Journal of Medicinal Chemistry
ARTICLE
(A) containing 0.05% HCOOH, with a linear gradient elution program
from 5% to 95% eluent B within 25 min at a 1.0 mL/min flow rate. The
detection was carried out at UV wavelengths of 254 nm. HPLC
purification was performed on a Gilson preparative HPLC. Melting
points were uncorrected and measured using a Yanaco MP-J3 micro-
melting point apparatus (Japan). Optical rotation was measured using a
model 341LC polarimeter (Perkin-Elmer). All NMR experiments were
carried out on a Varian Mercury 300, 500, or 600 MHz NMR spectro-
meter (Palo Alto, CA). Chemical shifts were reported in ppm (δ)
relative to the solvent signal, and coupling constants (J) were reported in
Hz. IR spectra were recorded on a Nicolet Impact 400 (San Jose, CA).
High resolution LCꢀMS was performed by Agilent Technologies LC/
MSD TOF using a column of Agilent ZORBAX SB-C18 (rapid resolu-
tion, 3.5 μm, 30 mm ꢁ 2.1 mm) at a flow of 0.4 mL/min. The solvent
was MeOH/H2O = 75:25 (v:v) containing 0.1% HCOOH. The ion
source is electrospray ionization (ESI). All the synthetic compounds
reported in this article possess a purity of at least 95%, which are all
identified through HPLC analysis (Supporting Information).
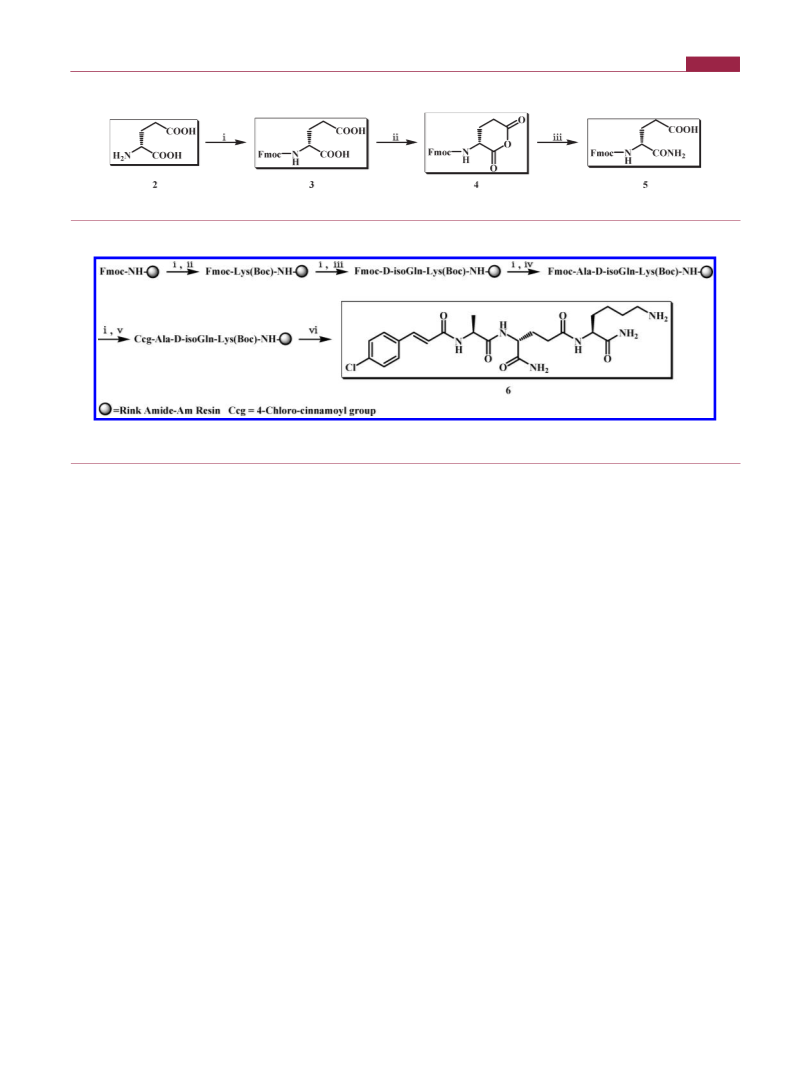
Preparation of Fmoc-isoGln-OH (5). To a vigorously stirred
solution of D-glutamic acid (2) (29.4 g, 1.0 equiv) in a mixture of acetone
and H2O with an equal volume ratio in a waterꢀice bath, NaHCO3
(18.5 g, 1.1 equiv) and Fmoc-OSu (67.4 g, 1.0 equiv) were added slowly
and reacted for an additional 3 days at rt. The mixture was then cooled in
a waterꢀice bath again, and 2.0 N HCl was carefully added until pH 2ꢀ3
was attained. After removal of acetone under reduced pressure, the
remaining solution was successfully extracted with EtOAc (400 mL, 3
times). The organic layer was separated and combined, dried with
MgSO4 overnight, and concentrated to a small volume under reduced
pressure. Then cyclohexane (600 mL) was added into the remaining
solution to suspend the solid. After filtration, 58.9 g of aimed product 3
was obtained as a white solid with a yield of 79%. 3 (58.9 g, 1.0 equiv)
was dissolved in anhydrous THF (324 mL). DCC (40.1 g, 1.2 equiv) was
then added while stirring in a waterꢀice bath. The reaction mixture was
allowed to warm to rt and stirring was maintained for an additional 10 h
to produce 4. The precipitates were filtered off. Dry ammonia gas was
then bubbled through the reactants while stirring in a NaCl saltꢀice
bath. When no more white solid was precipitated after 1.5 h, the reaction
was completed. MeOH (300 mL) was added to dissolve the solid. The
mixture was cooled in a waterꢀice bath again. Then 2.0 N HCl was
carefully and slowly added until pH 2ꢀ3 was attained. The solvent was
