A. Li, D. Li, J. Mao et al.
Polyhedron 200 (2021) 115140
that obtained with Pt/CdS photocatalyst [14]. A F-doping of Co3O4
films catalyst was prepared by chemical vapor deposition and dis-
played remarkable photocatalytic activity from ethanol in the
near-ultraviolet region, which is 5 times hydrogen yield higher
than undoped Co3O4 [15]. The noble-metal-free Co(OH)2/CdS nano-
wires catalyst exhibited excellent hydrogen generation rate from
water splitting under visible light irradiation, which is 206 times
higher than the CdS NWs [16].
mechanism of the composite was proposed through the study of
the optical properties.
2. Experimental
2.1. General procedures
All chemicals used were of reagent grade, purchased from Alad-
din or Beijing Chemical Company. The actual chemical composi-
tions were measured by Inductively Coupled Optical Emission
Spectrometer (ICP-OES) on a PerkinElmer Optima 8000. The size
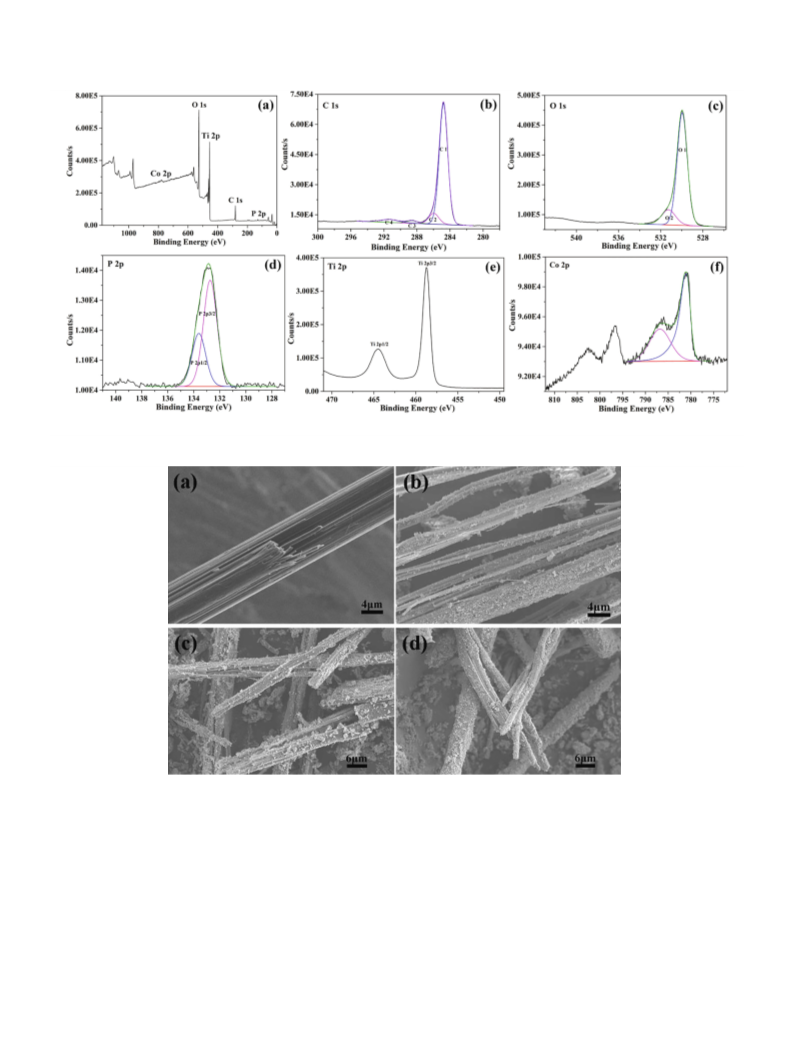
and morphology of the composites were investigated using a Hita-
chi S-3400N microscope scanning electron microscope (SEM).
