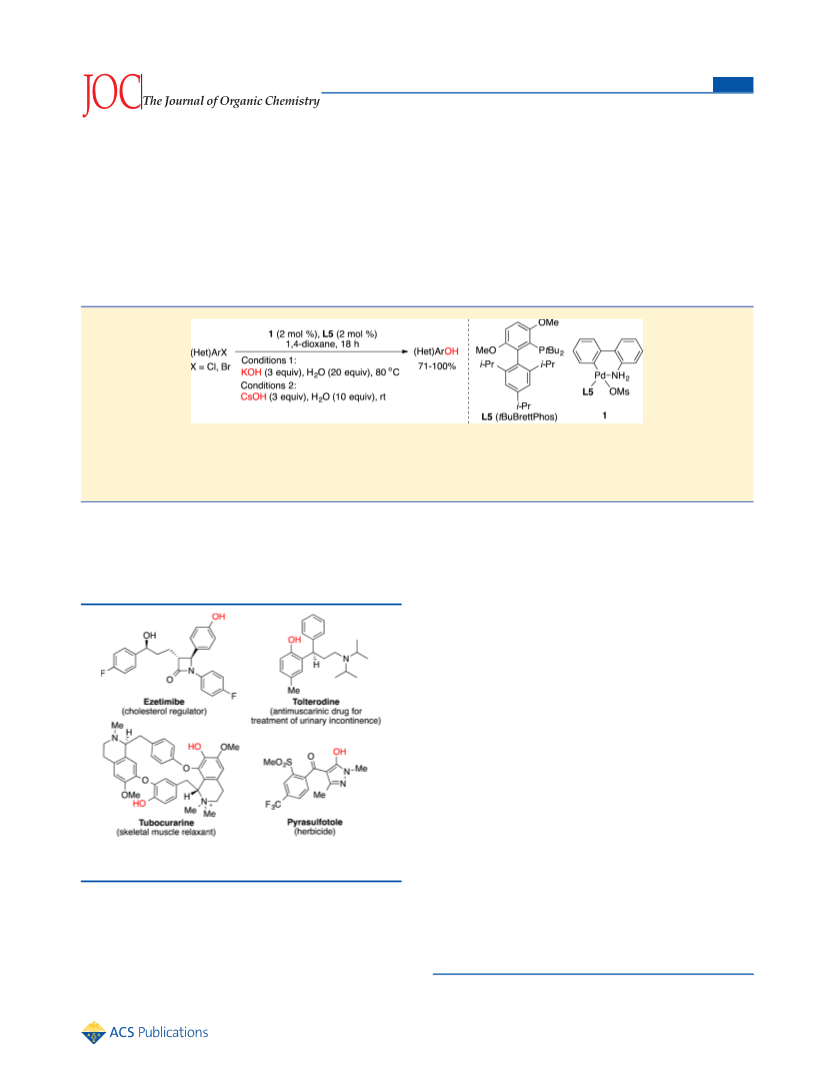
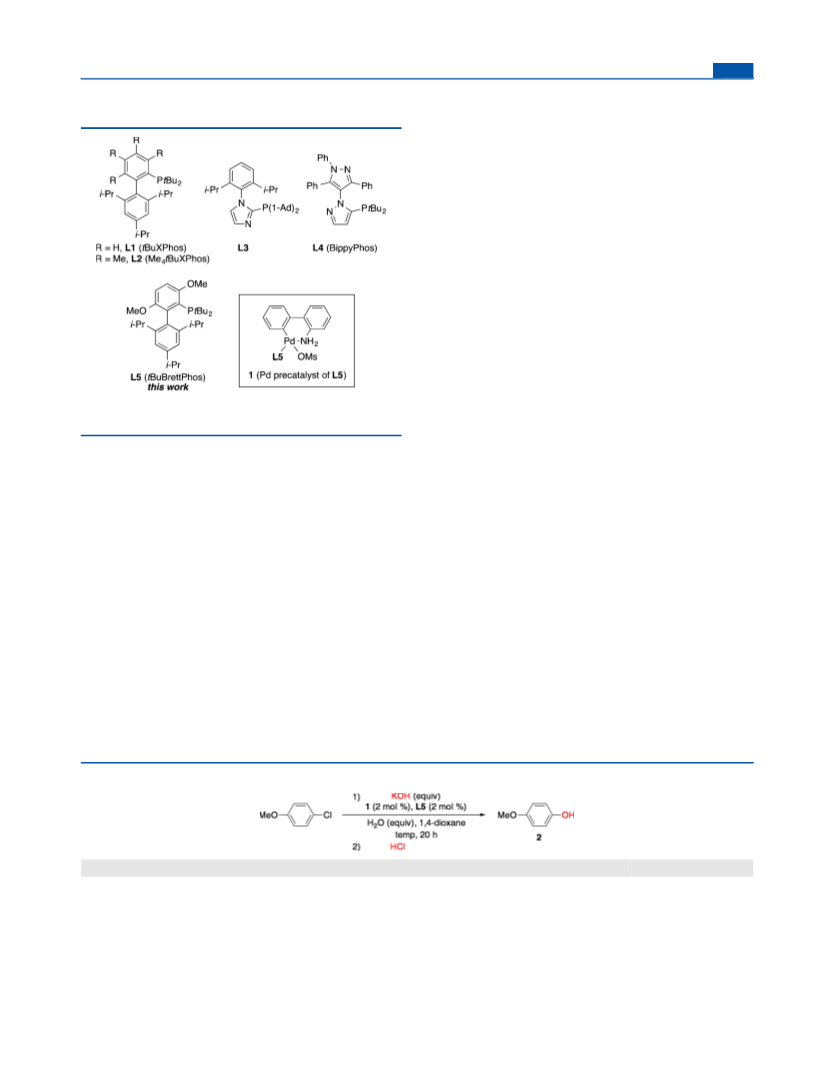
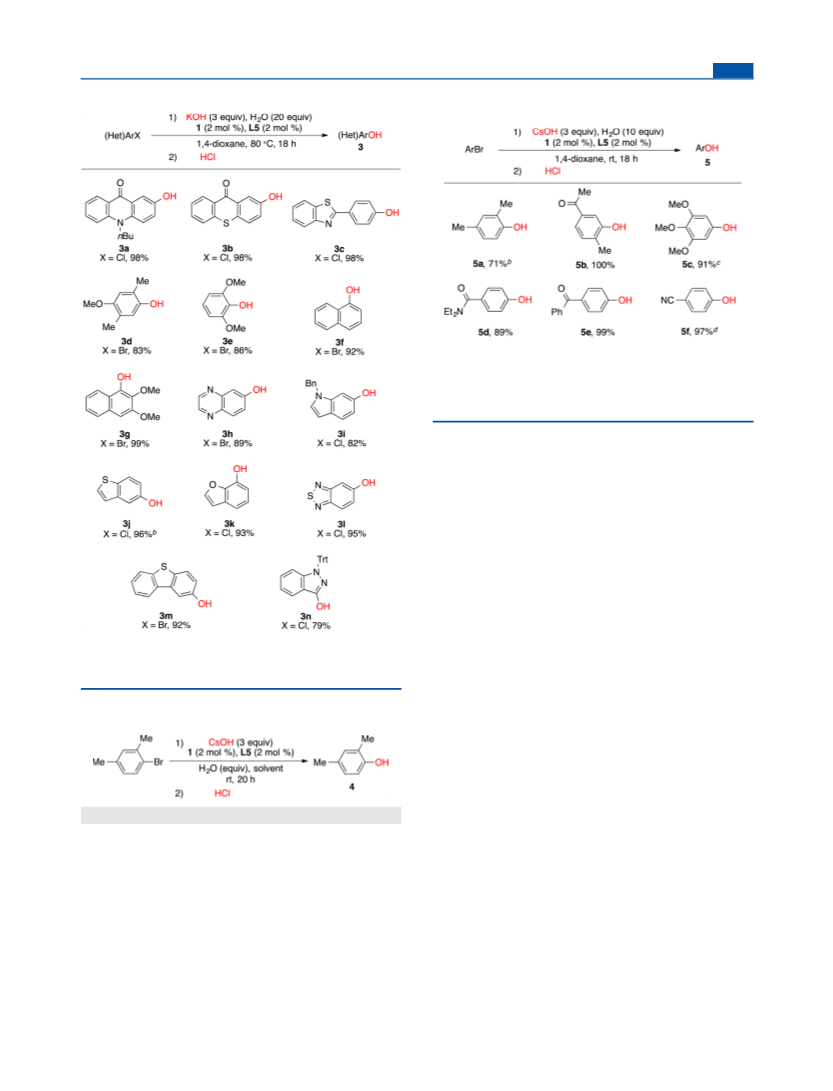
The Journal of Organic Chemistry
Note
Hz, J = 1.2 Hz, 1 H), 7.86 (d, J = 2.8 Hz, 1 H), 7.79 (dd, J = 8.0 Hz, J
= 0.8 Hz, 1 H), 7.73 (ddd, J = 8.4 Hz, J = 6.8 Hz, J = 1.6 Hz, 1 H),
7.67 (d, J = 8.8 Hz, 1 H), 7.54 (ddd, J = 8.4 Hz, J = 7.2 Hz, J = 1.2 Hz,
1 H), 7.27 (dd, J = 8.8 Hz, J = 2.8 Hz, 1 H). 13C NMR (100 MHz,
DMSO-d6) δ: 178.6, 156.5, 137.0, 132.6, 129.6, 129.0, 128.0, 127.7,
126.5, 126.3, 126.1, 122.7, 113.2. Anal. Calcd for C13H8O2S: C, 68.40;
H, 3.53. Found: C, 68.14; H, 3.55. IR (neat cm−1) 3303, 1620, 1568,
1484, 1436, 1341, 1216, 1150, 1121, 1075, 814, 736, 633.
