Table 5 Comparison of TON and TOF
(b) T. V. Lee, in Comprehensive Organic Synthesis, ed. B. M. Trost and
I. Fleming, Pergamon, Oxford, 1991, vol. 7, ch. 2.8; (c) G. Procter,
in Comprehensive Organic Synthesis, ed. B. M. Trost and I. Fleming,
Pergamon, Oxford, 1991, vol. 7, ch. 2.9.
2 Recently, highly selective oxidations using transition-metal V2, Pt3, Rh4,
Pd5, Ru6, Co7, Ni8, Cu9, and Os10 complexes with molecular oxygen were
reported; (a) T. Hirao, Chem. Rev., 1997, 97, 2707; (b) M. Kirihara, Y.
Ochiai, S. Takizawa, H. Takahata and H. Nemoto, Chem. Commun.,
1999, 1387; (c) Y. Maeda, N. Kakiuchi, S. Matsumura, T. Nishimura
and S. Uemura, Tetrahedron Lett., 2001, 42, 8877; (d) Y. Maeda, N.
Kakiuchi, S. Matsumura, T. Nishimura, T. Kawamura and S. Uemura,
J. Org. Chem., 2002, 67, 6718; (e) S. Velusamy and T. Punniyamurthy,
Org. Lett., 2004, 6, 217; (f) S. R. Reddy, S. Das and T. Punniyamurthy,
Tetrahedron Lett., 2004, 45, 3561; (g) A. T. Radosevich, C. Musich and
F. D. Toste, J. Am. Chem. Soc., 2005, 127, 1090.
Entry
System
TON
TOF/h-1
1
2
0.024 mol% 2
0.1 mol% 1
2500
860
52
18
3 C.-G. Jia, F.-Y. Jing, W.-D. Hu, M.-Y. Huang and Y.-Y. Jiang, J. Mol.
Catal., 1994, 91, 139.
4 J. Martin, C. Martin, M. Faraj and J.-M. Bregeault, Nouv. J. Chim.,
1984, 8, 141.
cf . VO(acac)2: TON 45, TOF 15 h-1; V2O5: TON 19, TOF 2 h-1.
5 (a) T. F. Blackburn and J. Schwartz, J. Chem. Soc., Chem. Commun.,
1977, 157; (b) K. Kaneda, M. Fujii and K. Morioka, J. Org. Chem.,
1996, 61, 4502; (c) T. Nishimura, T. Onoue, K. Ohe and S. Uemura,
Tetrahedron Lett., 1998, 39, 6011; (d) T. Nishimura, T. Onoue, K. Ohe
and S. Uemura, J. Org. Chem., 1999, 64, 6750; (e) K. P. Peterson
and R. C. Larock, J. Org. Chem., 1998, 63, 3185; (f) G.-J. ten Brink,
I. W. C. E. Arends and R. A. Sheldon, Science, 2000, 287, 1636; (g) Y.
Uozumi and R. Nakao, Angew. Chem., Int. Ed., 2003, 42, 194; (h) M. J.
Schultz, C. C. Park and M. S. Sigman, Chem. Commun., 2002, 3034;
(i) T. Nishimura, Y. Maeda, N. Kakiuchi and S. Uemura, J. Chem. Soc.,
Perkin Trans. 1, 2000, 4301; (j) T. Nishimura, N. Kakiuchi, M. Inoue
and S. Uemura, Chem. Commun., 2000, 1245; (k) D. R. Jensen, M. J.
Schultz, J. A. Mueller and M. S. Sigman, Angew. Chem., Int. Ed., 2003,
42, 3810; (l) T. Iwasawa, M. Tokunaga, Y. Obora and Y. Tsuji, J. Am.
Chem. Soc., 2004, 126, 6554; (m) M. Yang, K.-T. Yip, J.-H. Pan, Y.-C.
Chen, N.-Y. Zhu and D. Yang, Synlett, 2006, 3057; (n) S. Shimazu, T.
Uehara, A. Asami, T. Hara and N. Ichikuni, J. Mol. Catal. A: Chem.,
2008, 282, 28.
6 (a) M. Matsumoto and N. Watanabe, J. Org. Chem., 1984, 49, 3435;
(b) I. E. Marko´, P. R. Giles, M. Tsukazaki, I. Chelle´-Regnaut, C. J. Urch
and S. M. Brown, J. Am. Chem. Soc., 1997, 119, 12661; (c) K. Kaneda,
T. Yamashita, T. Matsushita and K. Ebitani, J. Org. Chem., 1998, 63,
1750; (d) B. Hinzen, R. Lenz and S. V. Ley, Synthesis, 1998, 977; (e) T.