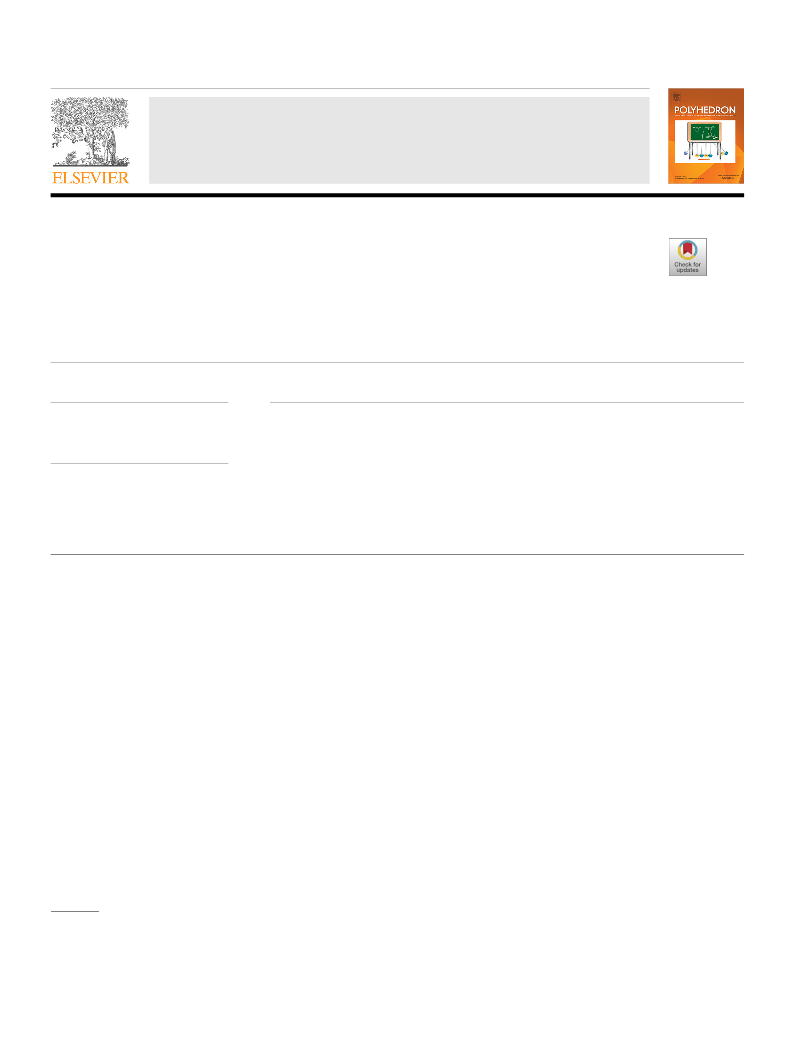
Chemical state analysis and relative atomic ratio were carried out
by X-ray photoelectron spectroscopy (XPS) in an AXIS Supra X-ray
photoelectron spectrometer. The detailed spectra of C1s, O1s, P2p,
Ti2p and Co2p were recorded in the following conditions. Based on
a Shirley-type background [23], the raw spectra were fitted using
nonlinear least-squares fitting program adopting Gaussian-Lorent-
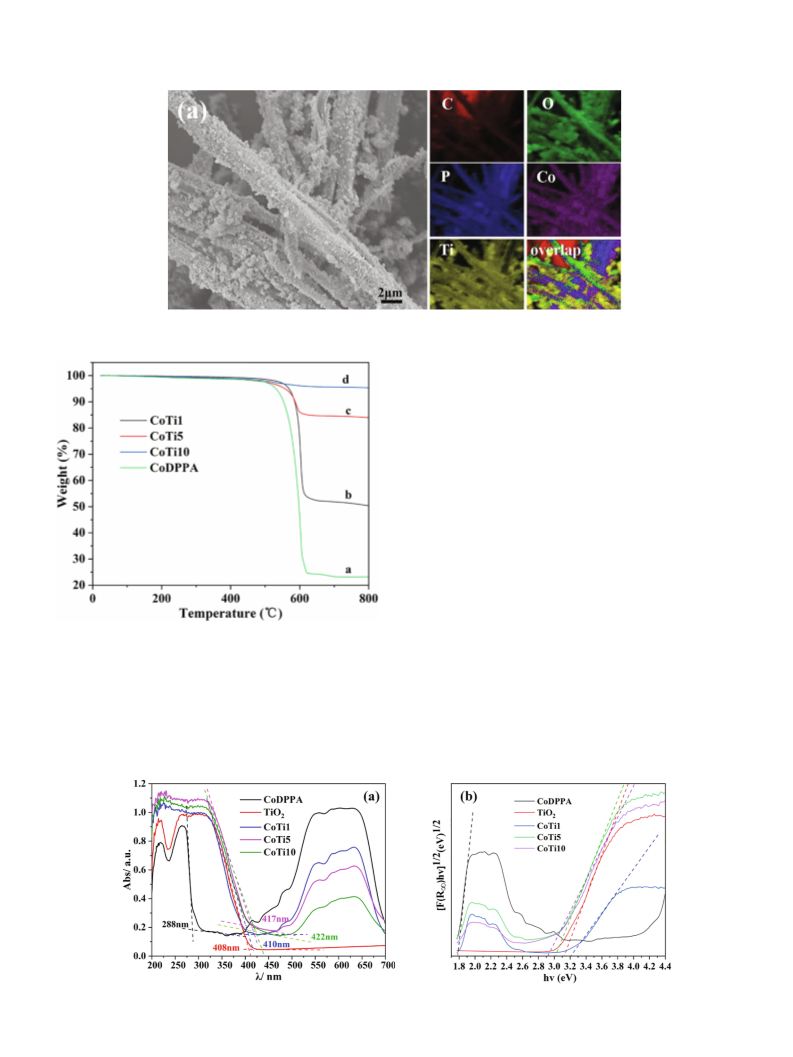
zian peak shapes for all the peaks. Thermogravimetric analysis (TG)
was performed on a TA Q500 apparatus between 20 and 800 °C
with a heating rate of 10 K/min under N2 atmosphere. The powder
XRD patterns were obtained on a Rigaku Ultima IV X-ray diffrac-
Hydrogen can be produced from diverse sources such as fossil
fuels, oils, alcohols, water and biomass [17]. Organic compounds
of methane and methanol as a substrate for H2 generation by
reforming reactions have been industrialized. However, as one of
the most important bio-alcohols, ethanol is considered as a poten-
tial substrate for hydrogen evolution in terms of its renewability
and lower toxicity. Various ethanol conversion technologies such
as steam reforming, partial oxidation, autothermal reforming, alka-
line-enhanced reforming, dehydrogenation, supercritical water
gasification, photocatalysis and electrocatalysis have been widely
used in H2 production from ethanol [17]. Among the above men-
tioned technologies, because of its low cost and mild experimental
conditions, the photocatalysis technology has great attractive for
the ethanol conversion to H2 generation. There are limited litera-
tures available on exploring pure ethanol as raw material conver-
sion to H2 evolution. Through a facile photodeposition method, a
