Journal of Agricultural and Food Chemistry
Article
with methanol and water (70:30 v/v) with a flow rate of 5 mL/min.
The eluent was monitored at 360 nm. Each fraction was collected,
evaporated, lyophilized, diluted to 0.2 mL with methanol, and further
analyzed on a MS/MS system.
Quercetin-3-O-docosahexaenoate. Yellow powder; ESI-MS: m/z
611.3 [M − H]−; 1H NMR (600 MHz, DMSO-d6): δ = 12.22 (s, 1H;
OH-5), 10.99 (s, 1H; OH-7), 9.87 (s, 1H; OH-4′), 9.50 (s, 1H; OH-
3′), 7.32 (d, J = 2.3 Hz, 1H; H-2′), 7.25 (dd, J = 8.4, 2.3 Hz, 1H; H-
6′), 6.90 (d, J = 8.5 Hz, 1H; H-5′), 6.47 (d, J = 2.1 Hz 1H; H-8), 6.25
(d, J = 2.1 Hz 1H; H-6), 5.41−5.24 (m, 12H, H-4″, H-5′′, H-7′′, H-
8′′, H-10′′, H-11′′, H-13′′, H-14′′, H-16′′, H-17′′, H-19′′, H-20′′),
2.83−2.74 (m, 10H; H-6′′, 9′′, 12′′, 15′′, 18′′), 2.71 (t, J = 7.3 Hz,
2H; H-2″), 2.41 (q, J = 7.0 Hz, 2H; H-3″), 2.07−1.96 (m, 2H; H-
21′′), 0.94−0.88 (m, 3H; H-22′′); 13C NMR (150 MHz, DMSO-d6):
δ = 174.85 (C-4), 170.00 (C-1″), 164.07 (C-7), 161.07 (C-9), 156.57
(C-5), 155.93 (C-4′), 149.29 (C-2), 145.48 (C-3′), 131.50 (C-3),
129.57 (C-20′′), 128.96 (C-5′′), 128.50 (C-17′′), 128.38 (C-8′′),
128.10 (C-11′′), 128.06 (C-13′′), 127.86 (C-10′′), 127.80 (C-7′′),
127.76 (C-14′′), 127.69 (C-4′′), 127.65 (C-19′′), 126.90 (C-10′′),
120.51 (C-1′), 119.55 (C-6′), 115.88 (C-2′′), 115.00 (C-5′), 103.43
(C-10), 99.03 (C-6), 94.04 (C-8), 33.06 (C-2′′), 25.19 (C-6′′), 25.15
(C-9′′), 25.12 (C-12′′), 25.10 (C-15′′), 25.06 (C-18′′), 22.17 (C-
3′′), 20.00 (C-21′′), 14.07 (C-22′′).
MS Analysis of Quercetin Derivatives. Quercetin derivatives were
analyzed on an Agilent 1290 liquid chromatography system equipped
with a Q-TOF 6550 mass spectrometer (Agilent Technologies, CA,
USA). Separation was carried out on a Waters BEH C18 column (1.7
μm, 100 × 2.1 mm). The mobile phase was composed of 0.1% formic
acid in water (solvent A) and methanol (solvent B). The flow rate was
0.3 mL/min in the following gradients: 0−2 min, 80% B. The MS
conditions: negative ion mode; mass range, 50−1000 m/z; capillary
temperature, 350 °C; spray voltage, 4 kV; collision energy 20 eV.
Synthesis of Quercetin Ester Derivatives as Reference
Compounds. EPA and DHA derivatives of quercetin were
synthesized by reacting of quercetin with EPA and DHA chlorides,
respectively. EPA and DHA were prepared by saponification of EPA
and DHA ethyl esters, respectively, according to previous studies.18,19
Briefly, 5 g of EPA ethyl ester (or DHA ethyl ester), 1.15 g of KOH,
2.2 mL of distilled water, and 13.2 mL of 95% ethanol were mixed
thoroughly and incubated under nitrogen reflux at 62 2 °C for 1 h.
Then, 50 mL of distilled water and 100 mL of hexane were added to
the mixture and the unsaponifiable matters (hexane layer) were
removed by using a separatory funnel. After that, the pH value of the
aqueous layer containing saponifiable matters was adjusted to 1.0 with
HCl (3M). The released free fatty acid in the aqueous layer was
extracted with 12.5 mL of hexane four times. The hexane layer was
combined, passed through anhydrous sodium sulfate, and evaporated
to a minimum volume at 35 °C under vacuum in a rotary evaporator.
EPA and DHA were converted to their corresponding acyl chlorides
by reaction with thionyl chloride. Briefly, 3 mL of thionyl chloride was
added dropwise to 5 g of EPA or DHA in a three-necked round-
bottom flask under nitrogen reflux at 70 °C in a water-bath for 1 h.