Matsushita, K. Ebitani and K. Kaneda, Chem. Commun., 1999, 265;
(f) M. Pagliaro and R. Ciriminna, Tetrahedron Lett., 2001, 42, 4511;
(g) K. Masutani, T. Uchida, R. Irie and T. Katsuki, Tetrahedron Lett.,
2000, 41, 5119; (h) M. Lee and S. Chang, Tetrahedron Lett., 2000,
41, 7507; (i) K. Yamaguchi, K. Mori, T. Mizugaki, K. Ebitani and
K. Kaneda, J. Am. Chem. Soc., 2000, 122, 7144; (j) A. Miyata, M.
Murakami, R. Irie and T. Katsuki, Tetrahedron Lett., 2001, 42, 7067;
(k) A. Hanyu, E. Takezawa, S. Sakaguchi and Y. Ishii, Tetrahedron Lett.,
1998, 39, 5557; (l) H.-Y. Shen, S.-C. Zhou, M.-H. Wei and H.-X. Zong,
React. Funct. Polym., 2006, 66, 827; (m) C. N. Kato, A. Shinohara, N.
Moriya and K. Nomiya, Catal. Commun., 2006, 7, 413; (n) C. N. Kato,
M. Ono, T. Hino, T. Ohmura and W. Mori, Catal. Commun., 2006, 7,
673.
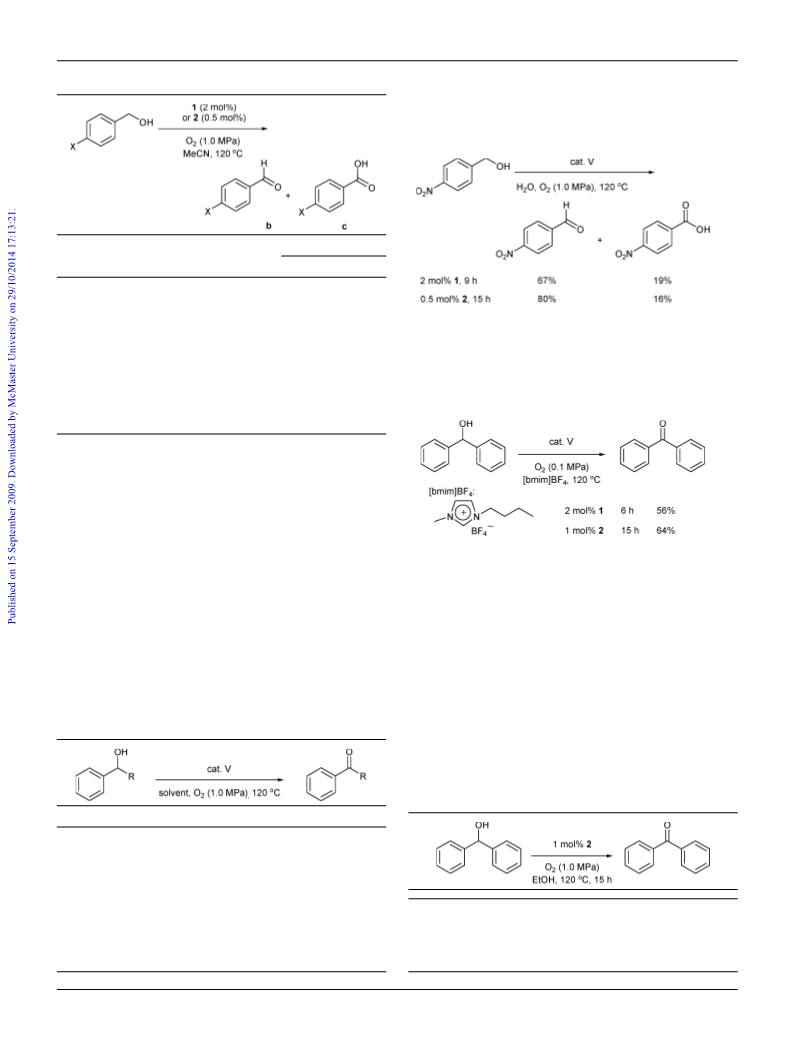
alcohols in comparison with other vanadium-catalyzed oxidation
systems (cf . VO(acac)2: TON 45, TOF 15 h-1; V2O5: TON 19, TOF
2 h-1).