evaporated to dryness in a vacuum. The resulting solid was dissolved in
EtOAc and then washed with diluted HCl, saturated aqueous NaHCO3
solution, and H2O sequentially. The organic layer was separated and
combined, dried with MgSO4 overnight, and evaporated in a vacuum.
The precipitate was dissolved in DMF (200 mL). DCM (800 mL) was
20% piperidine/DMF for 1 h at rt, followed by thorough washing with
DMF (500 mL) and DCM (500 mL), respectively, three times each.
Fmoc-Lys(Boc)-COOH (61.8 g, 1.5 equiv), HOBt (17.8 g, 1.5 equiv),
and DIC (20.8 mL, 1.5 equiv) were dissolved in DMF (500 mL), then
poured into the vessel, followed by gentle shaking for 8 h at rt. When the
Kaiser test was negative, the coupling reaction was completed. After the
Fmoc deprotection and washing were repeated, Fmoc-D-isoGln-OH
(48.5 g, 1.5 equiv, for 12 h at rt), Fmoc-Ala-OH (41.0 g, 1.5 equiv, for 8 h
at rt), and 4-chlorocinnamic acid (24.1 g, 1.5 equiv, for 8 h at rt) were
successfully assembled onto the resin, which led to the resin-bound 6.
Targeted compound 6 was finally cleaved off the resin by using 90%
TFA/H2O for 2 h at rt. The cocktail cleavage solution was concentrated
under reduced pressure. Then dried Et2O was added into the residue
under cooling conditions by an ice bath, followed by the same procedure
repeated three times. Rude product 6 (52.5 g) was obtained after
filteration in 96% yield. The aired dried powder was purified and
lyophilized to yield final product 6 as a white powder in 98% purity
by ODS column chromatography with gradient elution from 5:95 to
25
589
50:50 (v:v) MeOH/H2O, mp = 215ꢀ217 °C, [R] þ37.7° (c 11.0
mg/mL, DMF). 1H NMR (600 MHz, DMSO-d6), lysine part: δ 7.90 (d,
1H, J = 8.4 Hz, NH), 7.10 (s, 1H, CONHa), 7.30 (s, 1H, CONHb), 4.11
(m, 1H, R-H), 1.46 (m, 1H, β-Ha), 1.63 (m, 1H, β-Hb), 1.27 (m, 2H, γ-
H), 1.53 (m, 2H, δ-H), 2.73 (m, 2H, ε-H), 7.75 (br s, 2H, NH2). D-
Isoglutamine part: δ 8.21 (d, 1H, J = 8.4 Hz, NH), 6.98 (s, 1H, CONHa),
7.41 (s, 1H, CONHb), 4.14 (m, 1H, R-H), 1.71 (m, 1H, β-Ha), 1.97 (m,
1H, β-Hb), 2.15 (t, 2H, J = 7.2 Hz, γ-H). Alanine part: δ 8.39 (d, 1H, J =
6.6 Hz, NH), 4.38 (m, 1H, R-H), 1.26 (m, 3H, β-H). 4-Chlorocinna-
moyl part: δ 6.75 (d, 1H, J = 15.9 Hz, trans-R-H), 7.39 (d, 1H, J = 15.9
Hz, trans-β-H), 7.57 (d, 2H, J = 8.4 Hz, ph-o-H), 7.47 (d, 2H, J = 8.4 Hz,
ph-m-H). 13C NMR (150 MHz, DMSO-d6), lysine part: δ 173.3
(CONH2), 52.1 (R-C), 31.3 (β-C), 22.4 (γ-C), 26.8 (δ-C), 38.7 (ε-
C). D-Isoglutamine part: δ 173.8 (CONH2), 52.2 (R-C), 27.7 (β-C),
31.7 (γ-C), 171.6 (CONH). Alanine part: δ 172.4 (CONH), 48.8 (R-
C), 18.1 (β-C). 4-Chlorocinnamoyl part: δ 164.7 (CONH), 122.7
(trans-R-C), 137.6 (trans-β-C), 133.8 (ph-q-C), 129.2 (ph-o-C), 129.0
(ph-m-C), 134.0 (ph-p-C). IR (KBr): 3281.5, 3198.9, 3063.4, 2935.2,
1609.8, 1539.0, 1452.2, 1200.2, 1134.1, 973.6, 821.6, 799.8, 720.2 cmꢀ1
.
