
Journal of the American Chemical Society p. 10822 - 10827 (2021)
Update date:2022-08-16
Topics:
 Chao, Yuguang
Chao, Yuguang
 Gu, Lin
Gu, Lin
 Guo, Shaojun
Guo, Shaojun
 Li, Hongbo
Li, Hongbo
 Li, Menggang
Li, Menggang
 Lu, Shiyu
Lu, Shiyu
 Lv, Fan
Lv, Fan
 Tao, Lu
Tao, Lu
 Yin, Kun
Yin, Kun
 Zhang, Qinghua
Zhang, Qinghua
 Zhang, Weiyu
Zhang, Weiyu
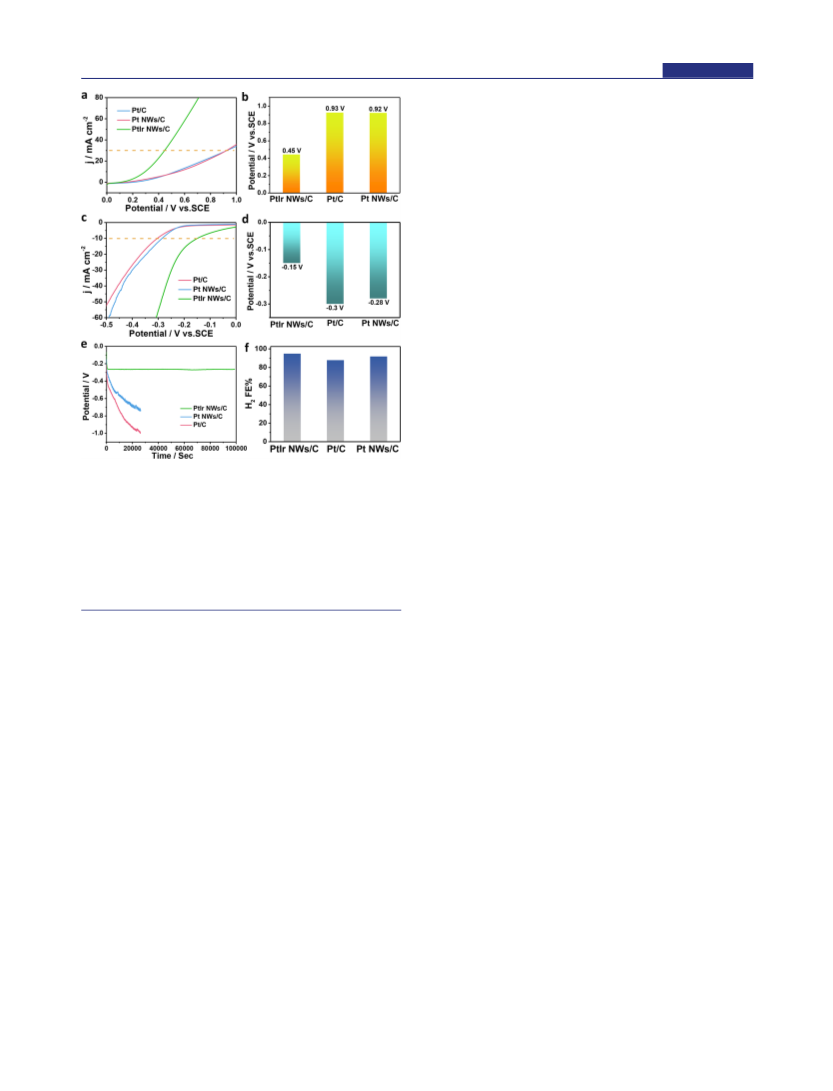
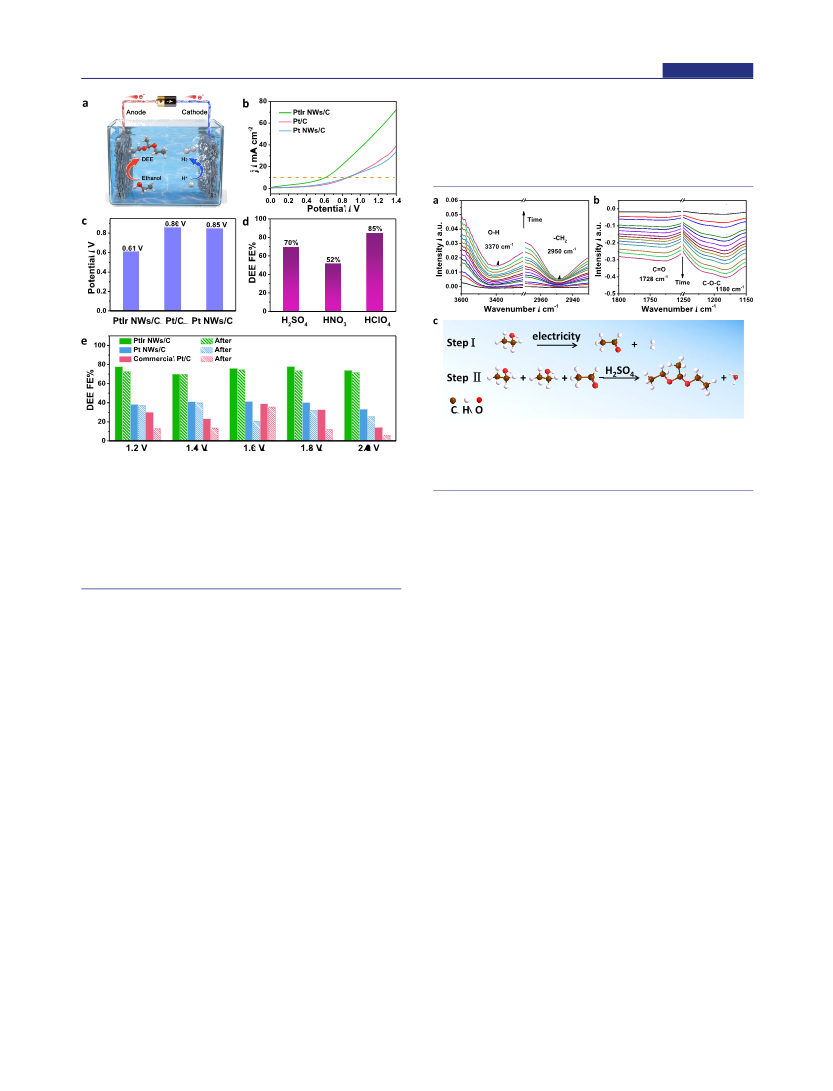
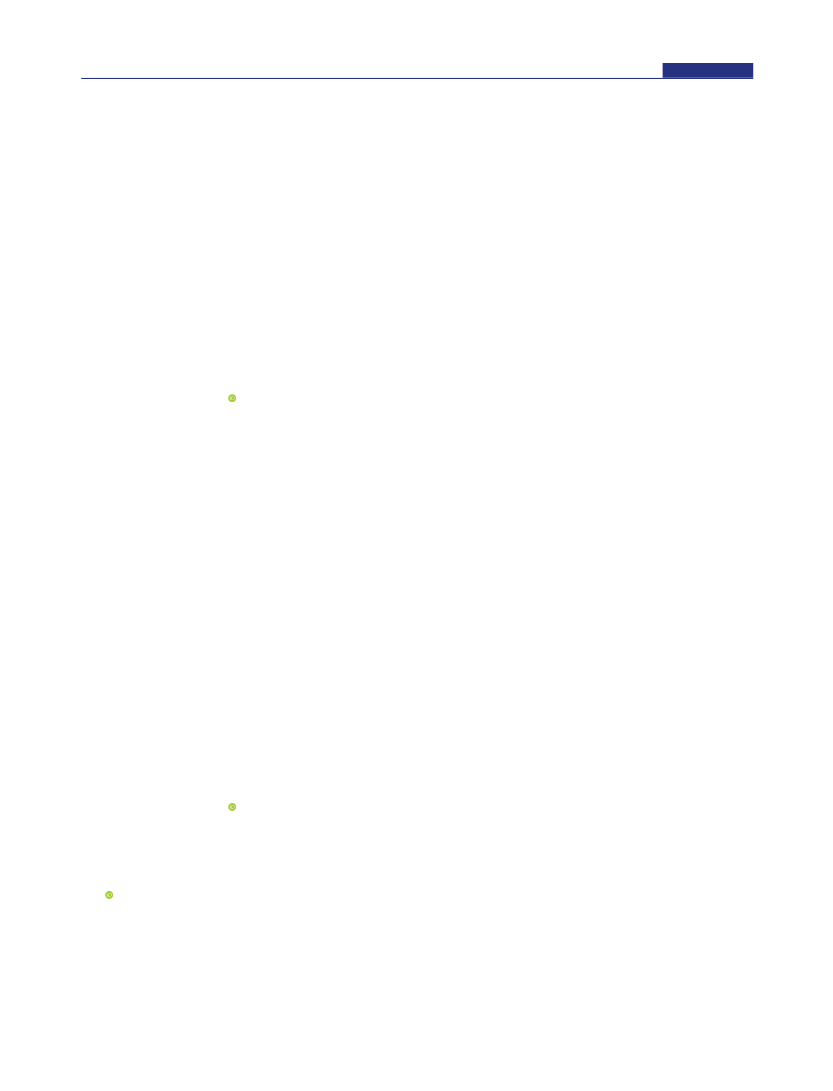
The electrosynthesis of high-value-added multicarbon compounds coupled with hydrogen production is an efficient way to achieve carbon neutrality; however, the lack of effective bifunctional catalysts in electrosynthesis largely hinders its development. Herein, we report the first example on the highly efficient electrosynthesis of high-value-added 1,1-diethoxyethane (DEE) at the anode