Quercetin was esterified with EPA chloride [C20:5] and DHA
chloride [C22:6] according to a reported method.19 Briefly, 0.8 mL of
pyridine was added dropwise to quercetin (10 mmol) dissolved in 250
mL of ethyl acetate. Acyl chloride solution (5 mL) was added
dropwise to the mixture. The mixture was then heated in a water bath
at 50 °C for 3 h with constant stirring. After cooled down, the mixture
was filtered using a filter paper and washed with distilled water (60
°C) three times. The ethyl acetate layer was collected, passed through
anhydrous sodium sulfate, and evaporated to a minimum volume
under vacuum in a rotary evaporator. The crude quercetin ester
mixtures were redissolved in hexane/ethyl acetate (50:50, v/v) and
purified by a silica column with a gradient of hexane/ethyl acetate
(90:10−50:50, v/v). The eluate was collected using an automatic
fraction collector. The collected fraction profile was checked by
HPLC. With this process, quercetin-EPA and quercetin-DHA
derivatives in high purity values (>95% by HPLC-PDA) were
obtained. The structures of the quercetin derivatives were determined
by ESI-MS and NMR spectroscopy (Bruker, AVANCE 600).
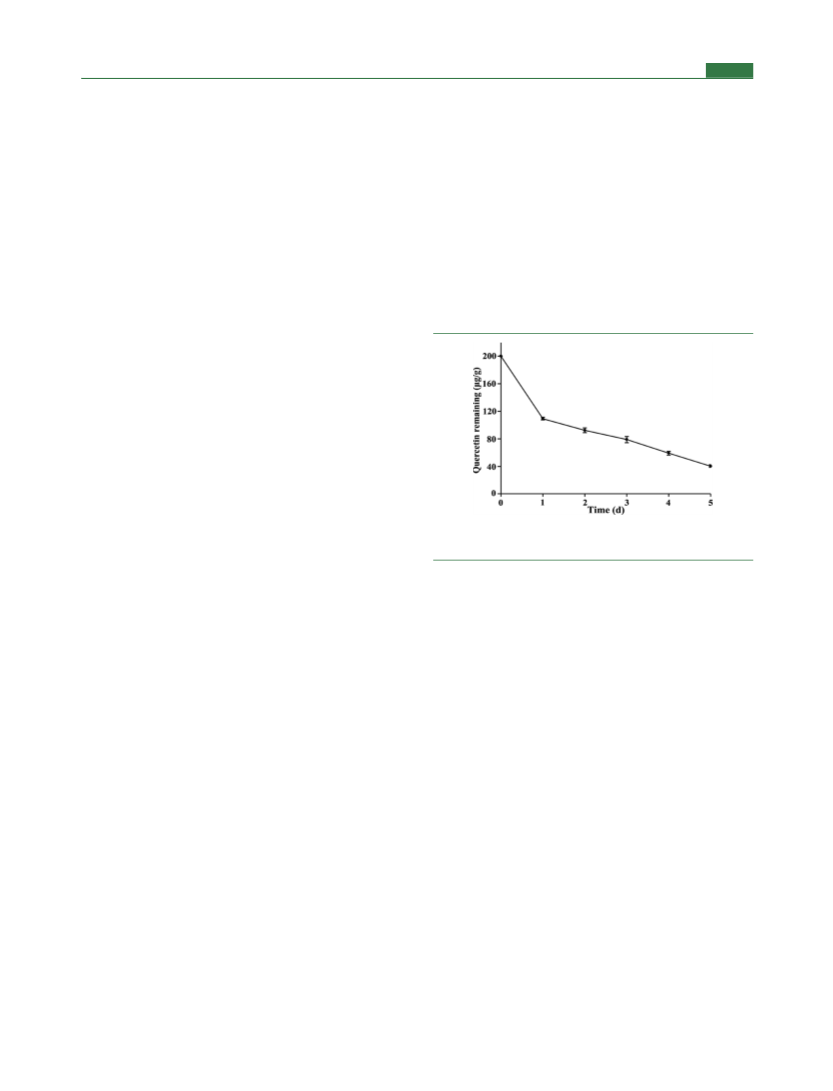
Quantitative Analysis of Quercetin Ester Derivatives in Fish
Oil by UPLC-MS/MS. Quercetin (20 mg) was mixed with fish oil (2
g) in a screw cap-sealed reaction vial. The oil samples were heated in a
water bath at 60 °C ( 2 °C) for 5 days. The mixture was extracted
with 8 mL of methanol and water (1:1, v/v) using a vortex mixer for 5
min. After centrifugation at 4000 rpm for 3 min, the supernatant was
transferred to a tube for extraction twice more, each time with 4 mL
of methanol and water (1:1, v/v). The lower layer was combined and
evaporated to a minimum volume at 40 °C under vacuum in a rotary
evaporator. The residue was then lyophilized and redissolved in 1 mL
of methanol, sonicated, and filtered through a 0.22 μm membrane
before UPLC-MS/MS analysis.
