2
0
K. Barbera et al. / Journal of Catalysis 323 (2015) 19–32
We may remark that oxides in mesoporous silica may be pres-
Luo et al. [11] showed recently that under pressure (70 bar) in
HMF etherification with 2-propanol, catalysts with Lewis acidity
or weak Brønsted acidity are active in the transfer of hydrogen
from the alcohol to HMF to produce 2,5-bis(hydroxymethyl)furan
(in their case), with subsequent reactions to form mono- or die-
thers. However, at lower pressure and using a primary alcohol, this
reaction was negligible, thus reducing in part the complexity of the
reaction network.
ent in various forms, although their specific catalytic role was not
clearly identified. The oxide may be present as nanocrystallites
deposited on the external surfaces of crystallites, inside the chan-
nels, or forming monolayer-type species (mono- or polynuclear
species formed by reaction with surface silanol groups), which
may lead to the reconstruction of the inner walls of the mesopor-
ous channels (the so-called corona area [18,19]). Katryniok et al.
[
20] observed that by grafting zirconia onto SBA-15, a very high
Therefore, HMF etherification with ethanol is a useful model sys-
tem (besides its applicative interest) to analyze the role of the loca-
tion of the oxide in ZrO /SBA-15-based catalysts on the acid
2
properties and in turn the catalytic reactivity. However, understand-
ing the role of location also requires having good model catalysts
with different locations of the oxide, for which analysis may thus
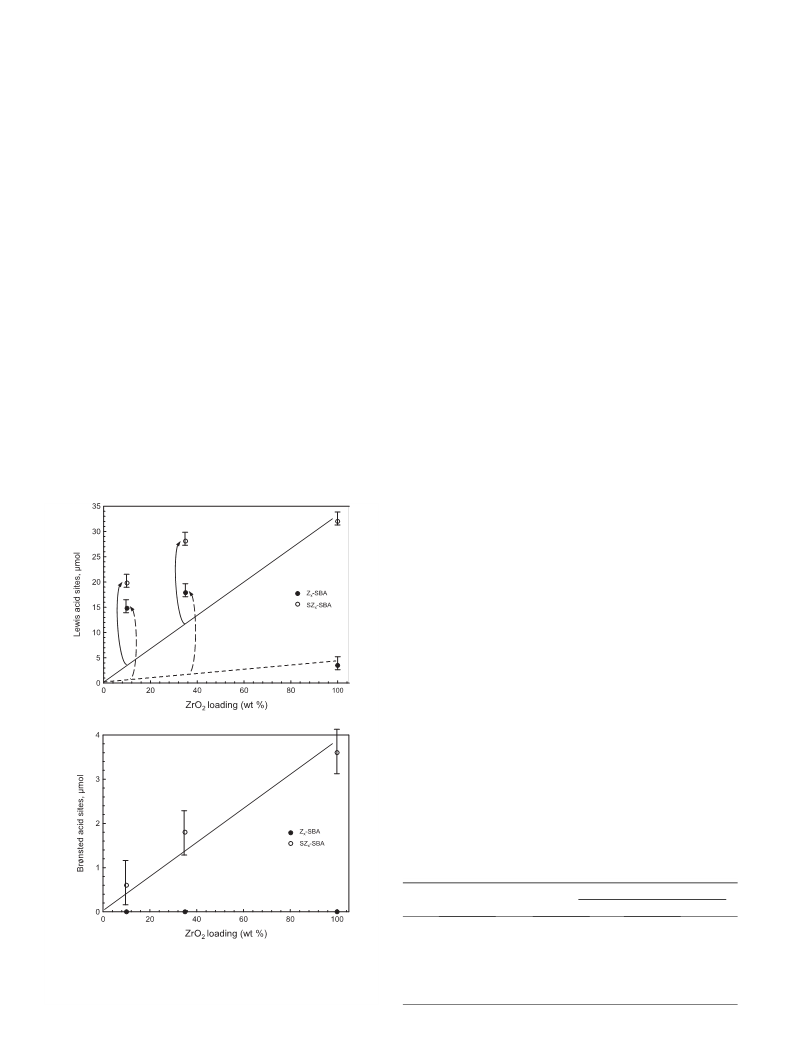
dispersion of zirconia within the mesoporous silica (indicated as
a nanocomposite) could be obtained, with a linear relationship
between the amount of introduced zirconia and the number of acid
sites. On the other hand, Schlögl and co-workers [21] observed that
upon introduction of molybdenum oxide species into the channels
of SBA-15, the limited availability of anchor silanol groups at high
provide insight into the above aspects. In preparing ZrO
catalysts, we observed (as detailed later) that a different location
of ZrO is observed depending on the initial loading, with preferen-
2
/SBA-15
4
loadings forced the MoO groups to form strained configurations,
giving enhanced reactivity and reasonably different acid-based
properties. A similar mechanism probably occurs for zirconia, sug-
gesting that a nonlinear correlation between the concentration of
zirconia species and acidity/reactivity is expected.
