Please do not adjust margins
ChemComm
Page 4 of 5
DOI: 10.1039/C6CC09050H
COMMUNICATION
Journal Name
3. M. Nielsen, H. Junge, A. Kammer and M. Beller, Angew. Chem.
Int. Ed., 2012, 51, 5711 -5713.
4. D. Spasyuk, S. Smith and D. G. Gusev, Angew. Chem. Int. Ed.,
2012, 51, 2772-2775.
5. G. E. Dobereiner and R. H. Crabtree, Chem. Rev., 2010, 110, 681-
703.
6. J. Zhang, E. Balaraman, G. Leitus and D. Milstein,
Organometallics, 2011, 30, 5716-5724.
7. C. Gunanathan and D. Milstein, Accounts of Chemical Research,
2011, 44, 588-602.
8. W. Kuriyama, Y. Ino, O. Ogata, N. Sayo and T. Saito, Advanced
Synthesis & Catalysis, 2010, 352, 92-96.
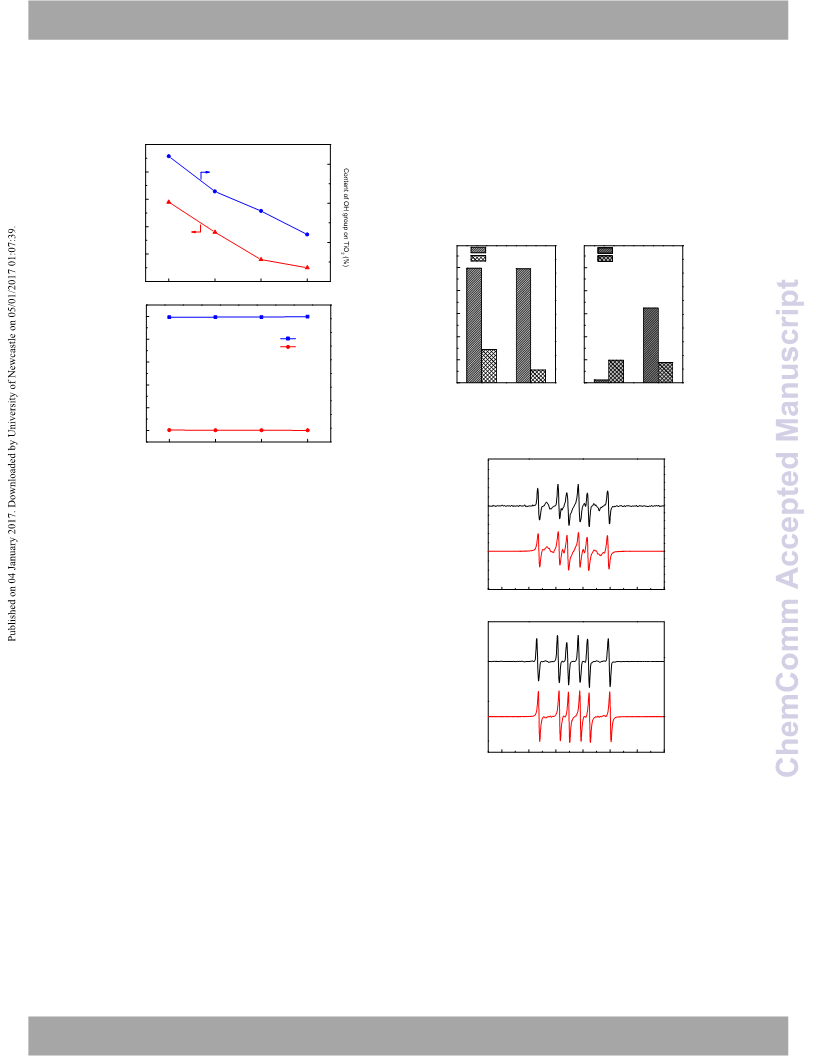
perform photocatalytic reaction in ethanol-D2O system. The
released hydrogen was detected by GC-MS technique. The
results showed that the most released hydrogen is D2 from
D2O with 62.0%, and only 17.4% H2 from ethanol (Figure 4a).
And then, we performed reaction in CD3CD2OD-H2O system.
91.7% H2 from H2O was detected, while only 4% D2 from
CD3CD2OD was detected (Figure 4b). The deuterium-labelling
experiments results revealed that H2O provided most
hydrogen proton to produce hydrogen. Meanwhile, it means
that ˙OH radical in photocatalytic cyclic process mostly
originated from H2O.
9. N. D. Schley, G. E. Dobereiner and R. H. Crabtree,
Organometallics, 2011, 30, 4174-4179.