new type Pt/TiO2 nanotube photocatalyst have been developed
for photocatalytic hydrogen production from ethanol system by
Lin and co-worker [18]. The research found that the preparation
procedure of noble metal Pt loading catalyst has a significant effect
on activity of hydrogen generation, the liquid phase product distri-
bution and the catalyst stability. The nanosheet photocatalyst Pt/
TiO2 was prepared by Xu etc., which demonstrated excellent cat-
alytic activity for simultaneous H2 production and selective oxida-
tion of ethanol under the irradiation of sunlight [19].
Moreover, as one of the most important bio-alcohols, ethanol
can be used to produce various chemicals through the oxidation
process. Among them, the 1,1-diethoxyethane has attracted atten-
tion because of its special quality and extensive application. It
could be used as raw material in organic synthesis and pharmaceu-
tical industry [20]. From the environmental point of view, adding
the 1,1-diethoxyethane to fuel can greatly reduce the emissions
of particulate and nitrogen oxides. Generally, the two steps is
needed for conversion of ethanol to 1,1-diethoxyethane. First the
ethanol is oxidized to acetaldehyde, then the ethanol reacts with
acetaldehyde to produce 1,1-diethoxyethane under the catalysis
of acid. This indirect synthetic process is complexity and the oxi-
dants are unfriendly to the environment. Therefore, the develop-
ment of simple and green synthetic process is essential. Zhang’s
group has developed a new synthetic route. The noble-metal-
loaded TiO2 was synthesized by photodeposition method, with
remarkable photocatalytic performance for the direct conversion
of ethanol to 1,1-diethoxyethane under illumination of ultraviolet
radiation [21].