2
tial external or internal deposition of small nanoparticles at 10 and
35 wt%, respectively. The reasons for this effect are still under inves-
tigation (probably related to the gradients of concentration realized
during the preparation), but these two catalysts, before and after
sulfation of zirconia, and in comparison with pure SBA-15 and
ZrO (before and after sulfation) samples, were good model systems
2
to understand the effect of oxide location in oxide/mesoporous sys-
tems in the acid characteristics and in turn the catalytic behavior.
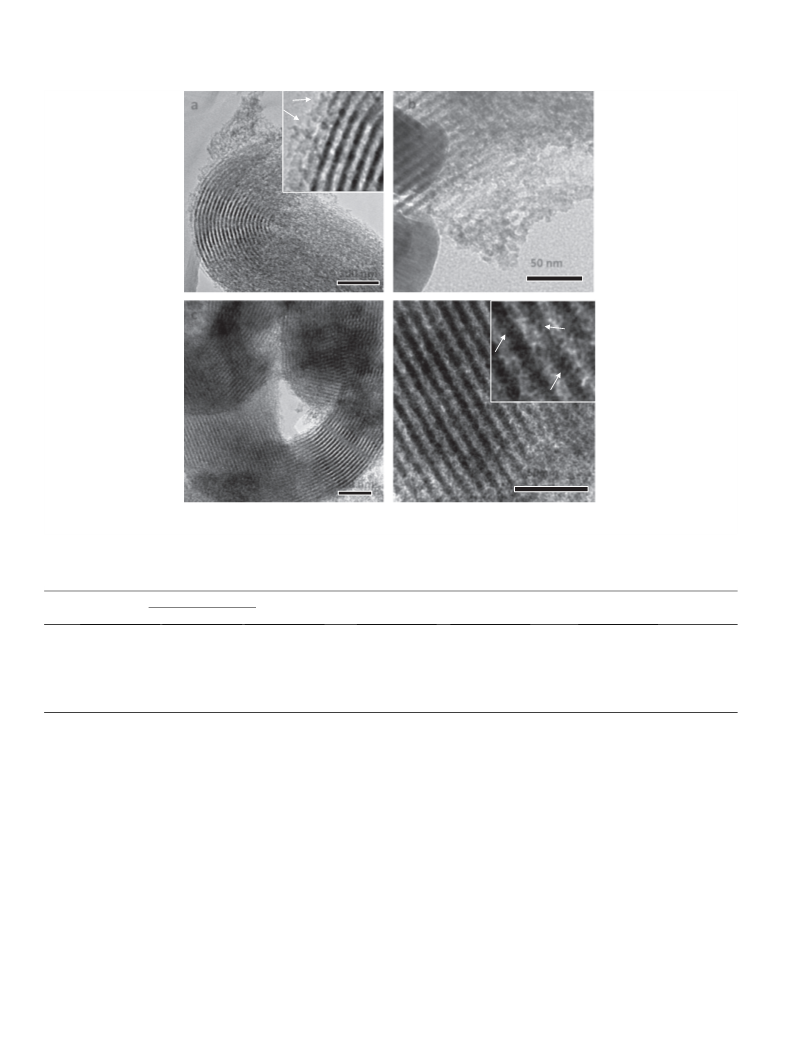
Ballem et al. [22] noted that zirconia nanoparticles inside SBA-
5 have small dimensions (ꢀ4 nm) and are faceted with 110 sur-
1
faces termination, suggesting enhanced reactivity and acidity with
respect to nanoparticles deposited on the external surface. Wang
et al. [23] observed that the acid strength of sulfated ZrO
2
/MCM-
4
1 (with monolayer coverage) is lower than that of bulk sulfated
zirconia, in contrast with the other indications discussed above
and for example with what was found by Gao et al. [24] in the
esterification of oleic acid.
There are thus contradictory results on the influence of the loca-
tion of oxide species in SBA-15 on their properties and reactivity.
Intuitively, small oxide nanoparticles deposited on the external sur-
face of a porous support could be preferable, because (i) the inter-
action with silanol groups is reduced (most are present inside the
channels [25,26]) and (ii) the accessibility of bulky molecules is
higher. Although in theory the channel diameter in SBA-15 (around
2
. Experimental
2.1. Catalyst preparation
Mesoporous SBA-15 was prepared as a support according to the
synthesis procedure in the literature and the detailed procedure
described elsewhere [34]. Different ZrO loadings (theoretical 10
and 35 wt%) were dispersed on the SBA-15 support by a urea
hydrolysis method using zirconium(IV) oxychloride (ZrOCl O)
ꢁ8H
as a zirconia source [35]. The mixture was refluxed at 90 °C for 5 h
pH about 8), and the resulting gel was filtered and washed with
distilled water until removal of chloride. After centrifugation, the
ZrO -SBA-15 gel was dried and calcined at 550 °C for 6 h. The sam-
ples are indicated hereafter as Zx%-SBA.
