P.E. Hoggard, A. Maldotti / Journal of Catalysis 275 (2010) 243–249
249
Table 9
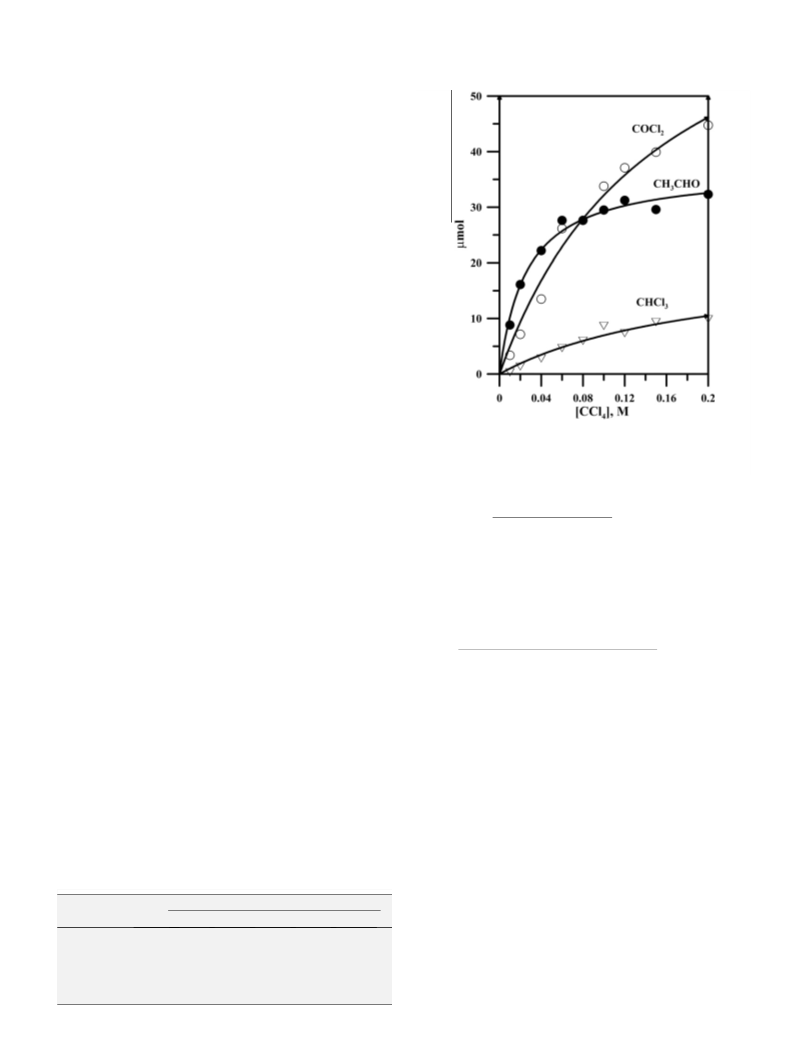
phosgene. Given this mechanism, the yield of phosgene should
never exceed that of acetaldehyde; however, phosgene was found
Products (in lmol) from 30-min irradiation (k > 350 nm)
of 1 mL of 5% EtOH in CCl
4
with 41 mg of Amberlite IRA-
ꢀ
in excess when the concentration of CCl
4
was above 5%. Thus, there
9
00 (Cl form).
appears to be an additional reaction channel, which might involve
the hydrolysis of CCl .
4
Total acetaldehyde
Total phosgene
32.7
18.0
0.7
1.0
0.4
CHCl
Ethyl acetate
Cl
HCl
3
References
C
2
6
3.8
[
[
1] X. Shen, J. Lind, T.E. Eriksen, G. Merenyi, J. Phys. Chem. 93 (1989) 553–557.
2] M.C. Gonzalez, G.C. Le Roux, J.A. Rosso, A.M. Braun, Chemosphere 69 (2007)
238–1244.
1
[
3] C. Bartocci, A. Maldotti, G. Varani, P. Battioni, V. Carassiti, D. Mansuy, Inorg.
