
New Journal of Chemistry p. 10003 - 10011 (2019)
Update date:2022-08-30
Topics:
 Paleti, Gidyonu
Paleti, Gidyonu
 Peddinti, Nagaiah
Peddinti, Nagaiah
 Gajula, Naveen
Gajula, Naveen
 Kadharabenchi, Vasikerappa
Kadharabenchi, Vasikerappa
 Rao, Kamaraju Seetha Rama
Rao, Kamaraju Seetha Rama
 Burri, David Raju
Burri, David Raju
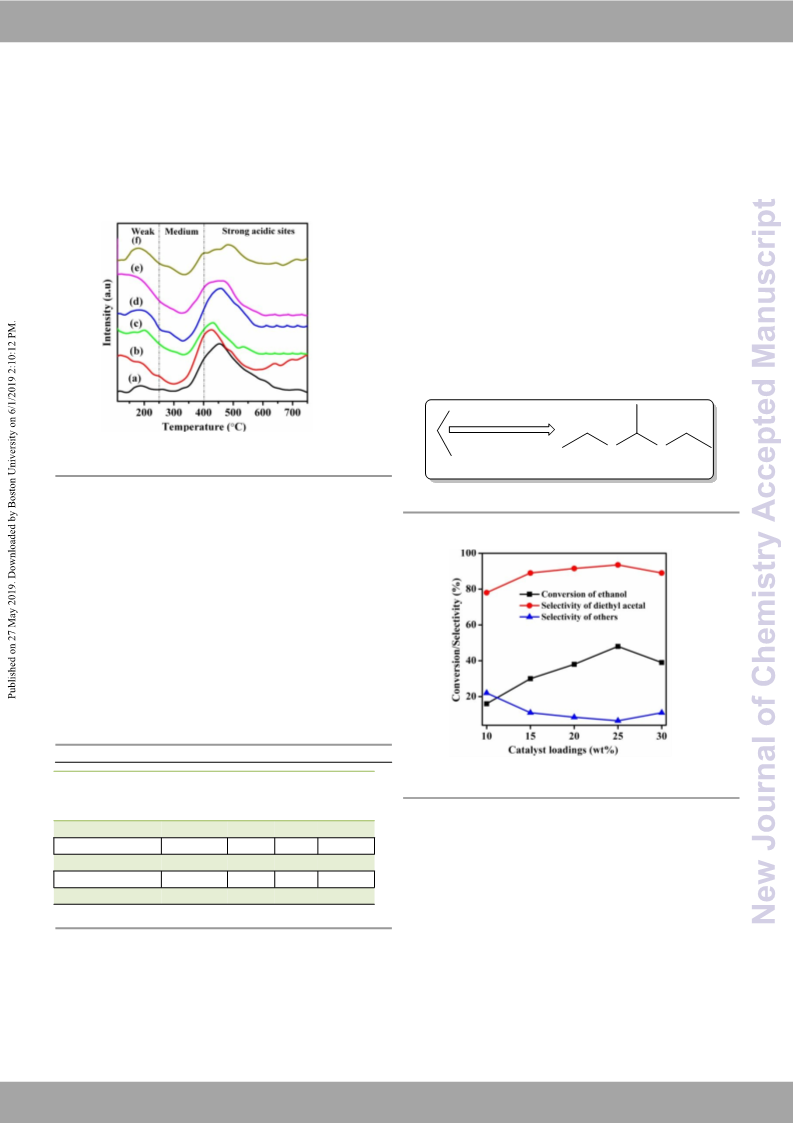
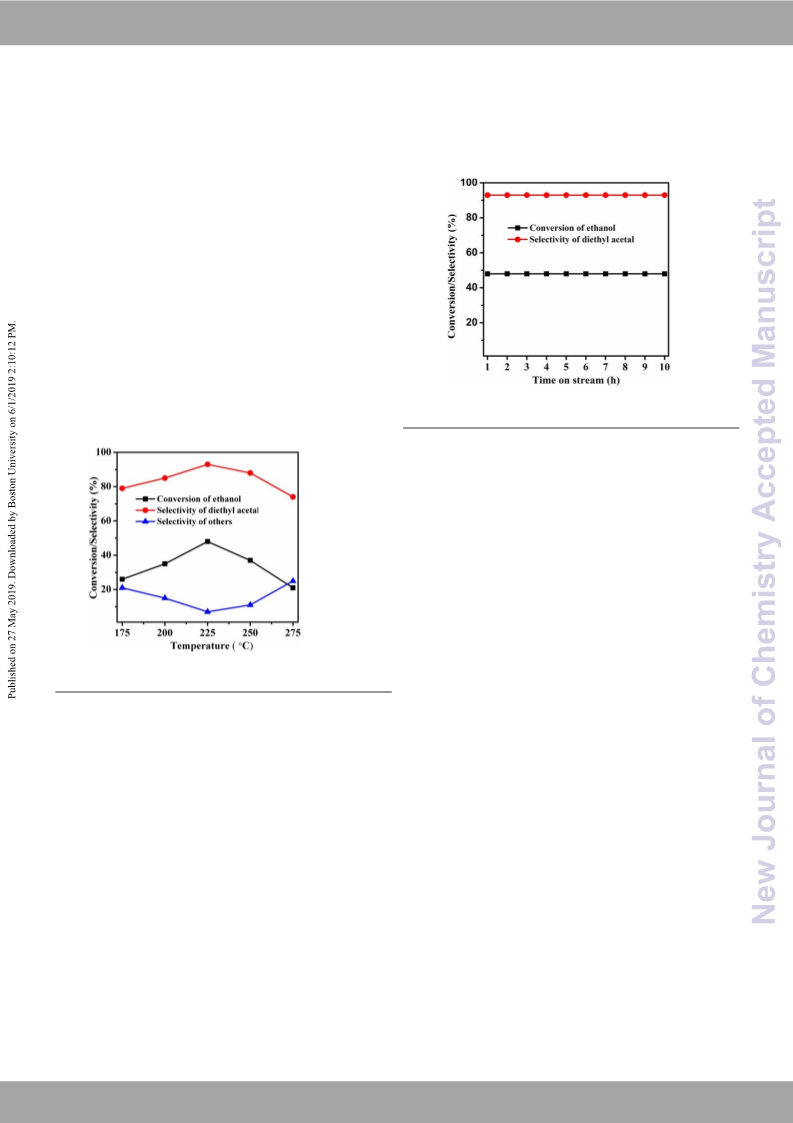
Of the biomass valorization technologies, bioethanol production and its selective conversion to diethyl acetal is of utmost importance to meet the increasing demand for bio-fuel additives. Direct synthesis of diethyl acetal in a single step from bioethanol using SBA-15-supported copper nanoparticle (CuNP/SBA-15) catalysts without using hydrogen acceptors and high pressure has been achieved as an alternative to the age-old two-step process via (i) partial oxidation of ethanol to acetaldehyde and (ii) acetaldehyde acetalization with ethanol. The prepared CuNP/SBA-15 catalysts with the SBA-15 support and copper nitrate (Cu (NO3)2·H2O) were characterized by using N2 adsorption, SEM, TEM, XRD, FT-IR, TPD of NH3, H2-TPR and