Quercetin derivatives were determined using an Agilent 1290
UPLC system equipped with an Agilent Q-TOF 6550 mass
spectrometer. Separation was conducted on an Agilent Extend C18
column (1.7 μm, 50 × 2.1 mm). The mobile phase was composed of
5% acetonitrile in water (solvent A) and methanol (solvent B). The
flow rate was 0.3 mL/min in the following gradients: 0−10 min, 80%
B. The MS conditions were as follows: negative ion mode; spray
voltage, 2.8 V; cone voltage, 30 V; source temperature, 120 °C;
desolvation temperature, 400 °C. Purified quercetin-EPA and
quercetin-DHA were used as reference standards, and MRMs at m/
z 585.2/301.03/178.9 and m/z 611.3/301.0/151.0 were used for MS
analysis, respectively.
Lipophilicity of Quercetin and Quercetin Derivatives. The
lipophilicity of quercetin and its derivatives was calculated using
ALOGPS 2.1 according to a previous work.20 The simplified
molecular input line entry (SMILE) structures of the tested chemicals
were drawn using ChemDraw V10.0.
Antioxidant Activity of Quercetin-EPA and Quercetin-DHA
Derivatives. DPPH Radical Scavenging Activity. The DPPH radical
scavenging ability of quercetin and quercetin derivatives was
measured according to the reported method with slight modifica-
tions.21 Briefly, 1 mL of quercetin and its derivatives (150 μg/mL)
dissolved in 95% methanol was mixed with 2 mL of methanolic
DPPH solution (0.05 mg/mL). After kept at room temperature for 30
min in the dark, the absorbance of the mixture was measured at 517
nm on a spectrophotometer (Unico 2100, Shanghai, China). Radical
scavenging ability (RSA) was expressed as percent DPPH inhibition
and calculated using the following equation: DPPH RSA (%) = [(A0
− As) / A0] × 100, where A0 is the absorbance of methanolic DPPH
solution; As is the absorbance of the sample.
ABTS Radical Cation Scavenging Activity. ABTS radical
scavenging activity of quercetin and quercetin derivatives was
measured by the ABTS cation decolorization assay as described
previously with slight modifications.21,22 In brief, the ABTS radical
cation (ABTS·+) was prepared by mixing of 0.2 mL 7.4 mM ABTS
solution with 0.2 mL of 2.6 mM potassium persulfate. The mixture
was stored at room temperature in the dark for 12 h to allow
completion of radical generation. The ABTS•+ solution was then
diluted with ethanol to achieve an absorbance of 0.70 0.02 at 734
Quercetin-3-O-eicosapentaenoate. Yellow powder; ESI-MS: m/z
1
585.2 [M − H]−; H NMR (600 MHz, DMSO-d6): 7.31 (d, J = 2.3
Hz, 1H; H-2′), 7.22 (dd, J = 8.4, 2.3 Hz, 1H; H-6′), 6.88 (d, J = 8.4
Hz 1H; H-5′), 6.38 (d, J = 2.0 Hz, 1H; H-8), 6.16 (d, J = 2.0 Hz, 1H;
H-6), 5.39−5.24 (m, 10H, H-5″, H-6′′, H-8′′, H-9′′, H-11′′, H-12′′,
H-14′′, H-15′′, H-17′′, H-18′′), 2.81−2.73 (m, 8H; H-7′′, H-10′′, H-
13′′, H-16′′), 2.63 (t, J = 7.3 Hz, 2H; H-2″), 2.16 (q, J = 7.2 Hz, 2H;
H-4″), 2.08 (q, J = 7.5 Hz, 2H; H-19″), 1.71(q, J = 7.5 Hz, 2H; H-
3″), 0.94−0.88 (m, 3H; H-20′′); 13C NMR (150 MHz, DMSO-d6): δ
= 174.49 (C-4), 170.47 (C-1″), 167.47 (C-7), 161.02 (C-9), 156.80
(C-5), 155.50 (C-4′). 150.07 (C-2), 145.88 (C-3′), 131.52 (C-3),
129.42 (C-18′′), 129.31(C-6′′), 128.87 (C-17′′), 128.61 (C-8′′),
128.08 (C-11′′), 128.04 (C-12′′), 127.87 (C-9′′), 127.79(C-15′′),
127.68(C-5′′), 126.93 (C-14′′), 120.31 (C-1′), 119.21 (C-6′), 115.90
(C-2′′), 114.75 (C-5′), 102.40 (C-10), 99.70 (C-6), 94.39 (C-8),
32.65 (C-2′′), 25.88 (C-4′′) 25.21 (C-16′′), 25.18 (C-13′′), 25.13
(C-7′′), 25.09 (C-22′′), 24.42 (C-3′′), 20.03 (C-19′′), 14.09 (C-
20′′).
1059
J. Agric. Food Chem. 2021, 69, 1057−1067

 Liu, Shaojun
Liu, Shaojun
 Zhu, Yamin
Zhu, Yamin
 Liu, Ning
Liu, Ning
 Fan, Daming
Fan, Daming
 Wang, Mingfu
Wang, Mingfu
 Zhao, Yueliang
Zhao, Yueliang