G.-K. Chuah et al.
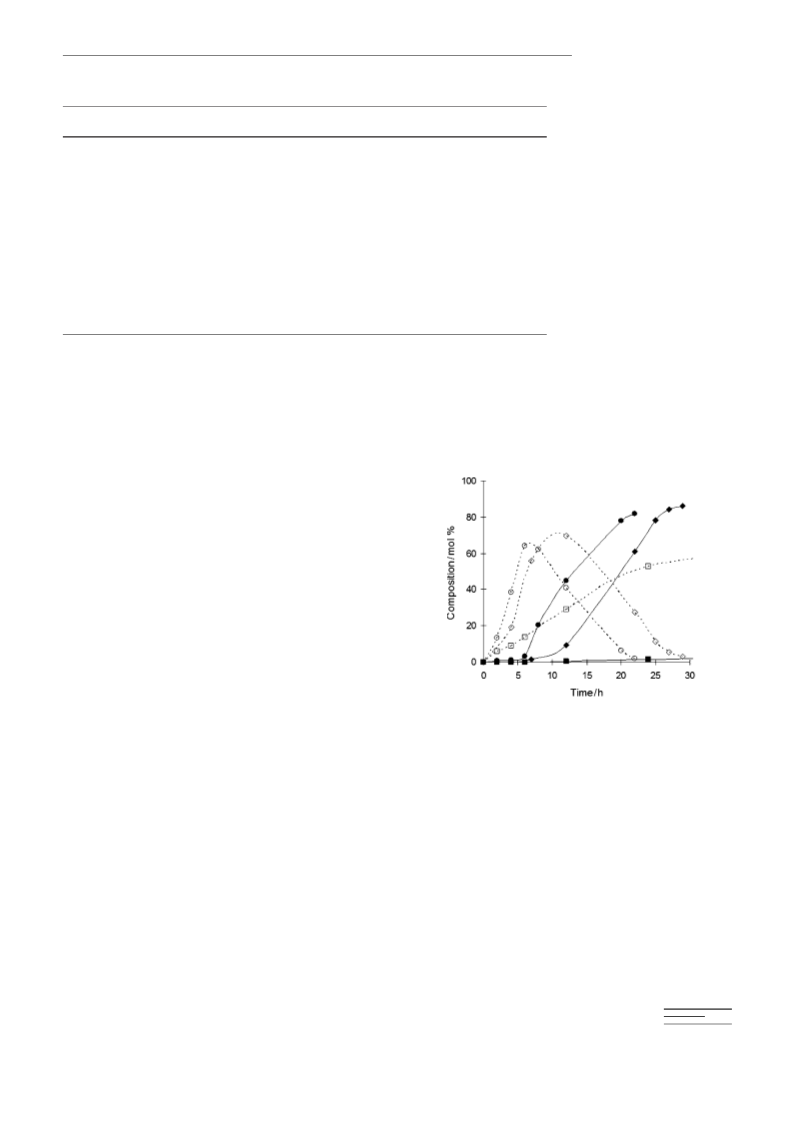
by the time all 2 had been converted, the selectivity for this
side product was already 22%. The final yield of menthols
was 72%. Although this does not reach the 95% yield ob-
tained with the Zr-beta–Ni/MCM-41 system, these results
show that it is possible to optimise a reaction with a dual
catalyst system because of the ease in adjusting the relative
amounts of the components.
beam slits. A step size of 0.028 and a dwell time of 1 s were used for the
measurements. A step size of 0.0048 was used for determining the crystal-
lite size of the deposited metal crystallites. The adsorption–desorption
isotherms for the solid samples were measured on a Micromeritics Tristar
porosimeter. The samples were degassed under a flow of nitrogen at
3008C for 4 h prior to measurement. TEM measurements were per-
formed with a JEOL JEM 3010 HRTEM microscope operated at 300 kV.
Infrared spectra were recorded on a Biorad Excalibur spectrometer with
À1
a resolution of 2 cm . The sample (5–8 mg) was pressed into a self-sup-
ported wafer and mounted in a Pyrex IR cell with NaCl windows. After
À3
evacuation under vacuum (10 mbar) for 2 h at 3008C, it was cooled to
Conclusions
room temperature and a background spectrum was recorded. Pyridine
was introduced at 22 mbar for 15 min and the system was evacuated for
an hour before measuring the spectrum at room temperature. Further IR
measurements were made after evacuation at 100 and 2008C.
The cascade transformation of citral to menthols occurs with
high selectivity when Zr-beta is used as a catalyst. With a Ni
loading of 10 and 15 wt%, the total yield of menthols was
Catalytic testing: The hydrogenation of citral (containing a mixture of cis
and trans isomers) was carried out in a Teflon-lined 100 mL autoclave
(Berghof) with stirring. The autoclave was charged with citral (1.7 mL,
8
8%, with a diastereoselectivity for (Æ)-menthol of 94%.
