Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
Green Chemistry
Communication
catalytic processes.35–39 The oxidation coupling reaction of
ethanol into diethoxyethane (DEE) was also studied by
Yoshida et al.,40,41 using silica-supported Ta2O5 and Nb2O5
photocatalysts. However, the reaction did not occur without
the presence of O2 over the employed photocatalysts, and the
aerobic conditions made the reaction quite complicated,
leading to the formation of considerable amounts of by-pro-
ducts such as acetaldehyde, acetate acid, and ethene. The
photocatalytic anaerobic dehydrogenation (oxidation) of alco-
hols in aqueous solution has been extensively studied for the
purposes of H2 generation from water and alcohols,42–44 in
which alcohols serve as sacrificial active reagents or hydrogen
donors and are degraded to CO2. We were recently intrigued by
the idea of selectively converting the “sacrificed” alcohols syn-
chronously into valuable products during photohydrogen pro-
duction. We found that over TiO2 photocatalysts, ethanol
could undergo an acceptorless dehydrogenation C–C coupling
reaction in aqueous solution and selectively produce 2,3-
butanediol during the H2 liberation.45 In this water-mediated
process TiO2 photocatalysts with lower reactivities, such as
rutile TiO2, are favorable for selective coupling, while highly
reactive photocatalysts, such as commercial TiO2 (Degussa
P25), tend to over-oxidize ethanol into acetic acid and CO2.
Herein we report that in organic media or neat reagents,
primary alcohols (including bio-renewable ethanol and
butanol) can undergo the ADC reaction under UV irradiation
with the highly active P25-TiO2 photocatalyst, selectively produ-
cing acetals; aldehydes first form via hole-induced dehydro-
genation of alcohols and then condense in situ with unreacted
alcohols into acetals. Simultaneously, the formed H+ ions are
reduced by photo-generated electrons into H2, as shown in
Scheme 1.
At the start of our investigation, we performed dehydro-
genation of butanol in cyclohexane solution in an argon
atmosphere, using a commercial TiO2 photocatalyst (Degussa
P25) under UV irradiation at room temperature. Pure TiO2-P25
did not show any detectable reaction after a long irradiation
time (52 hours). When 1 wt% of platinum co-catalyst was
loaded onto the P25 surfaces (Pt-P25), hydrogen was immedi-
ately generated. Analyses of the liquid products by GC-MS
showed that butanol was successfully converted into the coup-
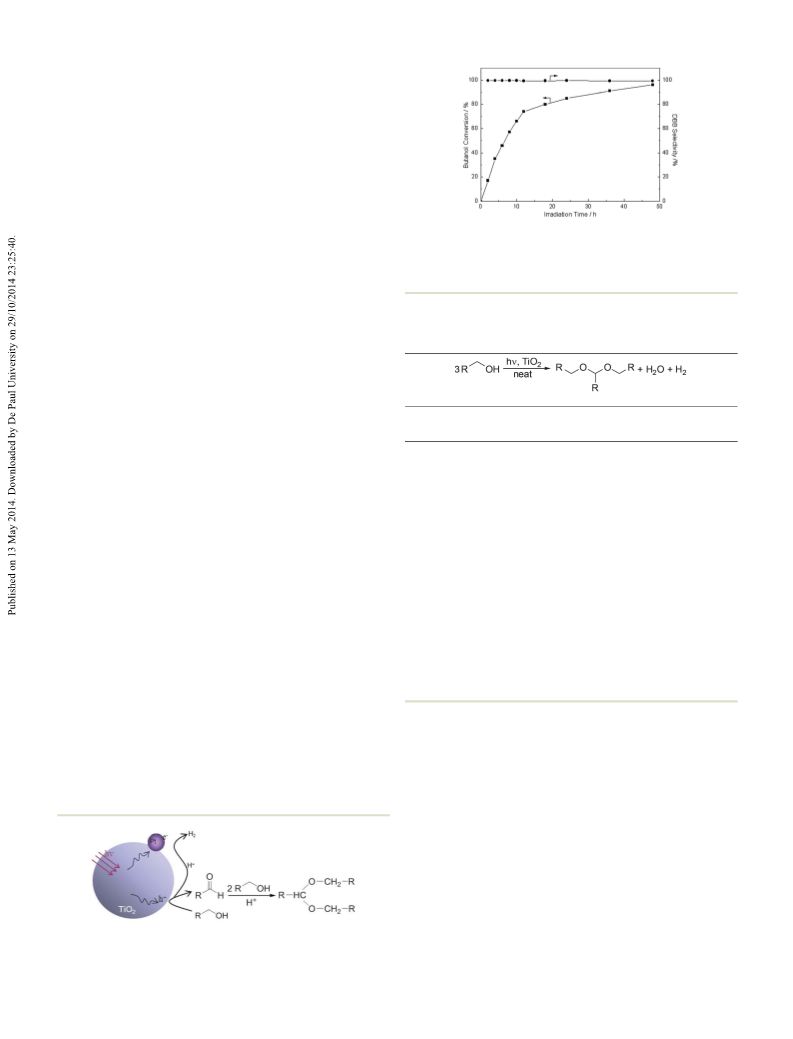
ling product, dibutoxybutane (DBB). The conversion of
butanol is more rapid initially, reaching 75% within 12 h, and
then it increases slowly to 96% after 50 h (Fig. 1). The reacted
butanol was nearly completely converted into DBB, with a
selectivity of 99.6%, although trace amounts of other products,
Fig. 1 Photocatalytic conversion of butanol into DBB in cyclohexane.
