4
40
P. Lanzafame et al. / Catalysis Today 175 (2011) 435–441
using the catalyst Al-MCM-41 (75), and only DE as reaction prod-
uct was observed with a yield of about 20%. The conversion of HMF
is also low, but higher than expected if only DE forms. No other
products of reaction were detected, apart from some formaldehyde.
Thus probably for this sample some heavier products which remain
on the catalyst may form. The carbon balance in the other samples
was instead typically close to 100%, apart for the SBA-15 meso-
porous silica, which also shows the formation of only DE as the
reaction product, but with a yield lower than expected from HMF
conversion. Therefore, both the samples showing the weaker acid-
ity (Al-MCM-41 (75) and SBA-15) have a similar behavior: lower
HMF conversion, DE as the main detected reaction product, and
defect in the carbon balance indicating the probable formation of
heavier reaction products.
On increasing the Al content in the Al-MCM-41 series, the
amount of DE decreases slightly, the amount of EOP increases sig-
nificantly becoming the main reaction product in Al-MCM-41 (25),
while the formation of the aimed product (EMF) passes through a
maximum.
In the case of zirconia supported over mesoporous silica, higher
selectivity to EMF is observed, while the formation of DE is absent.
The use of sulfated zirconia with respect to not-sulfated one
determines a lowering in the EMF yield, and a corresponding
increase in the yield of EOP. The mesoporous parent silica, e.g.
without zirconia, shows a lower activity in HMF conversion, and
only the formation of DE.
the degradation of HMF to levulinic acid (further then converted
to the ester), as discussed before. This result is also confirmed by
the data obtained using the MCM-41 based catalysts: when the
Brønsted acid sites strength on MCM-41 increases by introduction
of aluminum species in the framework (wall) of the mesoporous
structure (Al-MCM-41 (25)), EOP is the main product even if EMF
is also formed. In the sample with a lower Si/Al ratio (Al-MCM-
41 (50)), e.g. decreasing the amount of strong Brønsted acid sites,
the etherification of HMF to EMF is favored with respect to the
degradation reaction to EOP. Therefore, the EOP yield significantly
decreases with a parallel increase in EMF yield. As discussed below,
the formation of EMF is probably associated with the presence
of Lewis acid sites, due to extra-framework (isolated) Al3+ ions.
FTIR characterization (Fig. 4) confirms the presence of this type
of sites. Increasing the amount of Al in Al-MCM-41, increases
the amount of strong Brønsted acid sites, while probably isolated
3+
Al extra-framework sites tend to aggregate forming polymeric
Al-(hydr)oxide nanoparticles which do not show Lewis acidity.
Consistently, the formation of EMF decreases and significantly
increases the EOP yield.
The low acidity of Al-MCM-41 (75) catalyst is probably not
enough to activate the substrate to the degradation, and due to
the low amount of Al ions, Al3+ extra-framework sites were not
detected. Therefore, this sample results also inactive for the etheri-
fication of HMF. DE, deriving possibly from side reactions of ethanol,
is the only detectable product in low amounts. Note that the forma-
tion of this product is probably also favored by the catalyst, because
®
for other samples (zirconia on mesoporous silica, Amberlyst 15
4
. Discussion
and H SO ) it is not detected. NMR data (Tables 2 and 3) indi-
2
4
cate that the MCM-41 is highly defective, due to the presence
of hydroxyl nests. Probably these sites or traces of other transi-
tion metals such as iron present as impurities introduced during
the preparation catalyze the oxidation of ethanol to acetaldehyde,
which is the critical step for acetal (DE) formation. However, these
sites are also reasonably associated to slow side reactions respon-
sible for the conversion of HMF to undetected (probably heavier)
products. The mechanism should be better clarified, but being
present only in the less reactive and selective samples, it is of minor
interest.
