96
M. Honda et al. / Journal of Catalysis 318 (2014) 95–107
2.1% under the conditions of dibutyltin dimethoxide catalyst, CO2
300 atm, 453 K, 24 h [1d]) and far from satisfactory due to the
equilibrium limitation of Eq. (1) [20–23]. In order to improve the
low DMC yield, several attempts have been made for removal of
co-produced water from the reaction media, shifting the chemical
equilibrium to the carbonate side [20a–f,21a–c,24,25]. In particu-
lar, it has been reported that effective dehydrating reagents for
removal of water were acetal, carbodiimide and epoxide, affording
high yields based on the dehydrating reagents. For examples,
Bu2Sn(OMe)2/2,2-dimethoxy propane (DMP) [26], CuCl/dicyclohexyl
carbodiimide (DCC), [20d] and ZrO2–KCl–Mg/butylene oxide [21a]
systems provided 88%, 83%, and 49% DMC yields based on the
dehydrating reagents, respectively. However, DMC yields based
on methanol were very low (2%, 6%, 7%, respectively). From the
viewpoint of DMC purification, complete conversion of methanol,
that means high methanol-based DMC yields, is highly desirable
since methanol and DMC form an azeotrope [27]. However, DMC
yields based on methanol in the literatures were below 50% so
far [1b]. Recently, we have found that the combination of
CeO2-catalyzed carboxylation of methanol and CO2 and hydration
of 2-cyanopyridine (CeO2 + 2-cyanopyridine system) drastically
improves the DMC yield up to 94%, where the DMC yield in the
absence of 2-cyanopyridine is only 0.7% [24a]. In this reaction
system, hydration of 2-cyanopyridine gives 2-picolinamide, which
is also catalyzed by CeO2 (Eq. (2)) [28].
specific reaction time, the reactor was cooled to room temperature
and the gas was collected. Ethanol (30 ml, Wako Pure Chemical
Industries, 99.5% min.) and 1-hexanol (0.2 ml, Tokyo Chemical
Industry Co., Ltd., 98.0% min.) were added to the liquid phase as
a solvent and an internal standard substance for a quantitative
analysis, respectively. Products in the liquid and gas phases
were analyzed by using a gas chromatograph equipped with FID
(Shimadzu GC-2014) and GC–MS (Shimadzu QP-2020) with a
CP-Sil5 capillary column (length 50 m, i.d. 0.25 mm, film thickness
0.25 lm).
For the synthesis of various carbonates from CO2 and the corre-
sponding alcohols, the procedures are the same as the case of the
reaction of CO2 + methanol with 2-cyanopyridine. After the reac-
tion time, 30 ml of ethanol or acetone (Wako Pure Chemical Indus-
tries, 99.5% min.) was added to the liquid phase as a solvent, and
0.2 ml of 1-hexanol or 1-propanol (Wako Pure Chemical Industries,
99.5% min.) was added to the liquid phase as an internal standard
substance for a quantitative analysis. The products in the liquid
and gas phases were analyzed by gas chromatography equipped
with an FID or quadrupole mass spectrometer (GC–MS) using a
CP-Sil5 capillary column (length 50 m, i.d. 0.25 mm, film thickness
0.25
lm) or TC-WAX capillary column (length 30 m, i.d. 0.25 mm,
film thickness 0.25
lm).
2.3. Catalyst characterization and DFT calculation
O
C
The surface area of CeO2 was measured with BET method (N2
adsorption) using Gemini (Micromeritics). X-ray diffraction (XRD)
patterns were recorded by Rigaku Ultima IV with Cu Ka (40 kV,
40 mA) radiation. The thermogravimetric analysis (TG) and differ-
ential thermal analysis (DTA) data were collected under air using a
Shimadzu DTG-60. A sample with the weight of 10 mg was charged
into a Pt pan, and subsequently heated from room temperature to
873 K at a heating rate of 10 K/min.
N
CN
N
ð2Þ
NH2
+ H2O
CeO2
Toward the expansion of this catalyst system, we continued
fundamental studies on the applicability and the reaction mecha-
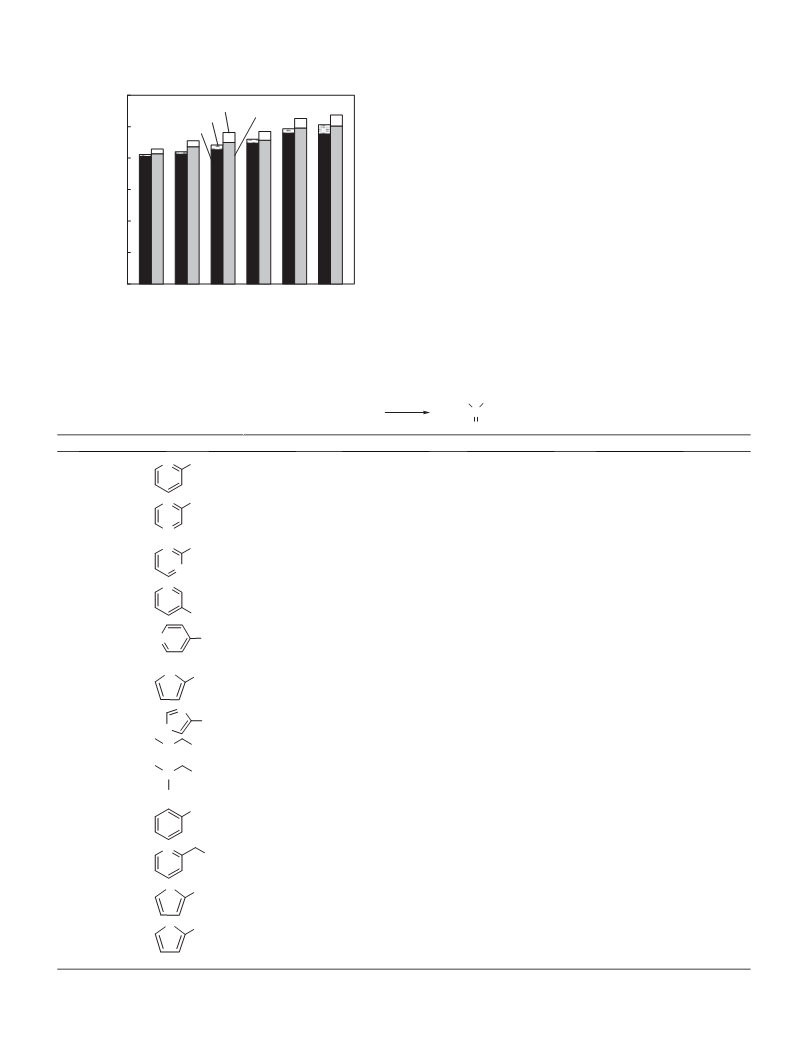
nism. This paper, thus, reports that various carbonates can be syn-
thesized from CO2, and the corresponding alcohols in the presence
of CeO2 and 2-cyanopyridine since the syntheses of carbonates
except for DMC have hardly been reported and the yields based
on alcohols are far from satisfactory (Table S1). In addition, the
reaction mechanism is proposed on the basis of kinetic and isoto-
pic studies, physicochemical analyses, and DFT calculation.
