Biosynthetic pathway of neuromelanin derived from the locus coeruleus
771
rate of 7.0 mL/min. Determination of melanin markers was
performed using HPLC as previously reported (Wakamatsu et al.
2002, 2003; Ito et al. 2011).
shows that 5-S-Cys-CMT-DA consists of a mixture of diastereomers
evidenced by a double series of signals. Mass spectrum: m/z 363.08
(M+H)+ for C13H18N2O6S2.
HCl hydrolysis of human NM
Ion trap liquid chromatography/mass spectrometry of CMT-DA
Ion Trap Liquid Chromatography/Mass Spectrometry was per-
formed using a liquid chromatography/mass spectrometry system by
Esquire HCT Plus Mass Spectrometer (Bruker) connected to an
HPLC system 1260 Infinity (Agilent Technologies) with a Cadenza
CD-C18 column (2.0 975 mm) (Imtakt Corp., Kyoto, Japan) with
10% CH3OH – 0.1% HCOOH, at flow rate of 0.1 mL/min.
One hundred lL of a suspension (1 mg/mL) of NM were transferred
to a sealed-capped tube with 300 lL 6 M HCl containing 5%
thioglycolic acid and 1% phenol. The tube was purged with a stream
of argon, then sealed and heated at 110°C for 16 h. After cooling, the
hydrolysate was mixed with 10 lL of an internal standard solution
(methyl-5-S-Cys-DOPA, 15 lmol/L), extracted twice with 1 mL
diethyl ether to remove phenol, and treated with alumina to extract
catecholic compounds as follows.
A 120 lL aliquot of the
Statistical analyses
hydrolysate was transferred into an Eppendorf tube containing
50 mg acid-washed alumina and 200 lL 1% Na2S2O5-1%
EDTA.2Na. To the mixture, 500 lL 2.7 M Tris.HCl-2% EDTA.2Na
(pH 9.0) was added and immediately mixed vigorously for 5 min on a
microtube mixer. After centrifugation, the aqueous layer was
removed by aspiration, and the remaining alumina was washed with
1 mL water three times. The catecholic compounds were then eluted
with 100 lL 0.4 M HClO4 by shaking for 2 min. A 30 lL aliquot of
the HClO4 extract was injected into the HPLC system.
We did not perform any statistical analyses, but values obtained by
H2O2-mediated oxidation and HI hydrolysis of synthetic pheome-
lanins prepared from the Cys-derivatives of DA, NE, DOPAC,
DOMA, DOPEG, DOPE, and DOPA, were obtained as a mean of
two determinations.
Results
Alkaline H2O2-mediated oxidation and reductive HI
hydrolysis
Synthesis of CMT-DA
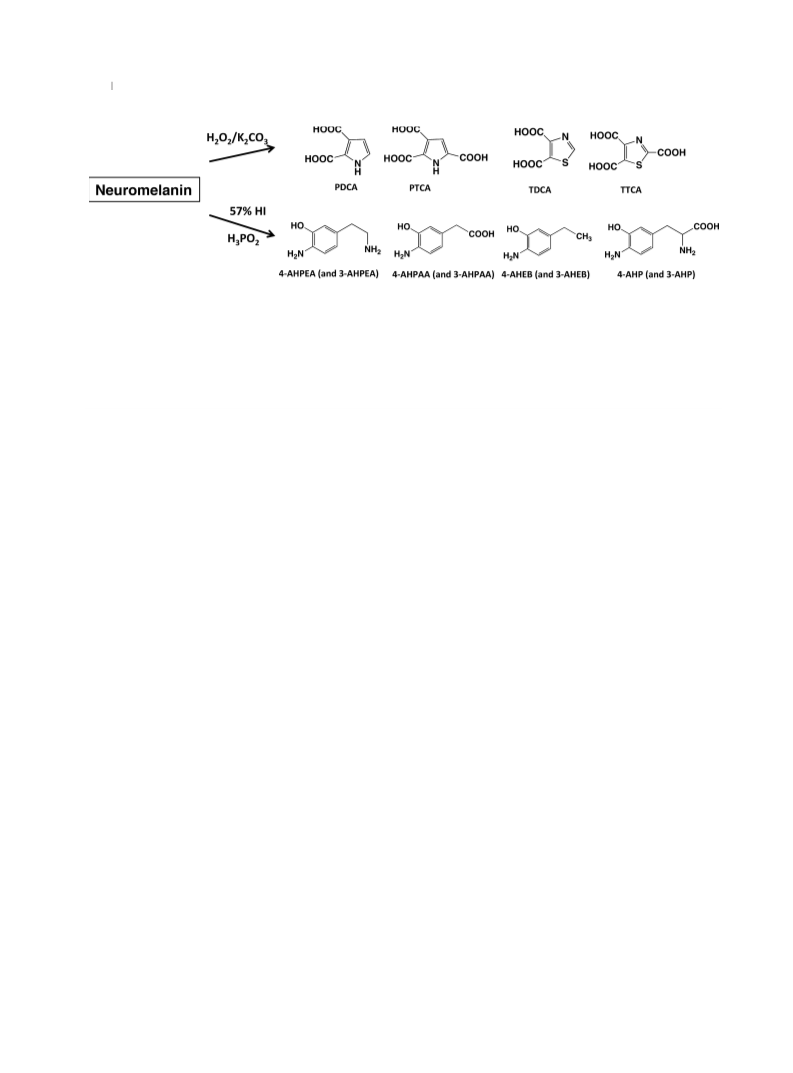
PDCA, PTCA, TDCA, and TTCA (Fig. 2) were measured
using H2O2-mediated oxidation of synthetic pheomelanins
prepared from Cys-DA, Cys-NE, Cys-DOPAC, Cys-DOPE,
Cys-DOPA, Cys-DOMA, and Cys-DOPEG (Table 1).
