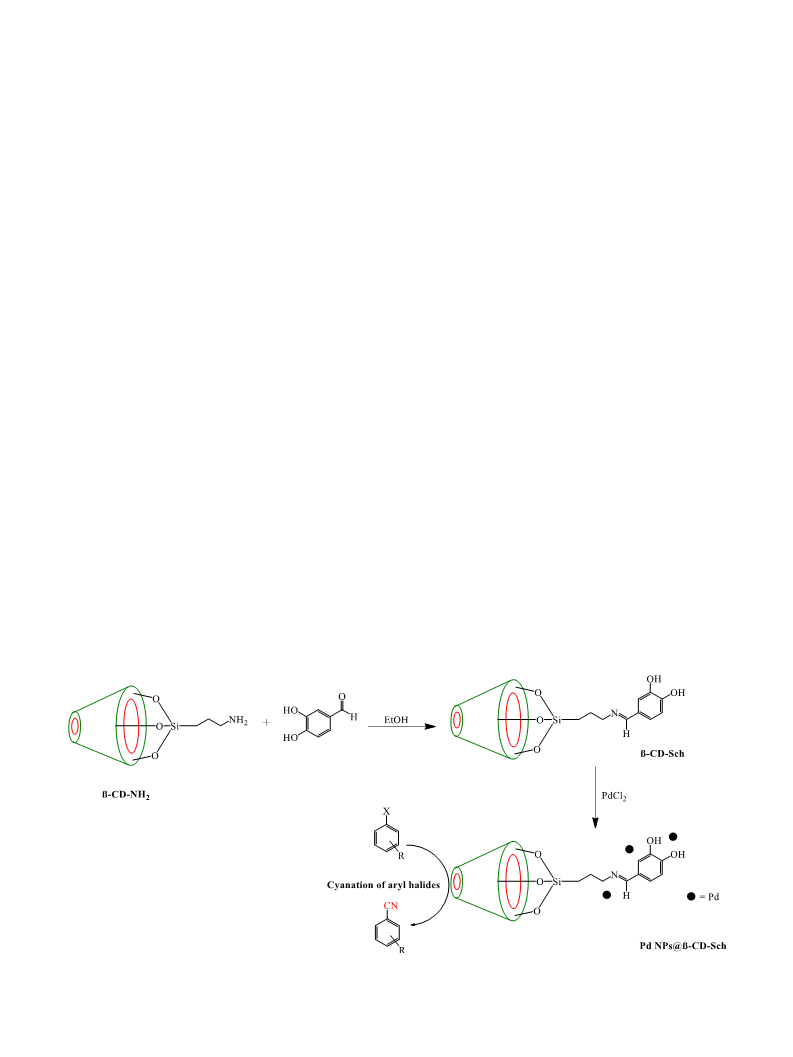
2.2. Synthesis of β-CD-Sch as stabilizer
In recent years, natural bio-polymers have gained considerable at-
tention due to their biodegradability, biocompatibility, and high affi-
nity for different transition metals. Among biopolymers, β-cyclodextrin
Firstly, amine attached β-CD was easily fabricated according to a
previously reported method [19,20] and entitled as β-CD-NH . Briefly,
2
2 g of β-CD and 5 mL of APTES were stirred at 100 °C in toluene solvent
(
β-CD), which is a cyclic oligosaccharide, is a natural, low cost, suitably
for 24 h. β-CD-NH (1 g) and 3,4-dihydroxybenzaldehyde (1 g) were
2
sized, biocompatible, non-toxic and edible compound [16,17]. Ad-
ditionally, β-CD has primary or secondary hydroxyl groups, pendant on
the polymer chain, by which it is easily modified. Thanks to its out-
standing properties and chemical structure, β-CD is an ideal stabilizer
for the construction of Pd NPs [18]. Therefore, different β-CD modified
supports can be prepared for the fabrication of different nanoscale
palladium and their catalytic performances can then be tested in var-
ious transformations.
then transferred to 40 mL of ethanol solvent and the mixture was re-
fluxed for 48 h. Finally, the support material, denoted as β-CD-Sch, was
filtered, washed with ethanol and dried (Scheme 1).
