CrystEngComm
Paper
8
,9
extensively investigated, because this might determine the
ease of resolution. Polymorphism has also been noted for
both the racemate and enantiomer of 3-chloromandelic
3-trifluoromethyl-, 4-bromo- and 4-chloromandelic acid were
prepared according to this approach.
1
0
acid. In attempting to repeat the work of Larsen and Marti
we observed several polymorphic forms and succeeded in
obtaining the crystal structure of a polymorph of their race-
mic 4-fluoromandelic acid. We have also obtained the crystal
structures of the methyl- and trifluoromethyl-mandelic acids
which have not been reported previously and include in our
comparisons the few structures of substituted mandelic
acids, and polymorphs 1 (ref. 11) and 2 (ref. 12) of mandelic
General description of the cyanohydrin route, as used
for 3-bromo
Sodium cyanide (1.5 g), and 3-bromobenzaldehyde (4.6 g =
1
/40 mol) and benzyltrimethylammonium chloride (10 mg)
were stirred and cooled in ice-water. A homogeneous mixture
of concentrated HCl (5 ml) and ether (4 ml) was added
dropwise over 1 h so as to maintain the temperature always
below 10 °C. The mixture darkened to deep orange during
the addition, but eventually became pale yellow. 20 ml con-
centrated HCl was added and stood overnight, then heated to
70 °C for 1 h. The mixture was extracted 2 × 10 ml ether. The
ether layer was basified and extracted 2 × 10 ml ether. The
aqueous layer was acidified and extracted 3 × 10 ml ether.
The ether extracts were dried with sodium sulfate, filtered
and evaporated to dryness to give 12.3 g of product, mostly
inorganic salts. This was recrystallized from dichloromethane
to give 2.0 g 3-bromomandelic acid (35% yield). 2-, 3- and
6
,8
acid itself, that are already in the literature.
For the sake of clarity, the present paper is confined to those
structures derived from 8 and 10 membered ring dimers i.e.
2
2
2 2
with graph set descriptors of R (8) and R (10) respectively.
These constitute the majority of structures so far encountered
and so form a relatively homogenous group linked by numerous
0
, 1, 2 and 3-dimensional relationships revealed by the XPac
4
program for structural similarity. It is also confined to the
polymorphs so far encountered during the attempts to obtain
suitable crystals for single crystal diffraction experiments.
4
4
3
-fluoro, 2-, 3- and 4-chloro, 2- and 4-bromo, 2-,3- and
-methyl, 2- and 3-trifluoromethyl, 2-, 3- and 4-methoxy and
-iodomandelic acid were all prepared in a similar fashion.
Commercial samples of 2-chloro-, 3-chloro-, 4-methoxy-
Experimental
The mandelic acids were synthesized from benzaldehydes
1
3
either via the cyanohydrin route or the Merz two-phase
and 4-trifluoromethylmandelic acid were also used, while
2-iodo and 4-iodo-, 3-chloro, 3-bromo- and 3-methylmandelic
acid were a gift from Jan von Langermann of the Max Planck
Institute, Magdeburg.
1
4
dichlorocarbene route. We were unable to obtain anything
other than benzoic acids by Compere's one-phase dichloro-
1
5
16,17
carbene route, although others appear to have done so.
The yields were incredibly variable, for example 6% for
-bromomandelic acid and 60% for 3-iodomandelic acid, but
The mandelic acids do not crystallise so well as, for example,
1
,2
20
2
substituted sulphonamides
or acetanilides. Hydroxylic
sufficient material was obtained in all cases for the present
purpose of obtaining a crystal structure. Larger quantities
will be needed for the chiral separations, so several sets of
solvents, especially ethanol, generally so useful for growing large
crystals, are particularly poor in the case of all the mandelic
acids tried in providing material for single crystal studies.
Ether, dichloromethane, chloroform, acetonitrile, toluene and
nitromethane were amongst the most successful solvents.
Single crystal experiments have been routinely performed
according to previously published procedures with details of
data collection and refinement parameters for all structures
summarised in the ESI.† It is not the aim of this paper to discuss
the individual structures and therefore experimental details in
the form of a summary table of data collection and refinement
parameters is provided for reference (Table S1†). All structures
compared in this study are presented in Table S1,† however
only pertinent crystal data (such as unit cell dimensions and
space group) are given for those previously reported. CIF files are
available as ESI† and have also been deposited with the
Cambridge Crystallographic Data Centre (CCDC 1007650 –
CCDC 1007667 inclusive and CCDC 1008570).
