G Model
CATTOD-8870; No. of Pages7
ARTICLE IN PRESS
X. He, H. Liu / Catalysis Today xxx (2014) xxx–xxx
2
involving the oxidative dehydrogenation of ethanol to acetalde-
hyde and its subsequent acetalization with ethanol. For example,
Bueno et al. reported a 92% DEE selectivity at 30% ethanol conver-
sion (corresponding to a ∼28% DEE yield) in a batch mode using
at 398 K overnight and calcination at 573 K for 4 h in air flow.
Amberlyst 15 (Amberlyst 15-wet polystyrene-polydivinylbenzene
sulfonic resin, Alfa Aesar) was grounded into powder, and evacu-
ated at 353 K for 3 h before its use. H-Y zeolite with Si/Al ratio of
5.3 (Tianjin Nanhua) was calcined in air flow at temperatures of
773, 873, 973 and 1073 K for 4 h, which was labeled as H-Y-773,
H-Y-873, H-Y-973, and H-Y-1073, respectively. H-ZSM-5 zeolite
with Si/Al ratio of 25 (Tianjin Nanhua) was calcined in air flow at
773 K for 4 h, and labeled as H-ZSM-5(25).
homogeneous Pd(OAc) /Cu(OAc) /paratoluene sulfonic acid cata-
2
2
lysts at 343 K and 10 atm O2 [9]. Thavornprasert et al. obtained a
4% DEE yield on Mo12V W1.2Cu1.2Sb0.5Ox in gas-phase selective
1