Conditions: butanol, 10 mmol; solution volume, 20 ml; catalyst, Pt-P25,
0.1 g with 1 wt% of platinum; atmosphere, argon; temperature, 20 °C;
light source, 300 W high-pressure Hg-lamp.
Table 1 Experimental data for the TiO2-photocatalytic ADC reaction
for the direct conversion of primary alcohols into acetalsa
Conv. Sel.
Rateb
Entry Alcohol
Catalyst
(%)
(%)
(mmol g−1 h−1
)
1
2
3
4
5
6
7
8
1-Butanol
1-Butanol
1-Butanol
1-Butanol
1-Butanol
1-Butanol
Ethanol
Pt-P25
22.2
2.5
0.9
99.2 40.4
Pt-rutile
Pt-Brookite
Pd-P25
Au-P25
Rh-P25
Pt-P25
Pt-P25
Pt-P25
Pt-P25
Pt-P25
99.5
99.5
4.6
1.6
21.3
16.1
8.6
27.7
24.8
17.9
12.1
5.1
99.3 38.8
99.5 29.3
99.6 15.7
99.3 79.2
99.5 55.3
99.3 27.4
97.2 16.2
1-Propanol
1-Pentanol
1-Hexanol
2-Propanolc
2-Butanolc
9
10
11
12
13
0
0
0
11.1
7.3
4.8
Pt-P25
4.0
3
Benzyl alcoholc Pt-P25
a Reaction conditions: alcohol, 20 ml; TiO2, 0.1 g; noble metal co-
catalyst, 1 wt%; atmosphere, argon; temperature, 20 °C; light source,
300 W high-pressure Hg-lamp; irradiation time, 12 h. b The rates were
calculated on the basis of the converted alcohols. c These alcohols are
only dehydrogenated into the corresponding ketones or aromatic
aldehydes, showing no coupling reaction.
such as 1-butyraldehyde, butyric acid, butyl butyrate, CO, and
CO2 were also detected. We additionally evaluated the reaction
using Pt-P25 over 12 h using neat butanol (butanol serves as
both a reactant and solvent). It showed that the reaction still
exhibited a very high selectivity for the formation of DBB
(99.2%) and a butanol conversion of 22. 2% (Table 1, entry 1).
Obviously, the reaction displays
a higher reaction rate
when using the neat alcohol, reaching 40.4 mmol g-1 h−1
(vs. 2.2 mmol g−1 h−1 in cyclohexane).
We also investigated the effect of the TiO2 structure on the
catalytic performance. The P25-TiO2 photocatalyst exhibits a
much higher activity than rutile and brookite TiO2, which is
likely to be due to its anatase–rutile-mixed phase structure that
facilitates the separation of electron–hole pairs.45–47 However,
the product selectivity does not seem to be dependent on the
Scheme 1 Schematic illustration of the TiO2-photocatalytic ADC reac-
tion for the direct conversion of primary alcohols into acetals.
This journal is © The Royal Society of Chemistry 2014
Green Chem., 2014, 16, 4076–4080 | 4077

 Zhang, Hongxia
Zhang, Hongxia
 Zhu, Zhenping
Zhu, Zhenping
 Wu, Yupeng
Wu, Yupeng
 Zhao, Tianjian
Zhao, Tianjian
 Li, Li
Li, Li