2.3. Synthesis of Pd NPs@β-CD-Sch catalyst
0.5 g of β-CD-Sch and 0.1 g of PdCl were added to 25 mL of ethanol
2
and the mixture obtained was stirred for 5 h at 70 °C in the absence of
In continuation of our interest in the development of heterogeneous
nanocatalysts on natural biopolymers, in this study, Pd NPs have been
prepared on a β-CD containing Schiff base and their chemical structure
has been verified by different analytical techniques. Afterwards, Pd
NPs@β-CD-Sch were evaluated as catalyst in the fabrication of benzo-
any reducing agent Afterwards, the reaction color changed to black.
Finally, Pd NPs@β-CD-Sch was filtered, rinsed with water and dried for
use in catalytic applications (Scheme 1).
2.4. Typical synthesis method for cyanation of aryl halides
nitriles via cyanation of aryl halides using K
4
[Fe(CN) ]. Pd NPs@β-CD-
6
Sch successfully catalyzed the conversion of various aryl halides to the
desired benzonitriles in good yields within 4 h. Moreover, Pd NPs@β-
CD-Sch serve as recyclable catalysts, which could be used eight times.
Pd NPs@β-CD-Sch catalyst (0.05 mol. %), aryl halide (1 mmol),
K
4
[Fe(CN)
6