3
oxidation of ethanol at 497 K [10]. However, the DEE yields from
this one-step route especially in a flow reactor are low because the
oxidative dehydrogenation of ethanol to the acetaldehyde interme-
diate on the existing catalysts requires higher temperatures, which
favor the desorption of acetaldehyde and as pervasively observed
in many selective oxidation reactions, its undesired over-oxidation,
relative to the acetalization step leading to DEE.
In this work, we study selective two-step ethanol conversion
to DEE in a high yield. The two steps include ethanol dehydro-
genation to acetaldehyde and its subsequent acetalization with
Iron molybdate catalyst with a Mo/Fe atomic ratio of 3, typically
for selective oxidation of methanol to formaldehyde, was prepared
by a coprecipitation method [23].
2.2. Catalyst characterization
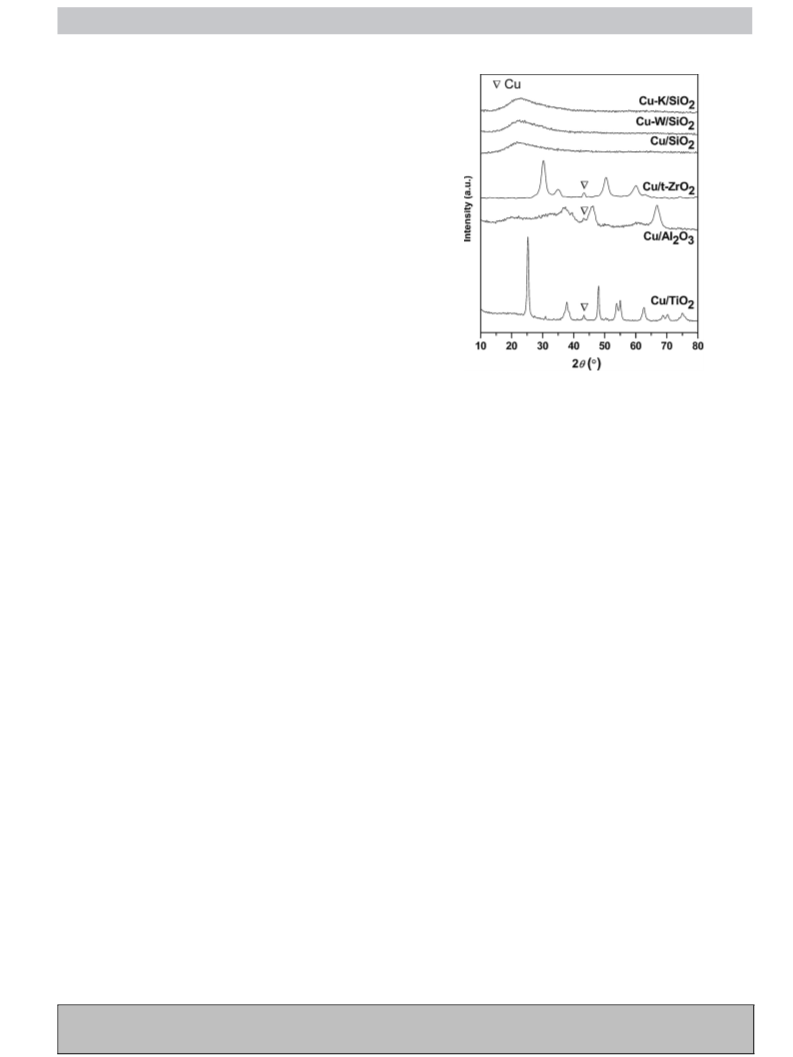
X-ray diffraction (XRD) patterns were obtained on a Rigaku
◦
D/Max-2000 diffractometer in the 2Â range 10–80 using Cu K␣
radiation (ꢀ = 1.5406 A˚ ). The applied voltage was 40 kV, and the
ethanol on supported copper (e.g. Cu/SiO ) and zeolite (e.g. H-Y)