tometer in transmission mode (flat sample holders, Cu-K
a radia-
tion) equipped with the D/tex Ultra detector (resolution 0.0001°
in 2h). The accelerating voltage and current were 40 kV and
40 mA, respectively. Infrared spectroscopy (IR) was measured on
a Thermo Scientific Nicolet iS10 at the range of 400–4000 cmÀ1
.
Elemental analysis (EA) was obtained by a Vario EL-III analysis
apparatus. The UV–vis diffuse reflectance spectra (DRS) was per-
formed on a PE Lambda 365 spectrophotometer, which was
equipped with an integrating sphere and a standard white board
was used as a reference. The photoluminescence (PL) spectra were
measured by FLS980 Spectrometer with a xenon arc lamp (Xe900)
and the entrance slit and the exit slit are both 4 nm.
2.2. Preparation of cobalt(II) diphenylphosphinate/TiO2
The composite of CoDPPA/TiO2 was prepared in following steps.
Cobalt(II) chloride hexahydrate (0.0476 g, 0.2 mmol),
diphenylphosphinic acid (denoted as DPPA) (0.0873 g, 0.4 mmol)
were dissolved in 7 mL ethanol. Then a different amount of com-
mercial P25 (TiO2), with molar ratio of Co/Ti = 1/X (X = 1, 5, 10)
was added to the solution, respectively. After ultrasonic and stir-
ring, the mixture was transferred into a 15 mL Teflon lined stain-
less steel autoclave. The autoclave was sealed, maintained at
180 °C for 3 days, and cooled to room temperature naturally. The
products were collected by filtration, washed with ethanol and
dried in air. The prepared samples with the molar ratio Co/Ti of
1, 0.2 and 0.1 were labeled as CoTi1, CoTi5 and CoTi10, respec-
tively. The EA results of the corresponding samples are as follows.
Calcd. for CoTi1: C 50.29, H 3.52; found: C 44.65, H 3.11. Calcd. for
CoTi5: C 32.19, H 2.26; found: C 16.08, H 1.13. Calcd. for CoTi10: C
22.31, H 1.56; found: C 8.09, H 0.60. The actual concentrations of
Co and Ti in the composites were given by ICP-OES, the ratio of
Co/Ti is basically the same as that of C and H to Ti.
In our previous reported work, the fibrous coordination poly-
mer cobalt(II) diphenylphosphinate (denoted as CoDPPA) was pre-
pared and the compound has excellent photocatalytic activity for
H2 production from ethanol solution, and ethyl acetate was pro-
duced in the same catalytic process [22]. For developing the cat-
alytic performance of CoDPPA further, based on the band gap
characteristics and optical properties of CoDPPA and TiO2, we
designed and synthesized the CoDPPA/TiO2 composite. The three
novel CoDPPA/TiO2 composites were prepared by the solvothermal
and characterized through various methods. By the studies of
hydrogen generation activity in ethanol solution, the composites
exhibited the higher photocatalytic activity for H2 production com-
pared with CoDPPA or TiO2. Meanwhile, the side catalytic product
1,1-diethoxyethane was obtained. The possible photocatalytic
2.3. Photocatalytic H2 evolution
The photocatalytic hydrogen evolution test was carried out in a
closed system of evacuation and gas circulation. A 300 W Xenon
lamp was used as light source. In detail, 50 mg CoDPPA/TiO2 com-
posite was dispersed in ethanol solution (100 mL). The suspension
solution was sealed in a quartz vessel and purged with N2 for
30 min to drive away the residual oxygen. The temperature of reac-
2

 Li, Aihong
Li, Aihong
 Li, Dongyang
Li, Dongyang
 Mao, Jianwei
Mao, Jianwei
 Ge, Zhimeng
Ge, Zhimeng
 Guo, Jianping
Guo, Jianping
 Liu, Bo
Liu, Bo