ORGANIC
LETTERS
2
011
Vol. 13, No. 9
180–2183
Intramolecular Pyridone/Enyne
Photocycloaddition: Partitioning
of the [4 þ 4] and [2 þ 2] Pathways
2
†
‡
‡
†
Svitlana Kulyk, William G. Dougherty Jr., W. Scott Kassel, Michael J. Zdilla, and
,†
Scott McN. Sieburth*
Department of Chemistry, Temple University, 1901 North 13th Street, Philadelphia,
Pennsylvania 19122, United States, and Department of Chemistry and Biochemistry, 800
Lancaster Avenue, Villanova University, Villanova, Pennsylvania 19085, United States
Received February 11, 2011
ABSTRACT
Intramolecular photocycloaddition (>290 nm) between a 1,3-enyne and a 2-pyridone is far more selective than the intermolecular version; a three-
atom linkage both controls regiochemistry and separates the [2 þ 2] and [4 þ 4] pathways. All four head-to-head, head-to-tail, tail-to-head, and tail-
to-tail tetherings have been investigated. Linkage via the ene of the enyne leads to [2 þ 2] products regardless of alkene geometry, whereas
linkage through the yne results in [4 þ 4] cycloadducts. The bridged 1,2,5-cyclooctatriene products of [4 þ 4] cycloaddition are unstable and
undergo a subsequent [2 þ 2] dimerization reaction.
1
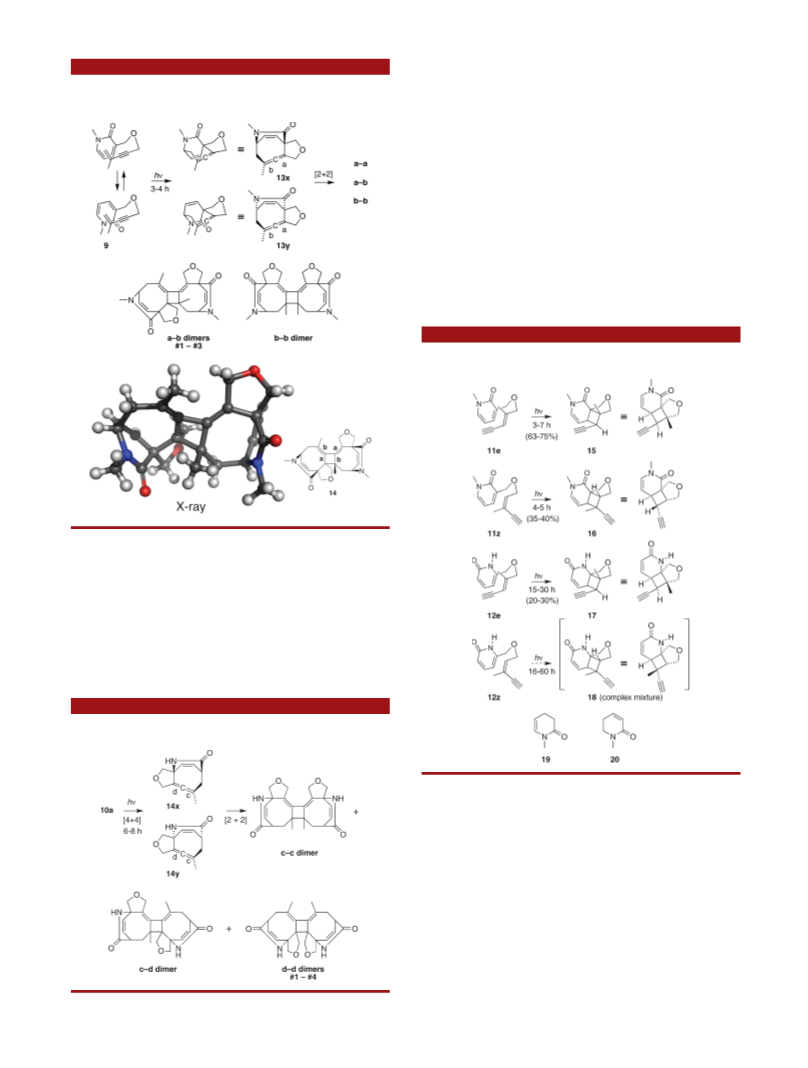
Pyridone photoreactivity is dominated by [4 þ 4] dimer-
ization reactions and, at low concentrations, 2-pyridones
will undergo isomerization to Dewar-pyridones, processes
that occur via a short-lived singlet excited state. When
mixed with1,3-dienes, furan, and napthalene, theycan also
undergo cross-[4 þ 4] cycloadditions.