9
.9 mmol), catalyst (0.15 g for Zr-beta-supported metal catalysts and Pd-
Modification of the catalyst at the nanoscale is essential for
high catalytic activity. Whereas smaller metal crystallites
were obtained by the deposition–precipitation method than
wet impregnation, the urea used in this preparation de-
stroyed the zeolitic structure. As a consequence, the yield of
menthols was decreased in favour of open-chain 5 and 6. A
systems; 0.30 g for other dual catalyst systems, typically a 1:1 mixture),
tert-butanol (50 mL) as solvent and heated to 808C before hydrogen was
introduced into the head space to a pressure of 1 to 5.5 MPa. Samples
were removed at regular intervals and analysed by gas chromatography
(Agilent GC 6890 equipped with a HP-5 capillary column and an FID
detector). The temperature program used for separation of the products
À1
À1
was as follows: 608C for 2 min, 88Cmin to 1308C, then 258Cmin to
008C. The identity of the products was verified by comparison with the
2
wt% Rh/Zr-beta catalyst also exhibited good activity with
2
an 86% yield of menthols, although the hydrogenation of 8
to menthols in the final cascade step was slower than with
Ni/Zr-beta. A higher loading of 5 wt% Rh increased the
rate of hydrogenation, but more 5 and 6 were also formed.
A dual catalytic system of Zr-beta and 15 wt% Ni on
MCM-41 gave a high menthol yield of 95% when used to-
gether with hydrogen pressure ramping from 0.2 to 2 MPa.
The 94% diastereoselectivity for (Æ)-menthol shows that
cyclisation of 2 occurs over active sites on Zr-beta. Hence,
the use of Zr-beta in the cascade transformation of citral
constitutes a green route to menthols, given the ease of cata-
lyst separation from the reaction medium, minimal loss of
activity in subsequent batch cycles and excellent product
yield with very high diastereoselectivity.
retention times of authentic samples and by GC–MS (Shimadzu GCMS-
QP5000, DB5MS column). Figure S1 in the Supporting Information
shows a typical chromatogram obtained on completion of the reaction.
Each experiment was repeated at least twice with fresh or regenerated
catalysts. The deviation in the yields is in the order of Æ4%. The varia-
tion in the diastereoselectivity for the various isomeric products is in the
order of Æ1%. For regeneration, the bifunctional catalyst or dual catalyst
system was recovered by centrifugation, washed with tert-butanol and cal-
cined at 5508C for 4 h. After reduction at the respective temperature, de-
pending on the metal, the sample was cooled to room temperature in
helium and introduced into the autoclave for reuse. Regenerated cata-
lyst/dual catalytic system was reused up to five times. Mass balance was
checked by adding m-xylene (0.2 mL) as an internal standard. In the
pressure ramping experiments, the hydrogen pressure was maintained at
0
.2 MPa until all citral was converted and no more 2 was detected before
increasing to 2 MPa. A two-fold scale up of the experiment was carried
out with citral (20 mmol) and the dual catalyst system of Zr-beta–
1
5 wt% Ni/MCM-41. On completion of the reaction, the catalysts were
separated from the reaction products and the solvent was removed by
rotary evaporation.
Experimental Section
Preparation and characterisation of Zr-beta and MCM-41-supported
metal catalysts: The syntheses and characterisation of the catalysts have
Acknowledgements
[
18b,24]
previously been described.
The metals were deposited on the sup-
ports by wet impregnation at room temperature. Besides incipient wet-
ness impregnation, the homogeneous deposition–precipitation method
was also used to prepare a 15 wt% Ni/Zr-beta-HDP sample. Zr-beta (~
The authors would like to thank Mr. Yong Lim Foo for his help in inter-
pretation of the TEM images. Financial support from the National Uni-
versity of Singapore under grant number R-143-000-329-112 is gratefully
acknowledged.
[
25]
0
.85 g) was immersed in an aqueous solution of Ni
3 2
ACHTUNGTRENN(UG NO ) (0.74 g in
1
50 mL) and the pH was adjusted to pH 2 with nitric acid. The suspen-
sion was heated to 908C with stirring before an aqueous solution of urea
0.4 g in 10 mL) was added to effect precipitation. After stirring for 18 h,
the suspension was cooled to room temperature, filtered, washed, dried
at 1008C and calcined at 5008C for 4 h. Prior to catalytic testing, the sup-
ported Pd and Ni catalysts were reduced for 2 h in a flow of hydrogen at
(
[
1] T. Kieboom in Catalysis for Renewables: From Feedstock to Energy
Production (Eds.: G. Centi, R. A. van Santen), Wiley-VCH, Wein-
heim, 2007, pp. 273–297.
[
[
1
3
50 and at 4508C, respectively, whereas the Rh catalysts were reduced at
008C for 4 h. The sample was cooled to room temperature in helium,
before it was transferred to the autoclave for reaction. No further in situ
reduction was carried out in the autoclave.
Catalyst characterisation: The crystal phase of the samples was deter-
mined by powder X-ray diffraction by using a Siemens D5005 diffractom-
eter equipped with a Cu anode and variable primary and secondary
1998
ꢀ 2009 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Chem. Eur. J. 2009, 15, 1991 – 1999

 Nie, Yuntong
Nie, Yuntong
 Jaenicke, Stephan
Jaenicke, Stephan
 Chuah, Gaik-Khuan
Chuah, Gaik-Khuan