
Journal of Catalysis p. 155 - 169 (2004)
Update date:2022-08-16
Topics:
 Maeki-Arvela, Paeivi
Maeki-Arvela, Paeivi
 Kumar, Narendra
Kumar, Narendra
 Nieminen, Ville
Nieminen, Ville
 Sjoeholm, Rainer
Sjoeholm, Rainer
 Salmi, Tapio
Salmi, Tapio
 Murzin, Dmitry Yu.
Murzin, Dmitry Yu.
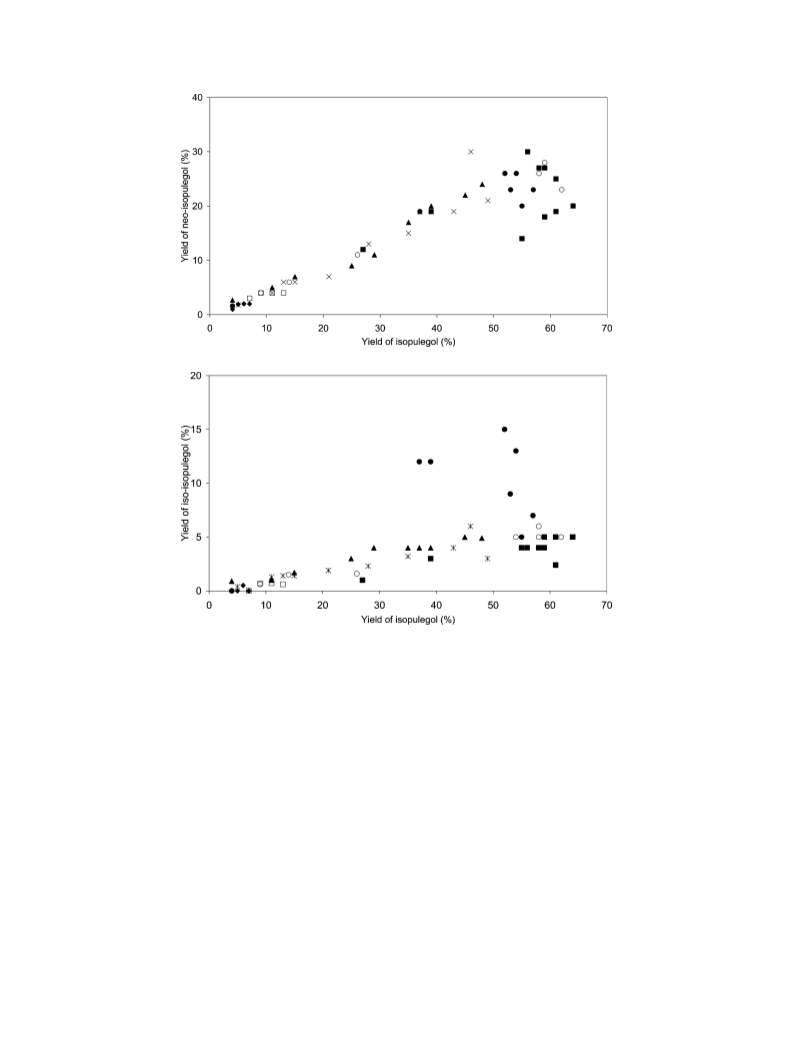
Cyclization of (+)-citronellal was investigated over zeolites and mesoporous materials as well as on silica under a nitrogen atmosphere in cyclohexane as a solvent. The highest cyclization rates were observed over mesoporous materials and 12-membered ring zeolites with high Bronsted acid concentration, while very low cyclization rates were achieved over silica with low or no Bronsted acidity, respectively. At the same a time low cyclization rate was observed over 10-membered ring pore H-ZSM-5 with a high Bronsted acid site concentration, which is due to diffusional limitation of the product in the narrow pores. The selectivity to cyclization products was very high over all the catalysts, being independent of the conversion of citronellal. Neither concentration of the Bronsted nor Lewis acid sites influenced the stereoselectivity to isopulegol. The support structure had only a minor effect on the stereoselectivity. Quantum mechanical calculations were carried out to explain the experimental results. The calculated stabilities of the carbocationic reaction intermediates correlated well with the observed stereoselectivity. The stereoselectivities were analogous when starting from racemic citronellal mixture or enantiopure (+)-citronellal; the former one gave 8 different pulegols, whereas only 4 pulegols were formed from (+)-citronellal.
View More











Henan Yellow River New Material Technology Co.Ltd.
website:http://www.yellowriverchem.com
Contact:0086-373-7278760
Address:Chengguan Town, Yuanyang County
AllyChem Co., Ltd., Dalian, China(BBChem)
Contact:+86-411-62313318/62313328
Address:No.5 of Jinbin Road, Jinzhou New District, Dalian City, Liaoning Province, P.R.China
Luzhou North Chemical Co., Ltd.
Contact:+86-830-2796784;+86-830-2796776
Address:Gaoba, Longmatan District, Luzhou, Sichuan Province
WUXI HONOR SHINE CHEMICAL CO.,LTD
Contact:+86-510-83593312
Address:No.1699 Huishan avenue,Huishan district,Wuxi ,Jiangsu,China,214177.(Wuxi Huishan Ecomonic Develop Zone )
Shanghai Hongbang Medical Technology CO.,. Ltd
Contact:13671516988 /18917636693
Address:Room1, No67 Building, Yongde Road369, Wujing Town, Minhang Districy, Shanghai CIty, China.
Doi:10.1134/S107042800702008X
(2007)Doi:10.1562/0031-8655(2003)078<0481:NAAEPP>2.0.CO;2
(2003)Doi:10.1039/C29700000523
(1970)Doi:10.1016/j.tet.2015.12.056
(2016)Doi:10.1039/c4cc01822b
(2014)Doi:10.1135/cccc19952057
(1995)