Chem. 30 (1991) 1255–1259.
peaks, but only about 10–15% of the total peak area found with
ethanol and 1-propanol, and likewise approximately 10% of the
HCl. Moving to 1-pentanol caused another large reduction in yield,
the total peak area and HCl yield being 1–2% of that found in eth-
anol and 1-propanol.
Based on the decrease in yield with the increase in hydropho-
bicity of the alcohol, we suggest that more polar alcohols solvate
4
the ionic sites in the resin, allowing dissolved CCl access to the
portion of the poly(styrene-co-divinylbenzene) resin in the vicinity
of chloride ions. Less polar solvents, including neat carbon tetra-
chloride, may be unable to approach the anions closely enough
to allow the chloride ion to stabilize the chlorine atom as it disso-
[4] C. Bartocci, A. Maldotti, G. Varani, V. Carassiti, P. Battioni, D. Mansuy, J. Chem.
Soc. Chem. Commun. (1989) 964–965.
[
[
5] G. Varani, A. Maldotti, C. Bartocci, New J. Chem. 16 (1992) 827–831.
6] A. Maldotti, C. Bartocci, R. Amadelli, V. Carassiti, J. Chem. Soc., Dalton Trans.
(1989) 1197–1201.
7] T. Hirose, Y. Maeno, Y. Himeda, J. Mol. Catal. A: Chem. 193 (2003) 27–32.
8] J. Li, W.H. Ma, Y.P. Huang, X. Tao, J.C. Zhao, Y.M. Xu, Appl. Catal. B 48 (2004) 17–
[
[
2
4.
[9] A. Maldotti, G. Varani, A. Molinari, Photochem. Photobiol. Sci. 5 (2006) 993–
95.
10] A. Molinari, G. Varani, E. Polo, S. Vaccari, A. Maldotti, J. Mol. Catal. A: Chem. 262
2007) 156–163.
9
[
(
[11] S.L. Buell, J.N. Demas, J. Phys. Chem. 87 (1983) 4675–4681.
[12] E.Y. Komarova, K.T. Ren, D.C. Neckers, J. Phys. Chem. B 107 (2003) 5675–5679.
[
[
[
13] A.E.C. Redpath, O. Pekcan, M.A. Winnik, J. Photochem. 23 (1983) 283–288.
com/catalog/ProductDetail.do?N4=216585|ALDRICH&N5=SEARCH_CONCAT_
PNO|BRAND_KEY&F=SPEC>, (accessed 12.05.10.
ciates from the CCl
4
molecule.
3.11. Conclusions
[
[
16] A. Stone, E.B. Fleischer, J. Am. Chem. Soc. 90 (1968) 2735–2748.
17] H. Du, R.-C.A. Fuh, J. Li, L.A. Corkan, J.S. Lindsey, Photochem. Photobiol. 68
(1998) 141–142.
Amberlite IRA-900 anion exchange resin in the chloride form
has been shown to be an efficient catalyst for the photodecompo-
sition of CCl /EtOH solutions, yielding acetaldehyde, phosgene, and
chloroform. This may be viewed as a potential means to degrade
unwanted carbon tetrachloride, but also for its potential to gener-
ate useful products, diethyl carbonate, for example. From a syn-
[
4
(
accessed 29.08.2010).
[19] P. Kubelka, J. Opt. Soc. Am. 38 (1948) 448–457.
[
[
20] D.N. Kevill, M.J. D’Souza, J. Org. Chem. 63 (1998) 2120–2124.
21] A.L. Sidel’kovskii, S.I. Orlov, L. Margolin, N. Vasil’ev, A.L. Chimishkyan, Org.
React. (Tartu) 24 (1987) 207–217.
thetic viewpoint, the photodecomposition of CCl
could be used to generate phosgene in situ, given the abundance
of reactions in which COCl is an important building block [25]
4
in ethanol
[22] N.S. Roh, B.C. Dunn, E.M. Eyring, R.J. Pugmire, H.L.C. Meuzelaar, Fuel Process.