2
catalysts, respectively, which are loaded in two sequential reactors
and operated at different temperatures. The dehydrogenation of
ethanol, instead of its selective oxidation, is employed to produce
acetaldehyde, which can avoid the inevitable formation of water
in the ethanol oxidation, and its inhibiting effect on the acetal-
ization of ethanol with acetaldehyde. Inspired by their excellent
performance in the dehydrogenation of alcohols [3,18], supported
copper catalysts are used for the dehydrogenation of ethanol. The
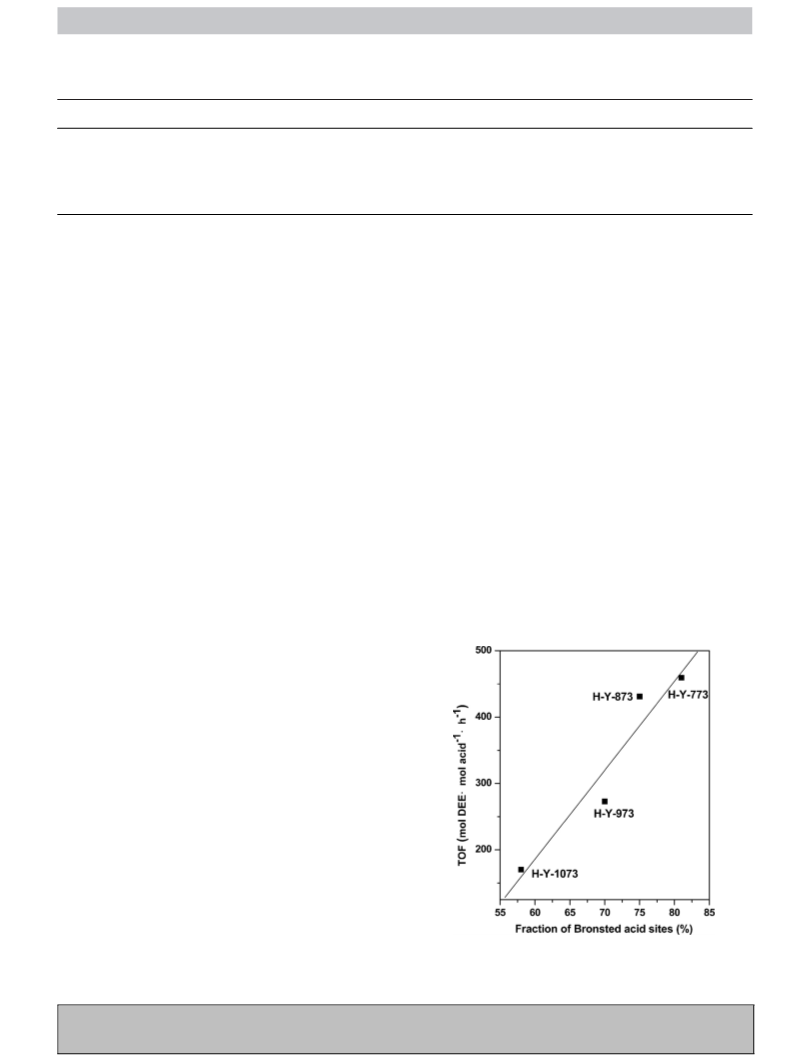
acetalization step is catalyzed by commercial zeolites (e.g. H-Y),
regarding their strong acidity, good stability, and large availability,
etc. [19]. This two-step route provides the convenience for opti-
mizing the temperatures of the dehydrogenation and acetalization
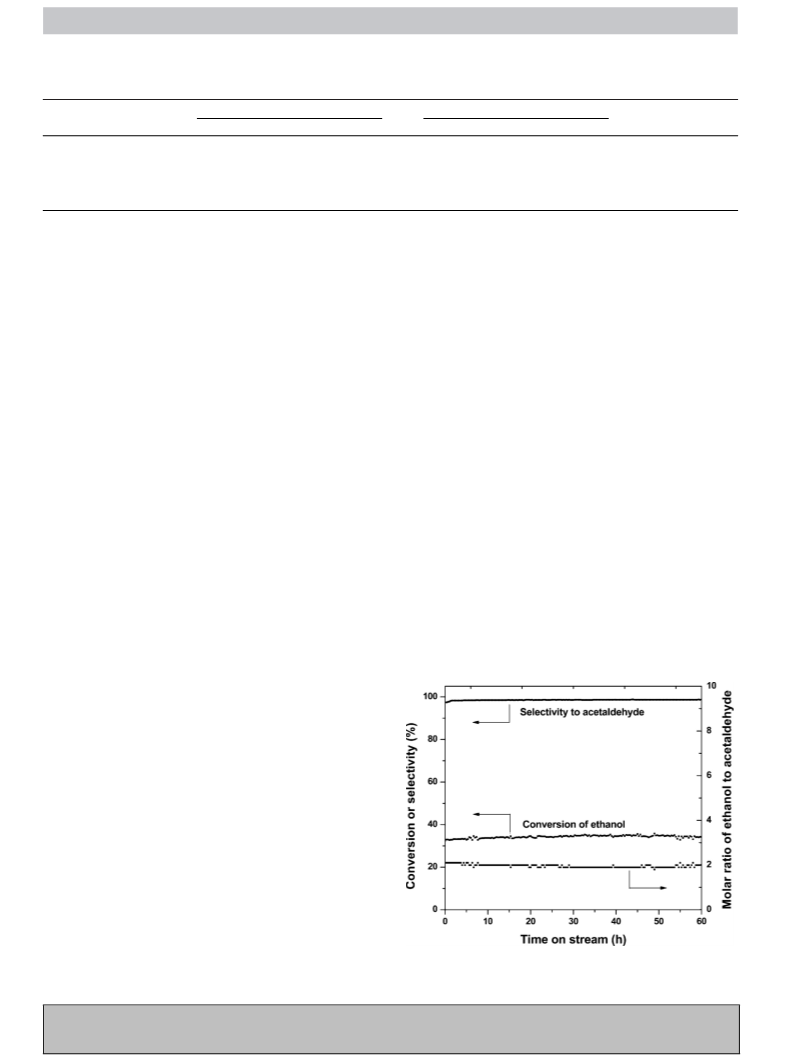
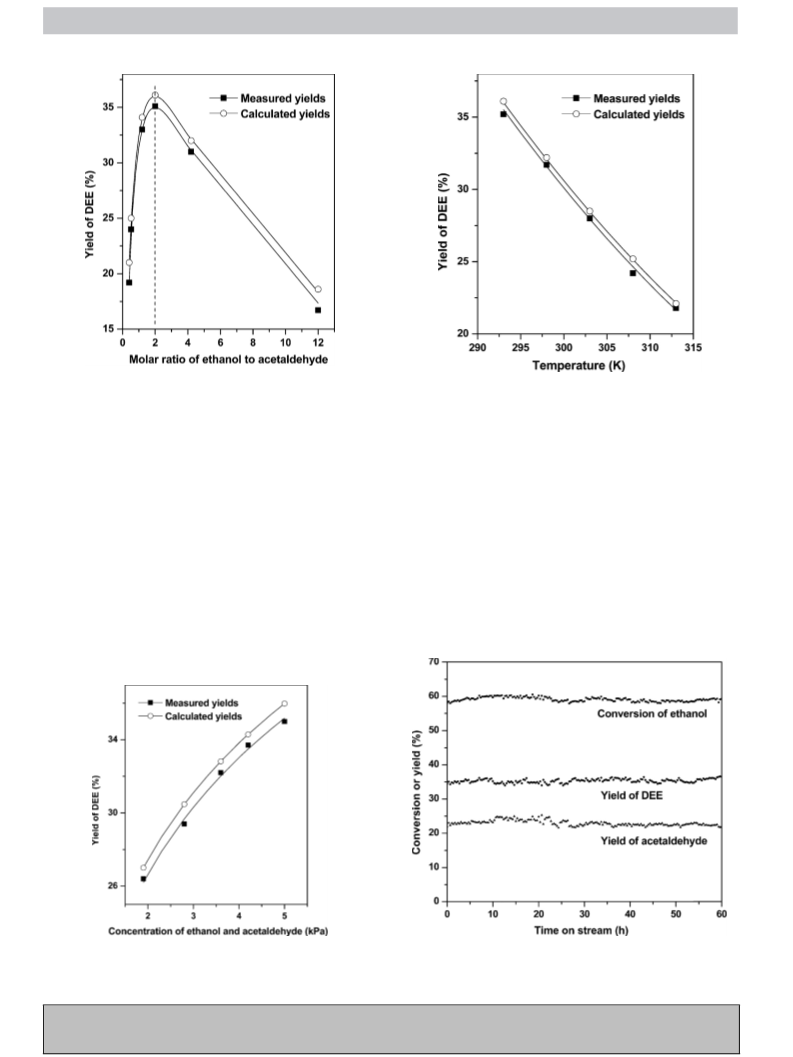
reactions, leading to a DEE yield of as high as 35.0%, analogous to
the estimated thermodynamic equilibrium yield (36.1%) at 293 K
under the conditions in this work.
current was 100 mA.