Pure ZrO was prepared by the sol–gel method. A portion of
0 ml of zirconium n-propoxide was mixed with 26.6 ml of n-pro-
2
2
2
6
–9 nm, depending on the preparation) avoids the presence of dif-
fusional problems, the length of channels (typically in the range
00–600 nm) may create issues of full access to active sites in the
(
3
2
inner part of the channels [27,28]. Similarly, the deposition of the
oxide may not occur in the whole channels, but only at or close to
the entrances of the mesopores [29]. Landau et al. [30] remarked
2
2
2 2
how TiO and ZrO can be inserted inside the pores of SBA-15 in dif-
panol and stirred with a magnetic stir bar. Then 2.8 ml of water
was added dropwise to carry out the hydrolysis and gelation of zir-
conium n-propoxide. The gel was aged for 1 h at room temperature
and then placed in an oil bath at 75 °C to remove alcohol. The solid
was then dried at 120 °C for 12 h and calcined at 550 °C for 12 h
ferent locations depending on the preparation. Janssen et al. [31]
observed that small zirconia particles (2–3 nm) inside the mesop-
ores of SBA-15 are not distributed uniformly in all the channels.
The objective of this paper is thus to understand better how the
2
location of zirconia in ZrO /SBA-15 catalysts influences the acidity
[
36]. This reference material is indicated hereafter as Z.
and catalytic activity in reactions of biomass conversion sensitive
to acidity characteristics and how the sulfation of these samples
would influence these properties. As a reaction highly sensitive
to the acid characteristics of the catalysts (type and strength of
the acid sites) in terms of different transformation pathways, we
have selected 5-hydroxymethyl-2-furfural (HMF) etherification
with ethanol. A reason for interest in this reaction, in addition to
its being a model reaction to analyze the acid characteristics of
the catalyst as discussed below, is that the products obtained are
of commercial interest as high-cetane-number biodiesel additives
All samples were further sulfated using H SO 1 N (15 ml/g) at
2
4
room temperature, dried and calcined at 550 °C for 3 h. These cat-
alysts are indicated hereafter as SZx%-SBA and SZ.
2.2. Catalyst characterization
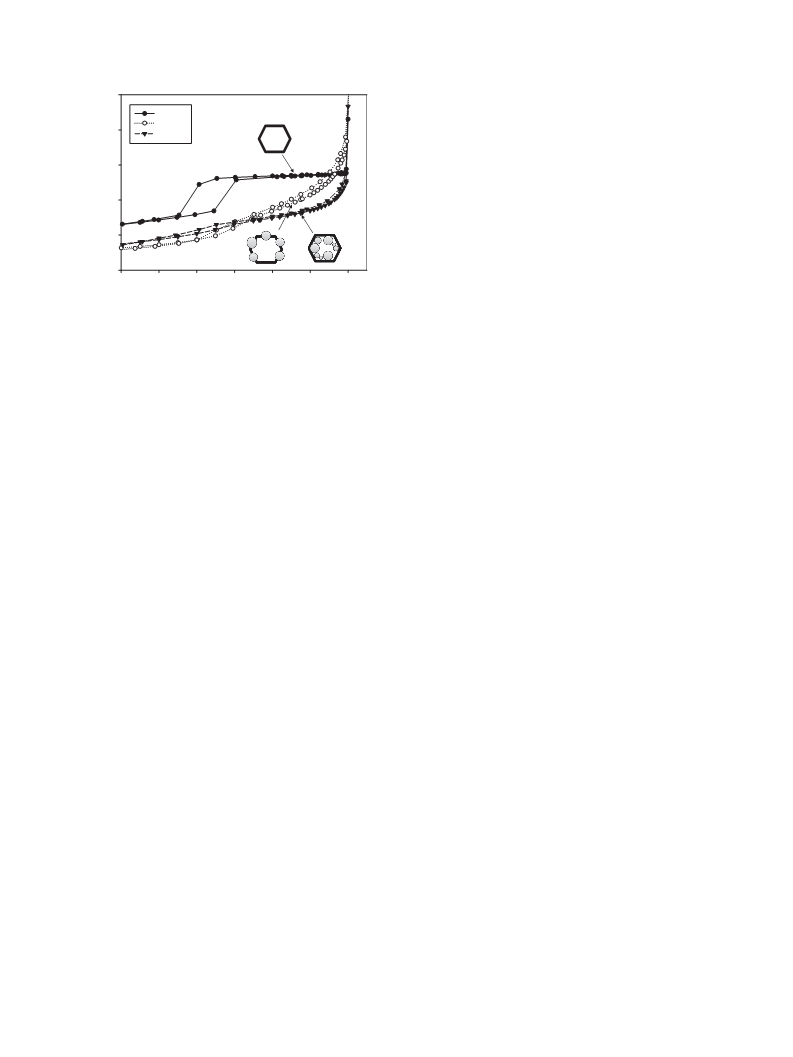
Small angle X-ray diffraction was performed using a Philips
PW1729 diffractometer with Bragg–Brentano geometry h–2h and
Cu K
were collected in the range of 2h from 0.5 to 5° with step interval of
0.02°. The d100 value was calculated from Bragg’s law, while a for
a radiation and a zero background quartz holder. The spectra
[32,33]. Three main types of products could be detected [12]: 5-
(ethoxymethyl)furan-2-carbaldehyde (EMF), 1,1-diethoxyethane
(DE), and ethyl 4-oxopentanoate (EOP) (Scheme 1).