Technol. 83 (2003) 27–38.
2
[
23] N.P. Cheremisinoff, Handbook of Industrial Toxicology and Hazardous
Materials, Marcel Dekker, New York, 1999.
but the general aversion to storing highly toxic chemicals.
Optimum efficiency of photodecomposition is achieved with a
partial pressure of O around half of atmospheric, under which
2
[24] G.G. Esposito, D. Lillian, G.E. Podolak, R.M. Tuggle, Anal. Chem. 49 (1977)
1774–1778.
[
25] J.-P. Senet, Comptes Rendus de l’ Academie des Sciences Serie IIc: Chimie 3
2000) 505–516.
conditions a decomposition rate over ten times that reported for
homogeneous catalysis by iron porphyrins results [3]. Less resin
is required to intercept the incident light if it is ground to a pow-
der, but the bead form offers advantages in sample handling.
The experimental evidence is fairly strong that the catalysis by
the chloride form of Amberlite IRA-900 of the photodecomposition
of carbon tetrachloride in ethanol may be ascribed to the absorp-
tion of light by the poly(styrene-co-divinylbenzene) matrix, energy
(
[
26] M.S. Kharasch, O. Reinmuth, W.H. Urry, J. Am. Chem. Soc. 69 (1947) 1105–
1110.
[
[
[
27] M.S. Kharasch, E.V. Jensen, W.H. Urry, J. Am. Chem. Soc. 69 (1947) 1100–1105.
28] M.W. Schmidt, E.K.C. Lee, J. Am. Chem. Soc. 92 (1970) 3579–3586.
29] X. Zhang, J. Wu, Y. Zhou, Radiat. Phys. Chem. 43 (1994) 335–338.
[30] V. Catoire, R. Lesclaux, W.F. Schneider, T.J. Wallington, J. Phys. Chem. 100
1996) 14356–14371.
(
[
[
31] R. Lesclaux, A.M. Dognon, F. Caralp, J. Photochem. Photobiol., A 41 (1987) 1–11.
32] E.M. Glebov, V.F. Plyusnin, V.P. Grivin, Y.V. Ivanov, N.V. Tkachenko, H.
Lemmetyinen, J. Photochem. Photobiol., A 113 (1998) 103–112.
transfer to CCl
4
, and its subsequent homolysis, assisted by chloride
[
33] R. Michalski, A. Sikora, J. Adamus, A. Marcinek, J. Phys. Chem. A 114 (2010)
ions, which stabilize the chlorine atoms and hinder recombination.
861–866.
There is also evidence that ethanol facilitates the transport of CCl
molecules to resin sites near chloride ions.
4
[
34] V. Nagarajan, R.W. Fessenden, J. Phys. Chem. 89 (1985) 2330–2335.
[35] X.Y. Yu, J. Phys. Chem. Ref. Data 33 (2004) 747–763.
[
[
[
36] B. Ruscic, J. Berkowitz, J. Chem. Phys. 101 (1994) 10936–10946.
37] R. Battino, Solubility Data Series: Oxygen and Ozone, Pergamon, Oxford, 1981.
38] A. Buchachenko, Russ. Chem. Rev. 54 (1985) 117–128.
To some extent, the data are also consistent with a partitioning
of the CCl radicals formed by photodissociation into two reaction
channels, one in which hydrogen is abstracted from ethanol and
the other in which reaction occurs with molecular oxygen to form
the trichloromethylperoxy radical, which goes on to produce
3
[39] G.C. Bond, R.C. Francisco, E.L. Short, Appl. Catal. A 329 (2007) 46–57.
[40] W. Minder, H. Knuchel, P. Gurtner, Experientia 4 (1948) 219–220.
[
41] H.F. Ryan, Aust. J. Chem. 21 (1968) 2933–2943.

 Hoggard, Patrick E.
Hoggard, Patrick E.
 Maldotti, Andrea
Maldotti, Andrea