4
.1. Reaction pathways
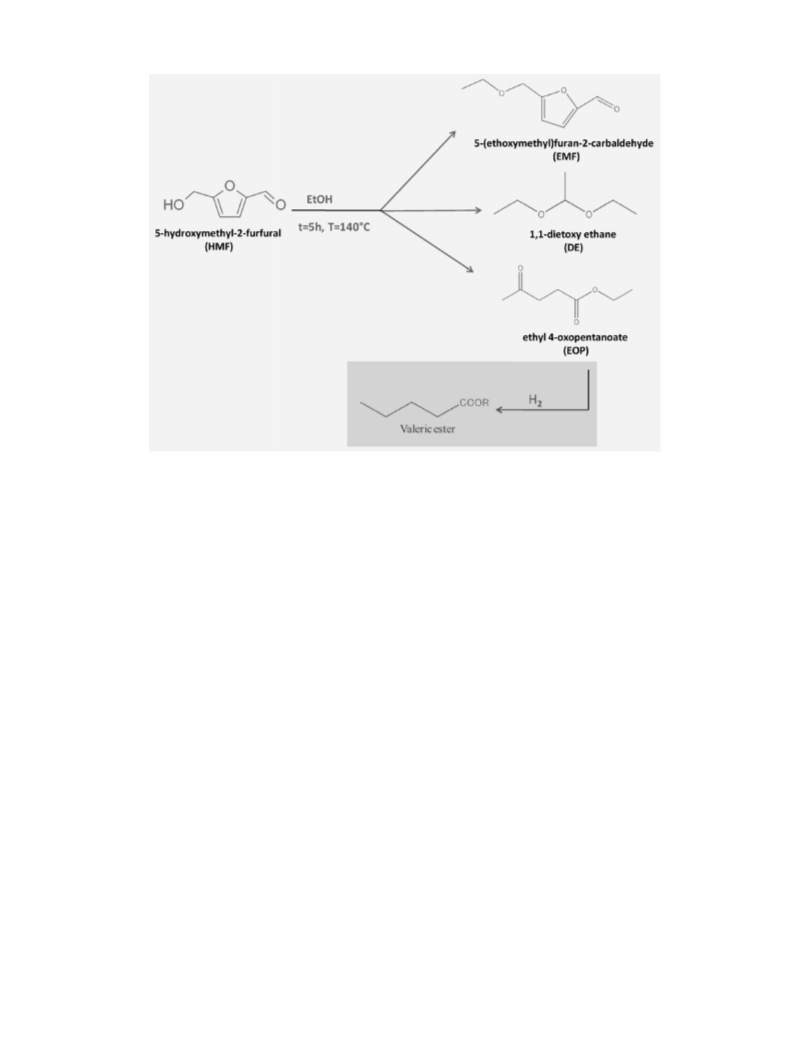
The main reaction network of the studied reaction is summa-
rized in Scheme 1. In addition, to the aimed biodiesel component
EMF, other products of interest for biodiesel and in general bio-
fuel production, such as EOP and DE were obtained, apparently in
parallel reactions.
Table 4 evidences the considerable effect on the selectivity
induced from the change of the acid properties of the mesoporous
catalyst.
Although no study on the reaction mechanism was made, the
formation of EMF can be easily explained with the well-known
mechanism of nucleophilic attack of ethanol on the oxonium ion
generated by interaction between the acidic sites on the catalyst
and the hydroxylic group on the HMF. The formation of EOP is
instead coherent with the acid catalyzed degradation mechanism of
HMF to levulinc acid [21] and the subsequent esterification reaction
with ethanol to give the corresponding ester (EOP). This pathway is
quite interesting because it could allow to obtain directly valerate
ester in a one pot reaction by direct hydrogenation of obtained EOP.
The formation of DE, although always associated to a moder-
ate conversion of HMF, can be not understood through a reaction
between HMF and ethanol, but it is probably due to the reac-
tion between ethanol and the small amount of acetaldehyde in
equilibrium with it [22]. HMF could be converted to products not
detectable in our analytical conditions. However, the study on the
mechanism and the conditions of this pathway should be investi-
gated more in detail, considering the possible use of DE as diesel
fuel additive.