10. C. Chen, Y. Zhang and S. H. Hong, The Journal of organic
chemistry, 2011, 76, 10005-10010.
11. K. Imamura, H. Tsukahara, K. Hamamichi, N. Seto, K. Hashimoto
and H. Kominami, Applied Catalysis A: General, 2013, 450, 28-
33.
12. H. Zhang, Y. Wu, L. Li and Z. Zhu, ChemSusChem, 2015, 8, 1226-
1231.
13. H. Zhang, Z. Zhu, Y. Wu, T. Zhao and L. Li, Green Chem., 2014,
16, 4076-4080.
14. W. Feng, G. Wu, L. Li and N. Guan, Green Chemistry, 2011, 13,
3265-3272.
15. Q. Wang, M. Zhang, C. Chen, W. Ma and J. Zhao, Angew. Chem.
Int. Ed., 2010, 49, 7976-7979.
Because ˙OH radical was directly part of the reaction, the
strong oxidation of ˙OH resulted in low selectivity of 2,3-BD
(2.6%). After OH group was replaced by -F, the amount of ˙OH
decrease from 32.5% to 20.8%, which lessened the over-
oxidation of ˙CH(OH)CH3 and resulted in increase of selectivity
from 2.6% to 65%. Similarly, R-TiO2 with less OH group also
exhibited higher selectivity of 2,3-BD. Thus, in ethano-H2O,
selectivity of 2,3-BD is preferentially controlled by the amount
of OH group. The similar conversion of ethanol in both Pt/P25-
TiO2 and Pt/F-P25-TiO2 case hinted that the transformation of
ethanol into ˙CH(OH)CH3 was a rate-determining step. The
more OH group was not beneficial to the formation of
˙CH(OH)CH3. On the contrary, the more OH group caused over-
oxidation of ˙CH(OH)CH3 and resulted in low selectivity.
16. J. Schneider, M. Matsuoka, M. Takeuchi, J. Zhang, Y. Horiuchi,
M. Anpo and D. W. Bahnemann, Chem. Rev., 2014, 114, 9919-
9986.
17. T. Mallat and A. Baiker, Chem. Rev., 2004, 104, 3037-3058.
18. R. A. Sheldon, I. W. C. E. Arends, G.-J. T. Brink and A. Dijksman,
Acc.Chem. Res., 2002, 35, 774-781.
19. R. A. Sheldon, Chem. Soc. Rev., 2012, 41, 1437-1451.
20. G.-J. t. Brink, I. W. C. E. Arends and R. A. Sheldon, Science, 2000,
287, 1636-1639.
21. D. I. Enache, J. K. Edwards, P. Landon, B. Solsona-Espriu, A. F.
Carley, A. A. Herzing, M. Watanabe, C. J. Kiely, D. W. Knight and
G. J. Hutchings, Science, 2006, 311, 362-365.
22. V. Augugliaro, M. Bellardita, V. Loddo, G. Palmisano, L.
Palmisano and S. Yurdakal, Journal of Photochemistry and
Photobiology C: Photochemistry Reviews, 2012, 13, 224-245.
23. G. Palmisano, E. Garcia-Lopez, G. Marci, V. Loddo, S. Yurdakal,
V. Augugliaro and L. Palmisano, Chem. Commun., 2010, 46,
7074-7089.