] (0.2 mmol) and Na
2
CO (1.5 mmol) were added to 6 mL of
3
DMF and the mixture obtained was stirred at 120 °C for 4 h. The
mixture was then cooled to room temperature and the product was
2
. Experimental
extracted with Et
2
O (10 mL) and H O (5 mL) three times and dried over
2
MgSO . Afterwards, the organic solvents were evaporated to obtain
4
2.1. General remarks
benzonitriles.
β-Cyclodextrin, 3-aminopropyltriethoxysilane (APTES), 3,4-dihy-
3. Results and discussion
droxybenzaldehyde, aryl halides (Ar-X), phenylboronic acid (PhB
(
OH)
2
), NaOH, KOH, Na
2
CO
3
, K
2
CO3, PdCl2, DMF, DMSO, toluene, NMP
3.1. Pd NPs@β-CD-Sch characterization
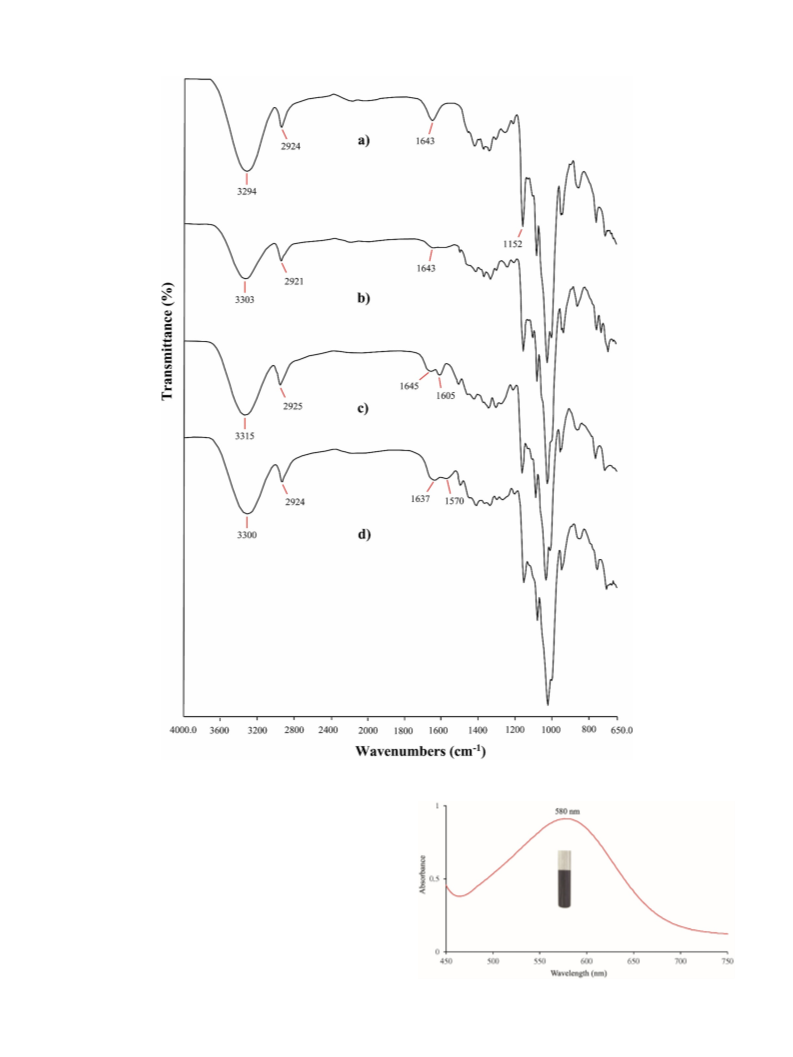
FT-IR analysis was used to confirm the chemical structures of β-CD,
and diethyl ether were obtained from Sigma Aldrich Chemical Co.
Modification of the products was confirmed by spectrophotometry
(
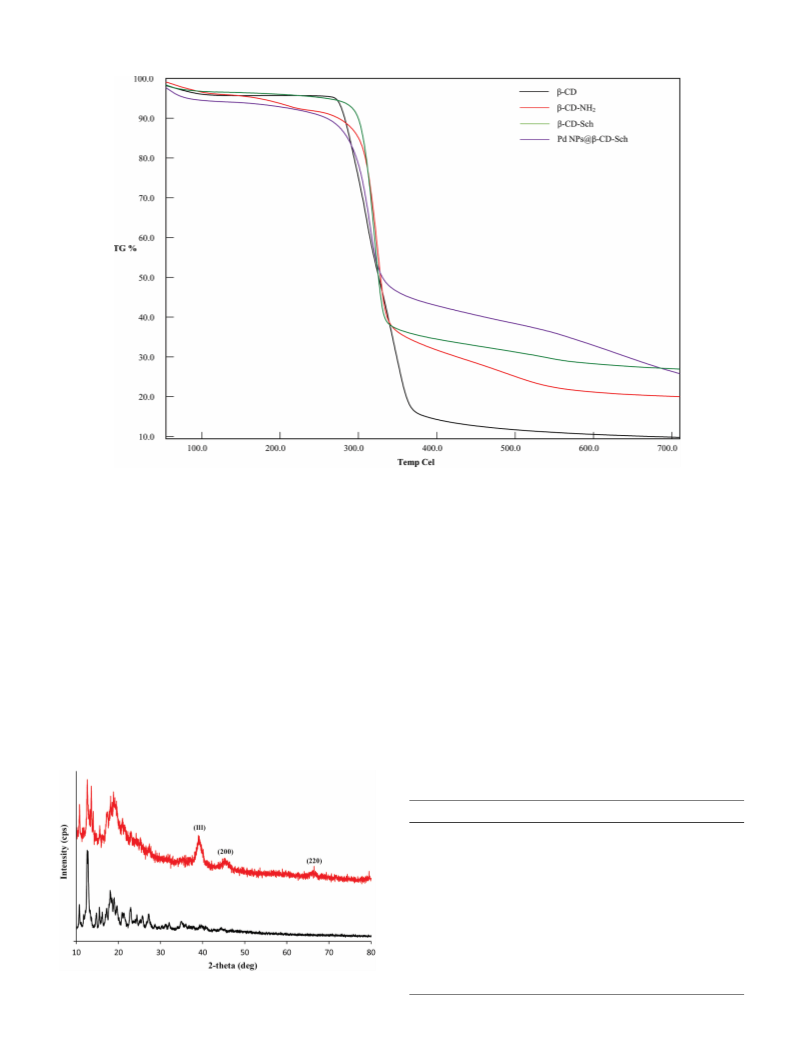
Perkin Elmer 100 FT-IR spectrophotometer). The X-ray spectra of β-CD
β-CD-NH , β-CD-Sch and Pd NPs@β-CD-Sch (Fig. 1). Important peaks
2
−1
and Pd NPs@β-CD-Sch were obtained using a Rigaku smart lab system.