0
the hexagonal unit cell was calculated using the equation
p
The selectivity to EMF and EOP was related to the presence of
a
0
= (2d100)/ 3.
Lewis and/or Brønsted acidity on the catalyst, while the formation
of DE depended on defect sites, which, being less reactive, cata-
lyzed the side reaction of co-reactant ethanol to DE only when
strong Lewis and/or Brønsted acid sites were absent.
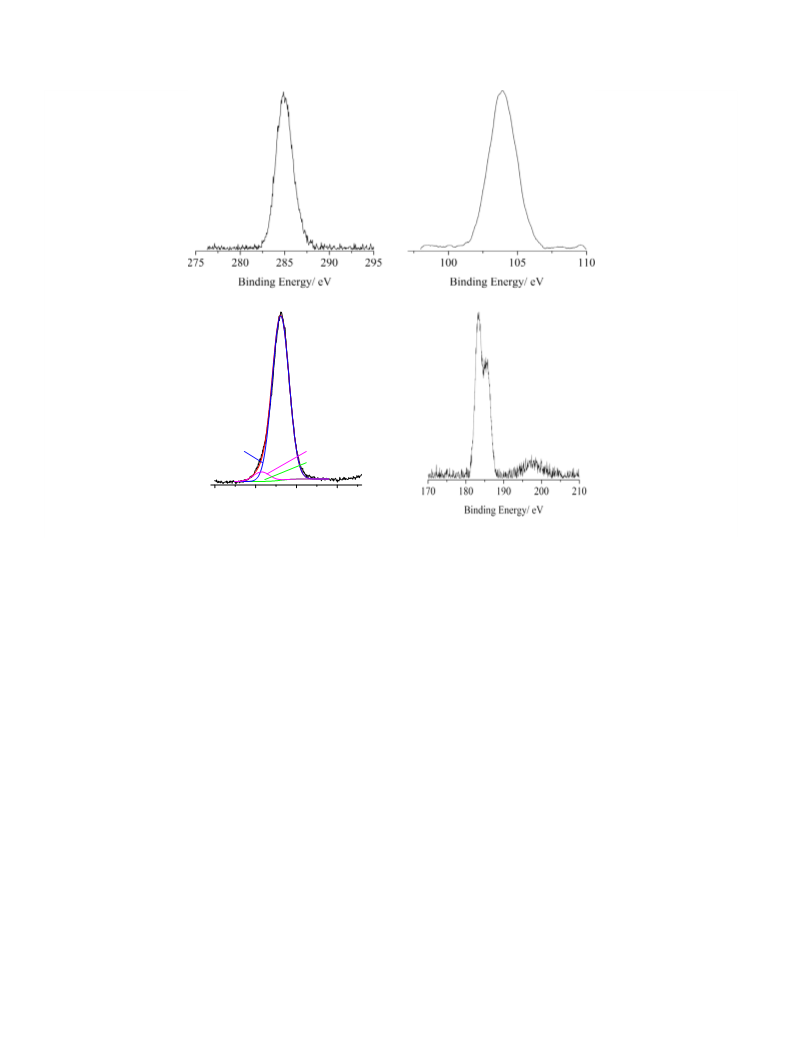
X-ray photoelectron spectra (XPS) were measured at 45° takeoff
angle relative to the surface plane with a PHI 5600 Multi Technique
System that offers good control of the photoelectron takeoff angle
ꢃ10
(base pressure of the main chamber 2 ꢂ 10
Torr) [37,38].

 Barbera
Barbera
 Lanzafame
Lanzafame
 Pistone
Pistone
 Millesi
Millesi
 Malandrino
Malandrino
 Gulino
Gulino
 Perathoner
Perathoner
 Centi
Centi