Pheomelanins prepared from Cys-DOPAC, Cys-DOPE
Cys-DOMA, and Cys-DOPEG did not give detectable levels
of PTCA or PDCA, but produced only TDCA and TTCA,
two specific markers of pheomelanin (Table 1). This is
consistent with the fact that those Cys-catechols do not
possess an amino group in their side chains.
On HI hydrolysis, DA-related pheomelanins, i.e., Cys-DA-
melanin, Cys-DOPAC-melanin and Cys-DOPE-melanin,
gave high levels of pheomelanin markers, i.e., 4-AHPEA,
4-AHPAA, and AHEB (as a mixture of 4-AHEB and
3-AHEB), respectively (Table 1 and Fig. 2). On the other
hand, NE-related pheomelanins, i.e., Cys-NE-melanin,
Cys-DOMA-melanin, and Cys-DOPEG-melanin, gave lower
(a half to one-fifth of the corresponding DA-related pheome-
lanins) levels of those markers. Cys-DOPA-melanin gave a
high level of 4-AHP.
A solution of 144 mg (0.7 mmol) NEÁHCl in 20 mL 6 M HCl
containing 5% thioglycolic acid and 1% phenol was refluxed for
30 min at 110°C. Thioglycolic acid and phenol were essential to
avoid polymerization of the starting material NE. The reaction
mixture was washed twice with 20 mL ether to remove phenol. The
water layer was evaporated to dryness and then subjected to Dowex
50W-X2 chromatography (1.6 9 10 cm in 2 M HCl). The column
was eluted with 2 M HCl and fractions of 10 mL were collected.
Fractions 9–16 were evaporated to give 159 mg (80% yield) of
CMT-DAÁHClÁH2O salt as a colorless solid (HPLC purity: > 99%).
UV kmax 282 nm (e 3,260) in 0.1 M HCl. 1H-NMR (DCl): d = 3.32
(dd, 2H, J = 16 Hz, 16 Hz), 3.47 (m, 2H), 4.19 (t, 1H, J = 8.0 Hz),
6.85 (dd, 1H, J = 2.3 Hz, J = 8.3 Hz), 6.93 (d, 1H, J = 8.3 Hz),
6.94 (d, 1H, J = 2.3 Hz). 13C-NMR (DCl): d = 35.24, 45.53, 49.08,
118.26, 119.18, 123.47, 131.55, 147.17, 147.20, 176.79. Mass
spectrum: m/z 244.0567 (M+H)+ for C10H14N1O4S1 (À2.1 mDa).
Elemental analysis: calculated for C10H13N1O4S1ÁHClÁH2O. C,
40.34%; H, 5.42%; N, 4.70%; S, 10.77%; Cl, 11.91%; found C,
40.72%; H, 5.27%; N, 4.72%; S, 10.79%; Cl, 13.05%.
Synthesis of 5-S-Cys-CMT-DA
A solution of 47 mg (0.16 mmol) 5-S-Cys-NE in 9.4 mL 6 M HCl
containing 5% thioglycolic acid and 1% phenol was refluxed for
30 min at 110°C. The reaction mixture was washed twice with
20 mL ether to remove phenol. The water layer was evaporated to
dryness and then subjected to Dowex 50W-X2 chromatography
(1.6 9 10 cm in 2 M HCl). The column was eluted with 2 M HCl
and fractions of 10 mL were collected. Fractions 16–26 were
evaporated to give 26 mg (14% yield) of 5-S-Cys-CMT-DAÁHCl
salt as a colorless solid (HPLC purity: 96%). UV kmax 292 nm (e
Alkaline H2O2-mediated oxidation of SN-NM and LC-NM
produced TDCA, TTCA, PDCA, and PTCA at levels similar
between SN-NM and LC-NM. Levels of TDCA and TTCA
were greater than those of PDCA and PTCA both in SN-NM
and LC-NM, which is consistent with the incorporation of
Cys as a benzothiazole moiety (Wakamatsu et al. 2012).
HI hydrolysis of SN-NM and LC-NM gave a similar
pattern of catechols, i.e., DOPA, DA, and DOPAC and their
aminohydroxyphenyl counterparts, i.e., 4-AHP, 4-AHPEA,
and 4-AHPAA. AHEBs isomers were also detected
(Table 1). DA was detected at the highest levels in both
types of NMs, while NE was not detected (Wakamatsu et al.
1
1,340) in 0.1 M HCl. H-NMR (DCl): d = 3.48 (m, 2H), 3.55 (m,
4H), 4.21 (m, 2H), 6.99 (d, 1H, J = 2.0 Hz), 7.10 (d, 1H,
J = 2.4 Hz). 13C-NMR (DCl): d = 35.2, 36.4, 45.4, 49.0, 54.7,
119.3, 121.0, 128.5, 131.7, 147.7, 148.6, 172.7, 176.7. 13C-NMR
© 2015 The Authors. Journal of Neurochemistry published by John Wiley & Sons Ltd on behalf of
International Society for Neurochemistry, J. Neurochem. (2015) 135, 768--776

 Wakamatsu, Kazumasa
Wakamatsu, Kazumasa
 Tabuchi, Keisuke
Tabuchi, Keisuke
 Ojika, Makoto
Ojika, Makoto
 Zucca, Fabio A.
Zucca, Fabio A.
 Zecca, Luigi
Zecca, Luigi
 Ito, Shosuke
Ito, Shosuke