In conclusion, we have found several unique characteristics of
vanadium complexes 1 and 2 as oxidation catalysts of benzyl
alcohols with molecular oxygen. For example, they can be
employed as catalysts for the oxidation of benzyl alcohols in
non-toxic solvents such as ethanol, water, and ionic liquids. In
particular, tetranuclear vanadium complex 2 is useful as a recycling
catalyst with excellent TON and TOF. These results suggest that it
is possible to control the oxidation potential of vanadium and
affinity of the vanadium catalysts to solvents by the selection
of appropriate ligands in vanadium complexes. In addition, we
believe that the preparation of the multi-nuclear catalyst such
as 2 enables us to develop economically and environmentally
friendly oxidation systems with a recycling catalyst having superior
durability. We are now challenging the development of further
useful oxidation of alcohols and amines by designing the catalysts
based on the results described in this paper.
This research was supported by a Grant-in-Aid for Scientific
Research on Priority Areas (Area 444, No. 19020061) from the
Ministry of Education, Culture, Sports, Science and Technology,
Japan, and Kansai Research Foundation for Technology Promo-
tion (KRF). S. K. acknowledges Research Fellowships of the
Japan Society for the Promotion of Science (JSPS) for Young
Scientists. We also acknowledge Ms Michiko Tabe and Ms Mihoko
Kato (Nara Women’s University) for their cooperation at the
initial stage of this study.
7 (a) T. Iwahama, S. Sakaguchi, Y. Nishiyama and Y. Ishii, Tetrahedron
Lett., 1995, 36, 6923; (b) V. B. Sharma, S. L. Jain and B. Sain,
Tetrahedron Lett., 2003, 44, 383; (c) T. Yamada and T. Mukaiyama,
Chem. Lett., 1989, 519; (d) V. B. Sharma, S. L. Jain and B. Sain, J. Mol.
Catal. A: Chem., 2004, 212, 55; (e) G. Blay, L. Cardona, I. Ferna´ndez
and J. R. Pedro, Synthesis, 2007, 3329.
Notes and references
† General procedure for the vanadium-catalyzed oxidation of benzyl
alcohols in acetonitrile or ethanol is as follows: In a 50 mL stainless steel
autoclave with a magnetic stirring bar were added VO(Hhpic)2 (2 mol%) or
(VO)4(hpic)4 (0.5 mol%), solvent (6 mL) and benzyl alcohols (1.5 mmol).
Oxygen was purged twice and then charged at 1.0 MPa. The reaction was
conducted with magnetic stirring for the appropriate time upon heating at
120 ◦C. The resulting mixture was filtered through a Celite pad and the
filtrate was analyzed by GC. Purification of the product was carried out
on a short pad of silica gel using ethyl acetate and hexane as eluent to
afford the analytically pure aldehydes and carboxylic acids, respectively.
All products were commercially available and identified by comparison of
the isolated products with authentic samples.
8 B. M. Choudary, M. L. Kantam, A. Rahman, C. V. Reddy and K. K.
Rao, Angew. Chem., Int. Ed., 2001, 40, 763.
9 (a) E. Saint-Aman, S. Me´nage, J.-L. Pierre, E. Defrancq and G. Gellon,
New J. Chem., 1998, 22, 393; (b) P. Chaudhuri, M. Hess, J. Mu¨ller,
K. Hildenbrand, E. Bill, T. Weyhermu¨ller and K. Wieghardt, J. Am.
Chem. Soc., 1999, 121, 9599; (c) I. E. Marko´, P. R. Giles, M. Tsukazaki,
S. M. Brown and C. J. Urch, Science, 1996, 274, 2044; (d) R. C. Pratt
and T. D. P. Stack, J. Am. Chem. Soc., 2003, 125, 8716; (e) P. Gamez,
I. W. C. E. Arends, J. Reedijk and R. A. Sheldon, Chem. Commun.,
2003, 2414; (f) I. E. Marko´, A. Gautier, R. Dumeunier, K. Doda,
F. Philippart, S. M. Brown and C. J. Urch, Angew. Chem., Int. Ed.,
2004, 43, 1588; (g) S. Velusamy, A. Srinivasan and T. Punniyamurthy,
Tetrahedron Lett., 2006, 47, 923; (h) N. Lu and Y.-C. Lin, Tetrahedron
Lett., 2007, 48, 8823.
1 (a) S. V. Ley and A. Medin, in Comprehensive Organic Synthesis, ed.
B. M. Trost and I. Fleming, Pergamon, Oxford, 1991, vol. 7, ch. 2.7;
9710 | Dalton Trans., 2009, 9708–9711
This journal is
The Royal Society of Chemistry 2009
©

 Kodama, Shintaro
Kodama, Shintaro
 Ueta, Yukihiro
Ueta, Yukihiro
 Yoshida, Jun
Yoshida, Jun
 Nomoto, Akihiro
Nomoto, Akihiro
 Yano, Shigenobu
Yano, Shigenobu
 Ueshima, Michio
Ueshima, Michio
 Ogawa, Akiya
Ogawa, Akiya