2
We recently reported the first [4 þ 4] cycloadditions of
enynes, performed by irradiation of an enyne/2-pyridone
mixture, Scheme 1.
†
Temple University.
3ꢀ5
‡
This reaction of commercially avail-
Villanova University.
1) Sieburth, S. McN. In CRC Handbook of Organic Photochemistry
(
able reagents yields a rich mixture of products, derived from
a competition between [2 þ 2] and [4 þ 4] pathways, both of
which produce an array of regio- and stereochemical iso-
mers. Of the six potential constitutional isomers 3ꢀ8,
examples of all but 8 were isolated. Adding yet another
layer of complexity, the 1,2,5-cyclooctatriene products 3
and 4 were apparently unstable, giving [2 þ 2] dimers
and Photobiology; Horspool, W., Lenci, F., Eds.; CRC Press: Boca Raton,
FL, 2004; pp 103/1ꢀ103/18.
(
2) Sato, E.; Ikeda, Y.; Kanaoka, Y. Liebigs Ann. Chem. 1989, 781–
788. Sato, E.; Ikeda, Y.; Kanaoka, Y. Heterocycles 1989, 28, 117–120.
Sieburth, S. McN.; Zhang, F. Tetrahedron Lett. 1999, 40, 3527–3530.
Sieburth, S. McN.; McGee, K. F. J.; Zhang, F.; Chen, Y. J. Org. Chem.
2
000, 65, 1972–1977.
3) Kulyk, S.; Dougherty, W. G., Jr.; Kassel, W. S.; Fleming, S. A.;
Sieburth, S. McN. Org. Lett. 2010, 12, 3296–3299.
4) For examples of photosensitized intramolecular cycloaddition of
(
(
Scheme 2). The viability of the enyne [4 þ 4] cycloaddition
(
6
alkenes and alkynes with 2-pyridones, see: Somekawa, K.; Oda, H.;
Shimo, T. Chem. Lett. 1991, 12, 2077–2078. Somekawa, K.; Okuhira, H.;
Sendayama, M.; Suishu, T.; Shimo, T. J. Org. Chem. 1992, 57, 5708–
however, was demonstrated.
The postphotocycloaddition dimerization of 1,2,5-cy-
clooctatrienes 3 and 4, Scheme 2, dramatically enhances
5712.
(
5) For triplet-mediated intermolecular photocycloaddition of 1,3-
enynes with enones, and related chemistry, see: Witte, B.; Margaretha, P.
Org. Lett. 1999, 1, 173–174. Witte, B.; Meyer, L.; Margaretha, P. Helv.
Chim. Acta 2000, 83, 554–561. Schwebel, D.; Margaretha, P. Helv. Chim.
Acta 2000, 83, 1168–1174. Soltau, M.; G o€ wert, M.; Margaretha, P. Org.
Lett. 2005, 7, 5159–5161. Inh u€ lsen, I.; Margaretha, P. Org. Lett. 2010,
(6) The initially formed bridged 1,2,5-cyclooctatrienes 3 and 4 are
highly strained and most likely have allenes that are substantially bent,
possibly as much as 20°; see: Price, J. D.; Johnson, R. P. Tetrahedron
Lett. 1986, 27, 4679–4682. Johnson, R. P. Chem. Rev. 1989, 89, 1111–
1124. Daoust, K. J.; Hernandez, S. M.; Konrad, K. M.; Mackie, I. D.;
Winstanley, J.; Johnson, R. P. J. Org. Chem. 2006, 71, 5708–5714.
1
2, 728–730. Vall ꢀe e, M. R.; Inh u€ lsen, I.; Margaretha, P. Helv. Chim.
Acta 2010, 93, 17–24.
1
0.1021/ol200390j r 2011 American Chemical Society
Published on Web 03/29/2011

 Kulyk, Svitlana
Kulyk, Svitlana
 Dougherty, William G.
Dougherty, William G.
 Kassel, W. Scott
Kassel, W. Scott
 Zdilla, Michael J.
Zdilla, Michael J.
 Sieburth, Scott M.
Sieburth, Scott M.