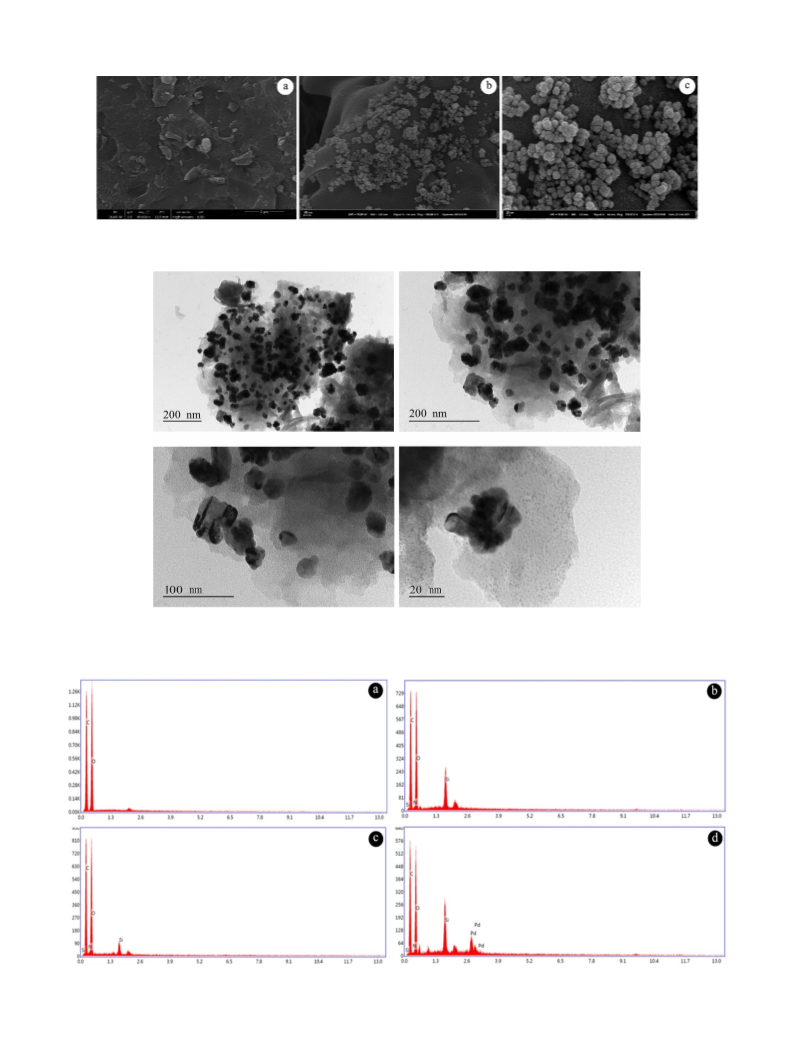
The surface profiles and EDS analyses of β-CD, β-CD-NH, β-CD-Sch and
NPs@β-CD-Sch were investigated using FEI Quanta 450 FEG. The
thermal behavior of the catalyst was studied by EXSTAR S11 7300.
are observed in the FT-IR spectrum of β-CD at 3294 cm (vibration of
symmetrical and asymmetrical stretching of the –OH groups),
−1
−1
2924 cm (vibration of C–H stretch), 1643 cm (HeOeH bending),
1152 cm−1 and 1020 cm
−1
(asymmetric and symmetric CeOeC
Scheme 1. Preparation of Pd NPs@β-CD-Sch.
2

 Baran, Talat
Baran, Talat
 Nasrollahzadeh, Mahmoud
Nasrollahzadeh, Mahmoud