H2 temperature-programmed reduction (H -TPR) was carried
2
out on the TP5000 flow unit (Tianjin Xianquan). Catalysts contain-
ing approximately 1.5 mg of Cu were placed in a quartz cell, and
exposed to a 5% H /N (Beijing Huayuan, certified mixture) flow at
2
−
2
1
a rate of 40 mL min . The temperature was increased linearly from
03 to 823 K at a rate of 10 K min 1 (denoted as bulk TPR). The H2
consumption was monitored by a thermal conductivity detector
TCD), and the TCD signals were calibrated by the reduction of CuO
powder in the identical conditions.
Dissociative N O adsorption–H -TPR reverse titration was per-
−
3
(
2
2
formed on TP5000 to measure the Cu dispersion. The Cu catalysts
were first treated in a 20% H /N2 flow (30 mL min ) at 623 K for
−
1
2
2
h, cooled to 323 K in N2 (Beijing Haikeyuanchang, >99.99%) flow,
and then exposed to 5% N O/Ar (Beijing Huayuan, certified mixture)
2
−
1
flow (40 mL min ) for 0.5 h. Finally, the samples were flushed with
N2 and cooled to 303 K to start a H -TPR run (denoted as surface
TPR). The Cu dispersions of the catalysts were calculated by the Eq.
2
2
. Experimental
(1).