The role of Lewis acidity in EMF synthesis is further demon-
strated from the performances of zirconia supported over
mesoporous silica. The parent sample (SBA-15) shows only very
weak acidity associated to the silanol group present in the inner
walls on the channels of SBA-15. The behavior is analogous to the
Al-MCM-41 (75). The introduction of ZrO2 determines an increase
of the Lewis acidity, associated to the surface exposed Zr4+ ions
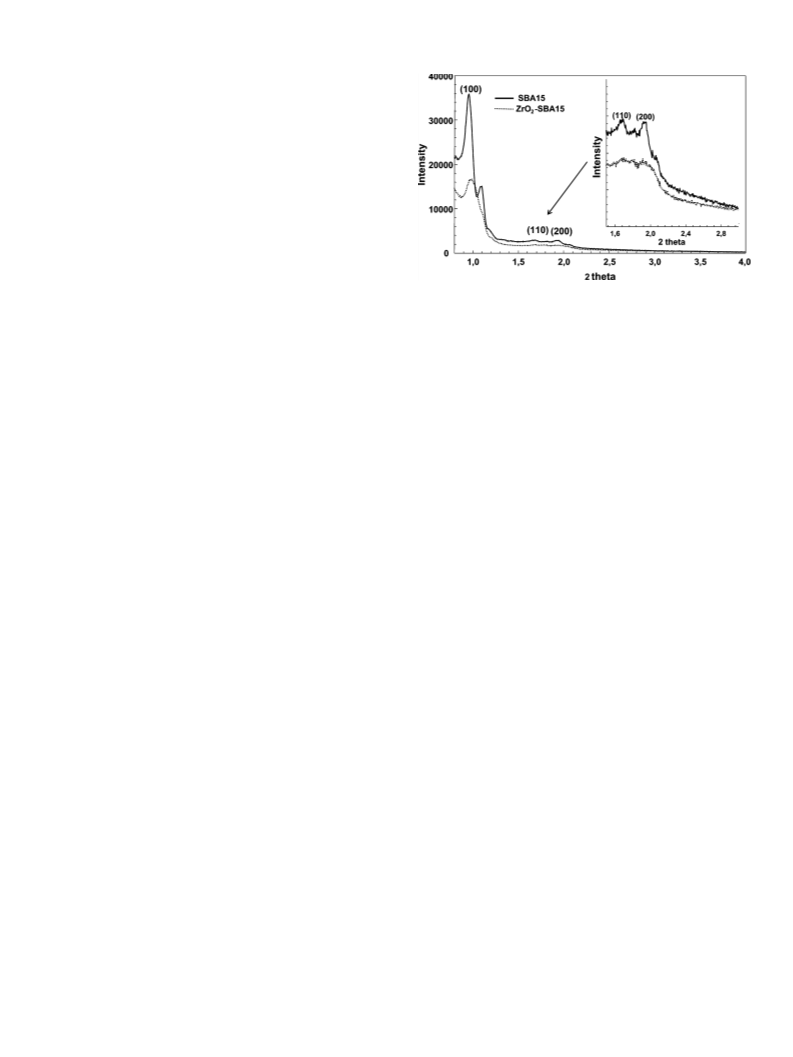
of XRD not detectable zirconia (amorphous or nanocrystalline)
well dispersed in the mesoporous silica (SBA-15). Electron micro-
graph characterization, XRD and porosity measurements confirm
the good dispersion of the zirconia. It is known that zirconia sup-
ported over silica shows both strong Lewis and Brønsted acidity
[23], and it is reasonable that the number of Lewis acid sites is
higher with respect to the limited amount of extra-framework Al3+
ions in Al-MCM-41. It is thus well consistent that Z-SBA-15 shows
a higher formation of the desired compound (EMF) together with
less amounts of EOP. By supporting the zirconia, the defect sites
present in the parent mesoporous silica are suppressed and thus
the formation of DE is inhibited.
As mentioned above, the sulfation of zirconia decreases the
Lewis acidity and increases the Brønsted acidity [13]. Consistently,
we observed that the yield in EOP on SZ-SBA-15 is higher with
respect to Z-SBA-15, while the yield in EMF decreases in parallel.
The adsorption of HMF on the Lewis acidic sites forms proba-
bly the oxonium ion and EMF is then obtained by the nucleophilic
attack of the ethanol. The presence of Lewis acidity is therefore
critical to address the reaction towards the etherification of HMF
4
.2. Role of the catalysts acidity
The remarkable influence of the catalyst characteristics on the
etherification of HMF (Table 4) can be rationalized in terms of
Lewis and/or Brønsted acidity properties of the catalysts. The high
selectivity to EOP showed by H SO4 and Amberlyst 15 is clearly
associated to the presence of strong Brønsted acidity that promotes
®
2

 Lanzafame
Lanzafame
 Temi
Temi
 Perathoner
Perathoner
 Centi
Centi
 MacArio
MacArio
 Aloise
Aloise
 Giordano
Giordano