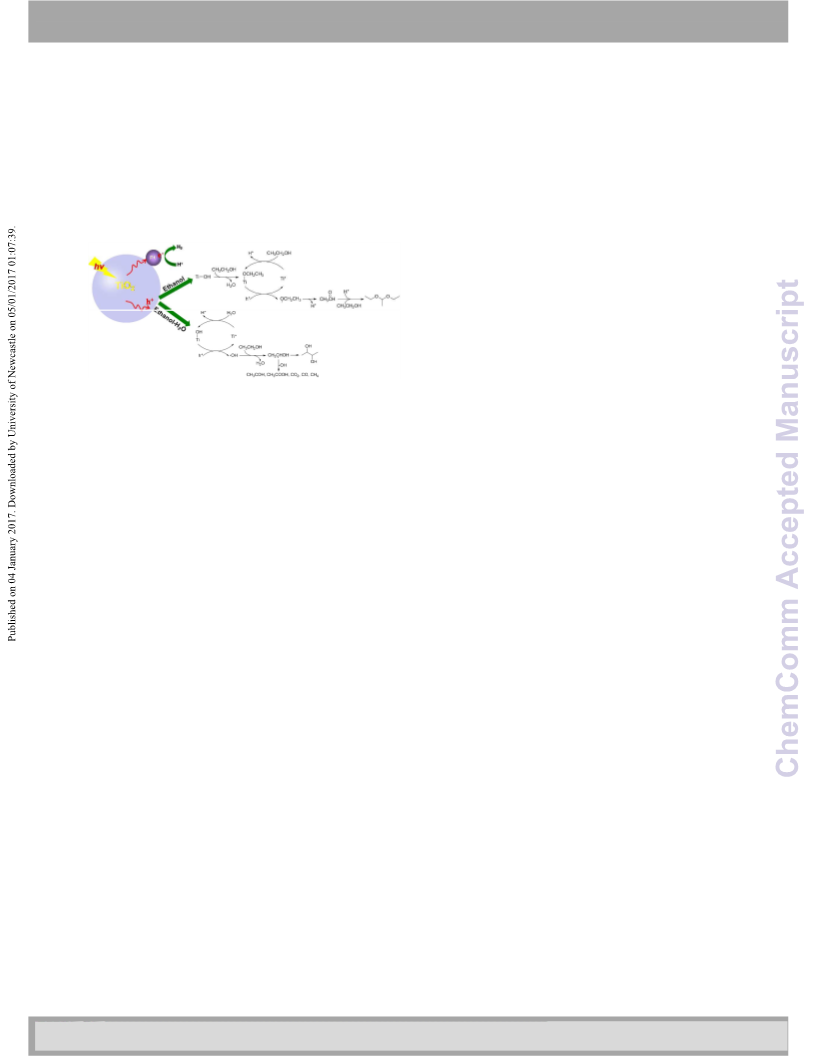
In summary, the study demonstrates how TiO2 surface
hydroxyl dominates the reaction process of the photocatalytic
C-O coupling of ethanol in neat ethanol and C-C coupling of
ethanol in aqueous. In neat ethanol, more hydroxyl group
results in higher activity, but because hydroxyl is not involved
in catalytic cyclic process, DEE exhibits high selectivity. Here,
hydroxyl plays an indirect site-holding role. In ethanol-H2O, the
strong oxidant ˙OH radical is involved in the photocatalytic
process, and less OH group can lead to higher selectivity of 2,3-
BD. The finding gives fundamental insight into the role of TiO2
surface hydroxyl during photocatalytic dehydrogenation
process of alcohols, which explain the role of a surface
function group of catalyst. The exchange between
reactant/solvent and surface group of catalyst is vital in the
activity and selectivity of photocatalytic reactions. The
proposed mechanism not only explains all experiment
phenomena, but also provides
a new direction for
24. M. Zhang, Q. Wang, C. Chen, L. Zang, W. Ma and J. Zhao,
Angew. Chem. Int. Ed., 2009, 48, 6081-6084.
25. P. Yang, J. Zhao, B. Cao, L. Li, Z. Wang, X. Tian, S. Jia and Z. Zhu,
ChemCatChem, 2015, 7, 2384-2390.
photocatalytic reaction design and opens a promising path to
obtain high selectivity and conversion in TiO2-based
photocatalysis process.
26. Q. Xiang, J. Yu and P. K. Wong, J. Colloid Interface Sci., 2011,
357, 163-167.
27. S. Stoll and A. Schweiger, J. Magn. Reson., 2006, 178, 42-55.
28. D. Dvoranova, Z. Barbierikova and V. Brezova, Molecules, 2014,
19, 17279-17304.
29. A. Molinari, M. Montoncello, H. Rezala and A. Maldotti,
Photochem. Photobiol. Sci., 2009, 8, 613-619.
30. V. Brezová, P. Tarábek, D. Dvoranová, A. Staško and S. Biskupic,
Journal of Photochemistry and Photobiology A: Chemistry, 2003,
155, 179-198.
This work was supported by the National Natural Science
Foundation of China (91545116 and 21573137), Natural
Science Foundation of Shanxi (2014011014-3) and the
Foundation of State Key Laboratory of Coal Conversion (J13-
14-913).
References
1. C. Gunanathan and D. Milstein, Science, 2013, 341, 1-11.
2. C. Gunanathan, L. J. W. Shimon and D. Milstein, J. Am. Chem.
Soc., 2009, 131, 3146–3147.
31. N. Hykaway, W. M. Sears, H. Morisaki and S. R. Morrison, J.
Phys. Chem., 1986, 90, 6663-6667.
4 | Chem. Commun., 2016, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Zhang, Hongxia
Zhang, Hongxia
 Zhang, Wenqin
Zhang, Wenqin
 Zhao, Min
Zhao, Min
 Yang, Pengju
Yang, Pengju
 Zhu, Zhenping
Zhu, Zhenping