2.1. Catalyst preparation
Cu dispersion = 2
× H consumption (surface TPR)
× 100%
(1)
2
SiO -supported copper (Cu/SiO ) catalysts were prepared by
deposition–precipitation (DP) method [20]. Briefly, a calculated
amount of SiO2 was added to an aqueous solution of Cu(NO3)2
2
2
H consumption (bulk TPR)
2
Brunauer–Emmet–Teller (BET) surface areas and pore volumes
measurements were performed on a Quantachrome instrument
(Autosorb-1) at liquid nitrogen temperature. Before the mea-
(
Beijing Yili Fine Chemical, AR grade), and then the resulting gel
was stirred vigorously for 3 h. Afterwards, an aqueous solution of
KOH (Beijing Chemical, AR grade) was constantly dropped to the gel
under vigorous stirring until its pH value was above 10, followed
by aging at 363 K for 4 h. Finally, the slurry of the gel was filtered,
thoroughly washed with distilled water, dried at 393 K overnight,
and calcined at 723 K for 3 h in oxygen flow (Beijing Haikeyuan-
surement, Amberlyst 15 and AlCl /SiO2 (Sigma–Aldrich) were
3
evacuated at 353 K for 4 h because of their poor thermal stability.
The other samples were evacuated at 473 K for 4 h. Surface areas
were calculated based on the BET equation, and pore volumes were
determined by the single point adsorption total pore volume of
chang, >99.9%). Similarly, Al O , TiO and tetragonal ZrO2 (t-ZrO2)
pores at P/P = 0.99.
2
3
2
0
supported Cu catalysts were prepared. SiO , Al O and TiO2 were
NH temperature-programmed desorption (NH -TPD) and NH
2
2
3
3
3
3
supplied by Alfa Aesar, and t-ZrO was prepared by a hydrothermal
method reported by Li et al. [21].
pulse titration were carried out on TP5080 (Tianjin Xianquan).
For NH -TPD, the reduced Cu catalysts were exposed to a NH
2
3
3
−
1
A K-doped Cu/SiO2 (Cu-K/SiO ) catalyst with 5 wt% K was pre-
(Beijing Huayuan, 99.9%) flow (60 mL min ) at 373 K for 1 h, and
2
−
1
pared by incipient wetness impregnation of the calcined Cu/SiO2
sample (prepared by the DP method described above) with an aque-
ous solution of KNO3 at 293 K, followed by drying at 393 K and
then heated linearly from 373 to 973 K at a rate of 10 K min in
−
1
a He (Beijing Haipu, >99.99%) flow (30 mL min ). Desorbed NH3
was detected with TCD, and the TCD signals were calibrated by a
calcining at 723 K under the same conditions for Cu/SiO . In a simi-
given volume of NH . The zeolite catalysts, H-ZSM-5(25) and H-
2
3
lar way, a W-doped Cu/SiO (Cu-W/SiO ) catalyst with 2 wt% W was
Y with a Si/Al ratio of 25 and 5.3, respectively, were treated in a
2
2
prepared using an aqueous solution of ammonium paratungstate.
N2 flow at 773 K for 0.5 h, before their NH -TPD experiments were
3
Different types of acid catalysts were employed. SO42 /ZrO2
−
carried out, as described above. The acidity for other acid sam-
ples (SO42 /ZrO , H PW12O40/SiO , Amberlyst 15, AlCl /SiO , and
−
was prepared by calcination of Zr(SO ) ·4H O (Alfa Aesar, AR
4
2
2
2
3
2
3
2
grade) in air flow at 973 K for 3 h [22]. Supported heteropolyacid
SiO –Al O (Sigma–Aldrich)) was measured by NH pulse titration
2 2 3 3
H PW12O40/SiO2 with a 30 wt% loading was prepared by incip-
3
2
ient wetness impregnation of SiO2 with an aqueous solution of
H PW12O40 (Sinopharm Chemical, AR grade), followed by drying
3
following the aforementioned NH -TPD procedures.
3
Please cite this article in press as: X. He, H. Liu, Efficient synthesis of 1,1-diethoxyethane via sequential ethanol reactions on silica-

 He, Xiaohui
He, Xiaohui
 Liu, Haichao
Liu, Haichao