DFT calculations of acidity and basicity were performed at
B3LYP/6-311++G(d,p) level using Gaussian 03 program package.
2.4. Details of the reagents used in experiments
The details of the reactants are described below. All the
chemicals were used without further purification. CO2 (>99.5%)
was purchased from Shimakyu Co. Ltd. Methanol (99.8% min.),
ethanol (99.5% min.), 1-propanol (99.5% min.), 1-butanol (99.0%
min.), 1-pentanol (98.0% min.), 1-octanol (98.0% min.), 1-decanol
(95.0% min.), allyl alcohol (99.0% min.), benzyl alcohol (99.0%
min.), 2-propanol (99.5% min.), acetonitrile (99.8% min.), acetamide
(98.0% min.), 3-cyanopyridine (98.0% min.), 4-cyanopyridine (98.0%
min.), pyridine (99.5% min.), piperidine (98.0% min.), benzoic acid
(99.5% min.), benzonitrile (98% min.), benzamide (95% min.), and
2-furancarbonitrile (98.0% min.) were purchased from Wako Pure
Chemical Industries. 2-Cyanopyridine (99.0% min.), 1-hexanol
(98.0% min.), b-methallyl alcohol (98.0% min.), 2-methyl-1-
propanol (99.0% min.), cyclohexanemethanol (98.0% min.),
cyanopyrazine (97% min.), 2-pyrimidinecarbonitrile (98.0% min.),
methylaminoacetonitrile (98% min.), (dimethylamino) acetonitrile
(95% min.), 2-picolinamide (98.0% min.), and picolinic acid (99.0%
min.) were purchased from Tokyo Chemical Industry Co., Ltd.
Pyrrole-2-carbonitrile (96% min.) was purchased from Aldrich.
2-Pyridineacetonitrile (99% min.) and thiophene-2-carbonitrile
(98% min.) were purchased from Alfa Aesar. 1H-Imidazole-4-
carbonitrile was purchased from Tyger Scientific Inc.
2. Experimental
2.1. Catalyst preparation
Preparation of CeO2 catalyst was carried out by calcining com-
mercially available cerium oxide HS (Daiichi Kigenso Kogyo Co.
Ltd., Japan) for 3 h under air atmosphere at 873 K. The calcination
temperature was optimized on the basis of the catalytic perfor-
mance in the reaction of methanol + CO2 with 2-cyanopyridine
[24a]. The BET surface area of calcined CeO2 was 84 m2 gꢀ1
.
2.2. Activity tests
All the reactions were carried out in an autoclave reactor with
an inner volume of 190 ml. A typical procedure of the reaction of
CO2 + methanol with 2-cyanopyridine was as follows: 0.34 g of
CeO2 catalyst, 0.64 g of methanol (20 mmol, Wako Pure Chemical
Industries, 99.8% min.), and 10.4 g of 2-cyanopyridine (100 mmol,
Tokyo Chemical Industry Co., Ltd., 99.0% min.) were put into the
autoclave together with a spinner, and then, the reactor was
purged and pressurized with CO2 (Shimakyu Co. Ltd., >99.5%).
Gas line was closed, and then, the reactor was heated to the reac-
tion temperature. The time when the temperature reached the
desired reaction temperature is defined as zero reaction time.
The mixture was constantly stirred during the reaction. After the
In order to determine the retention time and molar sensitivity,
commercially available reagents were used; dimethyl carbonate
(98.0% min.), diethyl carbonate (98.0% min.), methyl carba-
mate (98.0% min.), ethyl carbamate (98.0% min.), butyl carbamate
(98.0% min.), benzyl carbamate (98.0% min.), methyl picolinate

 Honda, Masayoshi
Honda, Masayoshi
 Tamura, Masazumi
Tamura, Masazumi
 Nakagawa, Yoshinao
Nakagawa, Yoshinao
 Nakao, Kenji
Nakao, Kenji
 Suzuki, Kimihito
Suzuki, Kimihito
 Tomishige, Keiichi
Tomishige, Keiichi