N2O pulse chemisorption techniques. Analysis of the results showed the physico-chemical characterestics of these catalysts responsible for the acceptorless partial oxidation of ethanol to acetaldehyde and its acetalization with ethanol to yield diethyl acetal with high selectivity.
View More







Guangxi Nanning Guangtai Agriculture Chemical Co.,Ltd
Contact:+86-771-2311266
Address:Room703,Building12, Software Park Phase II,NO.68,Keyuan Road,Nanning City,Guangxi,China
Contact:+86-20-62802632;62802633
Address:Room 330 GIGCAS Building,No.511 Kehua Street,Tianhe District
Contact:86-931-8272767
Address:Room 602, No.461, Nanchang Road, Chengguan District, Lanzhou City, China PRC
Suzhou Kangrun Pharmaceutical, Inc
Contact:86-512-63912376,63913329
Address:Building 2, No. 2358 ,Chang'an Rd, Wujiang Economic Development Zone Pioneering park, china
Jewim Pharmaceutical (Shandong) Co., Ltd
Contact:+8615621883869
Address:山东省泰安市高新技术产业开发区配天门大街西首
Doi:10.1246/cl.1986.299
(1986)Doi:10.1007/BF00574817
(1984)Doi:10.1246/cl.2008.1230
(2008)Doi:10.1039/b404614e
(2004)Doi:10.1021/ja00094a020
(1994)Doi:10.1126/science.141.3585.1039
(1963)