HRMS (TOF): observed for 509.2270 [M þ H]þ, calcd for 509.2274
[M þ H]þ, C23H33ClN6O5.
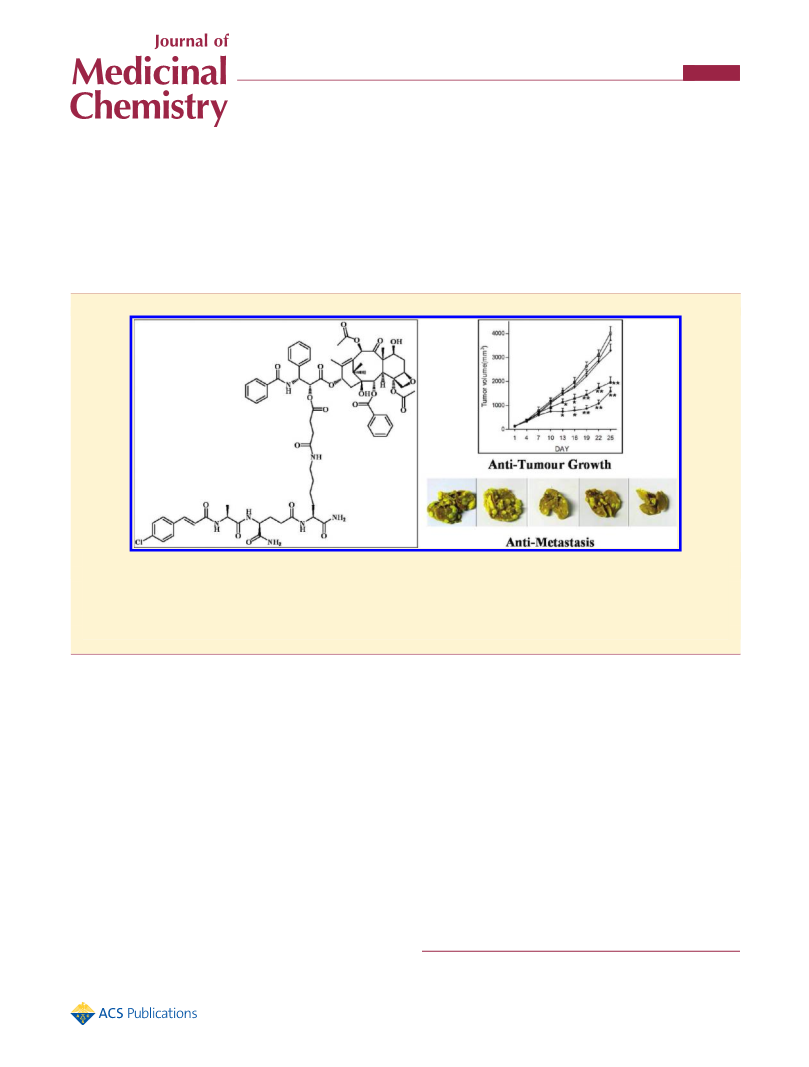
Conjugation of Building Blocks To Prepare 1. A solution of
20-succinylpaclitaxel (7) (9.53 g, 1.0 equiv), which was obtained by
Deutsch’s method,48 HOSu (1.15 g, 1.0 equiv), and EDC HCl (1.92 g,
3
1.0 equiv) in DMSO was stirred at rt for 8 h to produce 8. Then 6 (5.08 g,
1.0 equiv) was added to the solution slowly and the pH value of solution
was carefully adjusted to 7ꢀ8 by NMM. The reaction was completed
after an additional 30 min of stirring at rt. Then water was slowly added
to the solution, which precipitated out crude product 1. The filtrate was
purified by preparative HPLC with a linear gradient from 50:50 to 95:5
(v:v) ACN/H2O over 15 min at 3 mL/min flow rate at 254 nm UV
detected wavelength and lyophilized to give a white product 1 (13.0 g) in
then slowly added, resulting in the anticipated product 5 (37.0 g) as a
25
589
white solid with a yield of 63%, mp = 204ꢀ205 °C, [R] ꢀ4.2° (c 10.0
mg/mL, DMF). 1H NMR (300 MHz, DMSO-d6), Fmoc part: δ 7.88 (d,
2H, J = 7.8 Hz, ph-H), 7.72 (m, 2H, ph-H), 7.42 (m, 2H, ph-H), 7.32 (m,
2H, ph-H), 4.27 (m, 2H, CH2), 4.20 (m, 1H, CH). D-Isoglutamine part:
δ 7.40 (m, 1H, NH), 7.04 (br s, 1H, CONHa), 7.29 (br s, 1H, CONHb),
3.93 (dd, 1H, J = 13.5 and 8.5 Hz, R-H), 1.73 (m, 1H, β-Ha), 1.89 (m,
1H, β-Hb), 2.25 (m, 2H, γ-H), 12.08 (br s, 1H, COOH). 13C NMR (125
MHz, DMSO-d6), Fmoc part: δ 155.9 (OCONH), 143.8 (ph-C), 140.7
(ph-C), 127.6 (ph-CH), 127.0 (ph-CH), 125.3 (ph-CH), 120.0 (ph-
CH), 65.6 (CH2), 46.6 (CH). D-Isoglutamine part: δ 173.4 (CONH2),