and high-purity hydrogen at the cathode using 1 nm PtIr nanowires (NWs) as the bifunctional catalysts. We demonstrate that the cell using 1 nm PtIr nanowires as the bifunctional catalysts can achieve a reported lowest voltage of 0.61 V to reach the current density of 10 mA cm-2, much lower than those of the Pt NWs (0.85 V) and commercial Pt/C (0.86 V), and also can have the highest Faraday efficiencies of 85% for DEE production and 94.0% for hydrogen evolution in all the reported electrosynthesis catalysts. The in situ infrared spectroscopy study reveals that PtIr NWs can facilitate the activation of O-H and C-H bonds in ethanol, which is important for the formation of acetaldehyde intermediate, and finally DEE. In addition, the cell using PtIr NWs as bifunctional catalysts exhibits excellent stability by showing almost no obvious decrease in the Faraday efficiency of the DEE production.
View More


Shanghai Yudiao Chemistry Technology Co.,Ltd
Contact:0086-18964703211
Address:Building NO.5, NO.218,Rongtian Road,ganxiang town,Jinshan District,shanghai,201518,china
Contact:(1) 206-3550089
Address:5115 NE 8TH PL, Renton, WA 98059 USA
website:http://www.simagchem.com
Contact:+86-592-2680277
Address:21/F Hualong Office Building,No.6 Hubin East Road, Xiamen,China
Dalian RSD International Trade Co.,Ltd.
Contact:86-22-60875058 58610575
Address:Wantong International Areas, Hongqiao District, Tianjin, China.China
Contact:+86-574- 87178138; 87297407
Address:No. 809, Liudingxingzuo, cangsong road, Ningbo, China
Doi:10.1016/j.jcat.2006.11.027
(2007)Doi:10.1016/0039-128X(67)90119-5
(1967)Doi:10.1007/BF00497320
()Doi:10.1021/ja00377a023
(1982)Doi:10.1016/0008-6215(87)80108-8
(1987)Doi:10.1021/j100265a034
(1985)