1
8
Design of Experiment runs were undertaken on 4-chloro-,
-bromo- and 4-methyl-mandelic acid preparations to try to
4
optimize the yields. The yields were erratic and the results
inconclusive for both routes: lithium ions, the key to Com-
pere's synthesis, appeared ineffective in improving the yields
2
1
1
9
by the Merz route. A modification of Jenkins' procedure for
the cyanohydrin route without sodium bisulfate but with
phase transfer catalyst was generally the most successful.
General description of the dichlorocarbene route, as used
for 2-methyl
A mixture of chloroform (8 ml), 2-methylbenzaldehyde
(
(
5.8 ml = 1/20 mol) and benzyltriethylammonium chloride
1.14 g) was stirred vigorously with a magnetic stirrer and
heated to 60°. Sodium hydroxide (10 g in 10 ml water) was
added dropwise over 2.5 h. Maintenance of the temperature
below 60 °C is critical. Distilled water (30 ml) was added to the
orange–brown solution to dissolve any solids, and the solution
was extracted with 2 × 30 ml ether. The aqueous layer was acid-
ified with concentrated HCl, and extracted 2 × 30 ml ether. The
orange extract was dried over sodium sulfate and the resulting
oil crystallised from toluene. Yield 5.15 g = 62%. 3-Methyl-,
Discussion
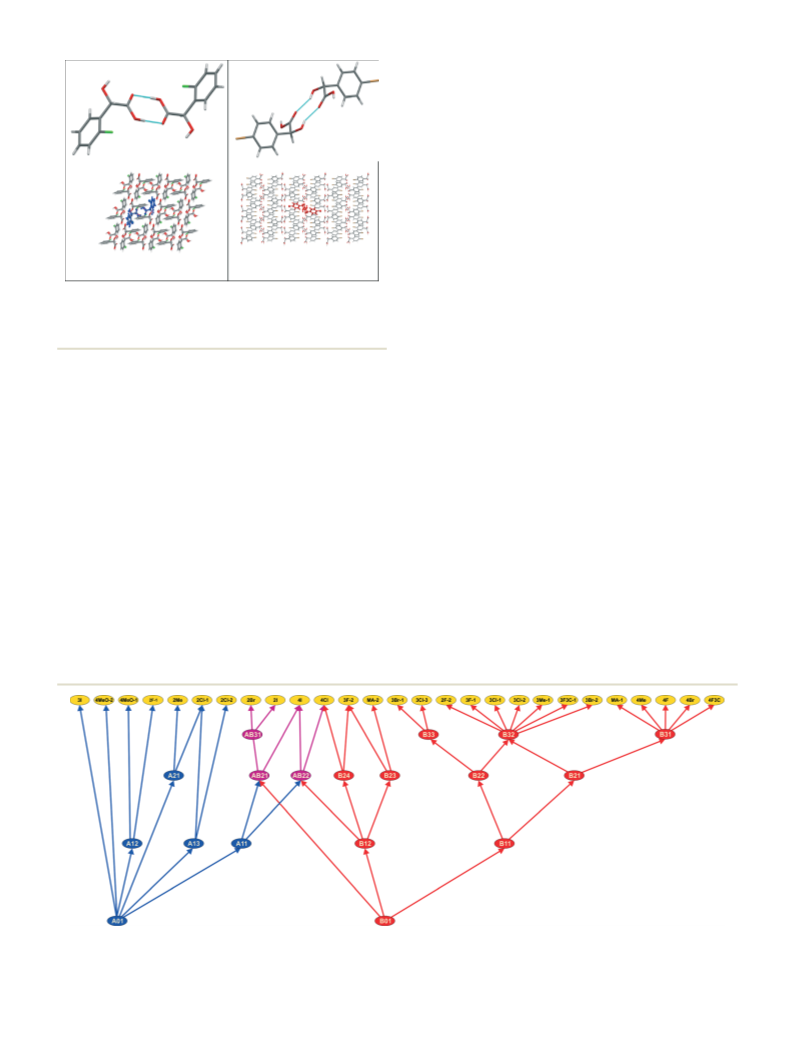
The monosubstituted racemic mandelic acids with fluoro,
chloro, bromo, iodo, trifluoromethyl, methyl and methoxyl
substituents at the ortho, meta and para positions, as shown
in Fig. 1, are presented (note we have not managed to
This journal is © The Royal Society of Chemistry 2014
CrystEngComm, 2014, 16, 10816–10823 | 10817

 Coles
Coles
 Ellis
Ellis
 Leung
Leung
 Sarson
Sarson
 Threlfall
Threlfall
 Tizzard
Tizzard