53.8 (R-C), 27.2 (β-C), 30.4 (γ-C), 173.9 (COOH). HRMS (TOF):
observed for 369.1439 [M þ H]þ, calcd for 369.1445 [M þ H]þ,
C20H20N2O5.
90% yield, mp = 180ꢀ181 °C, [R] ꢀ9.8° (c 10.0 mg/mL, DMF). 1H
25
589
NMR (600 MHz, DMSO-d6): paclitaxel part: δ 4.62 (br s, 1H, 1-OH),
5.40 (d, 1H, J = 7.2 Hz, 2-H), 3.56 (d, 1H, J = 7.2 Hz, 3-H), 4.89 (m, 1H,
5-H), 1.62 (m, 1H, 6-Ha), 2.30 (m, 1H, 6-Hb), 4.10 (m, 1H, 7-H), 4.90
(m, 1H, 7-OH), 6.28 (s, 1H, 10-H), 5.81 (t, 1H, J = 9.0 Hz, 13-H), 1.46
(m, 1H, 14-Ha), 1.79 (m, 1H, 14-Hb), 0.99 (s, 3H, 16-H), 1.01 (s, 3H,
17-H), 1.75 (s, 3H, 18-H), 1.49 (s, 3H, 19-H), 3.98 (d, 1H, J = 8.4 Hz,
20-Ha), 4.01 (d, 1H, J = 8.4 Hz, 20-Hb), 2.23 (s, 3H, 4-OCOCH3), 2.09
(s, 3H, 10-OCOCH3), 5.33 (d, 1H, J = 9.0 Hz, 20-H), 5.52 (t, 1H, J = 8.4
Hz, 30-H), 9.21 (d, 1H, J = 8.4 Hz, 30-NH), 7.48 (d, 2H, J = 7.8 Hz, ph-o-
H), 7.46 (m, 2H, ph-m-H), 7.54 (m, 1H, ph-p-H), 7.84 (d, 2H, J = 7.2
Hz, NBz-o-H), 7.42 (m, 2H, NBz-m-H), 7.17 (m, 1H, NBz-p-H), 7.97
(d, 2H, J = 7.2 Hz, OBz-o-H), 7.66 (t, 2H, J = 7.2 Hz, OBz-m-H), 7.72 (t,
1H, J = 7.2 Hz, OBz-p-H). Succinic part: δ 2.60 (m, 2H, OCOCH2),
Synthesis of Nr-[4-Chlorocinnamoyl-L-alanyl-D-isogluta-
minyl]-L-lysine (MDA, 6). Fmoc protected Rink amide AM resin
(100.0 g, 1.0 equiv) was put into a vessel and vacuumed under reduced
pressure. After 1 h, dried DCM (500 mL) was added to swell the resin for
45 min. The Fmoc group of resin was next removed with treatment of
2774
dx.doi.org/10.1021/jm101577z |J. Med. Chem. 2011, 54, 2767–2777

 Ma, Yao
Ma, Yao
 Zhao, Nan
Zhao, Nan
 Liu, Gang
Liu, Gang