144.0, 142.2, 137.6, 130.4, 122.9, 109.5. Anal. Calcd for C8H6N2O: C,
65.75; H, 4.14. Found: C, 65.63; H, 4.12. HRMS (ESI) Calcd for
C8H7N2O [M + H]: 147.0553. Found: 147.0548. IR (neat cm−1)
1610, 1478, 1409, 1304, 1231, 1197, 1117, 1036, 941, 831, 750.
1-Benzyl-1H-indol-6-ol (3i). Following general procedure A, the
title compound was prepared using 1-benzyl-6-chloro-1H-indole (241
mg, 1.0 mmol); EtOAc/hexanes (1:4); 182.1 mg, 82%; pale-brown
solid. 1H NMR (400 MHz, DMSO-d6) δ: 8.99 (br s, 1 H), 7.35 (d, J =
8.4 Hz, 1 H), 7.30 (t, J = 7.2 Hz, 2 H), 7.26 (d, J = 3.2 Hz, 1 H), 7.24
(t, J = 7.2 Hz, 1 H), 7.15 (d, J = Hz, 2 H), 6.73 (d, J = 2.0 Hz, 1 H),
6.60 (dd, J = 8.4 Hz, J = 2.0 Hz, 1 H), 6.36 (dd, J = 3.2 Hz, J = 0.4 Hz,
1 H), 5.29 (s, 2 H). 13C NMR (100 MHz, DMSO-d6) δ: 153.2, 138.4,
136.9, 128.5, 127.3, 127.2, 126.8, 121.6, 120.9, 109.7, 100.8, 95.4, 49.1.
m.p.: 103−104 °C. HRMS (ESI) Calcd for C15H12NO [M − H]:
222.0924. Found: 222.0941. IR (neat cm−1) 3308, 1625, 1580, 1507,
1468, 1354, 1321, 1267, 1168, 1074, 943, 827, 799, 745, 705.
Benzothiophen-5-ol (3j).27 Following general procedure A, the
title compound was prepared using 5-chlorobenzothiophene (169 mg,
1.0 mmol) at 100 °C; EtOAc/hexanes (1:6); 143.6 mg, 96%; off-white
solid. 1H NMR (400 MHz, DMSO-d6) δ: 9.42 (br s, 1 H), 7.74 (d, J =
8.8 Hz, 1 H), 7.65 (d, J = 5.6 Hz, 1 H), 7.27 (dd, J = 5.6 Hz, J = 0.4
Hz, 1 H), 7.21 (d, J = 2.0 Hz, 1 H), 6.88 (ddd, J = 8.4 Hz, J = 2.0 Hz, J
= 0.4 Hz, 1 H). 13C NMR (100 MHz, DMSO-d6) δ: 154.9, 140.9,
129.4, 128.0, 123.5, 123.1, 114.8, 108.2. m.p.: 105−106 °C (lit: 103−
105 °C).28 HRMS (ESI) Calcd for C8H6OS [M]: 150.0134. Found:
150.0133. IR (neat cm−1) 3235, 1599, 1565, 1502, 1422, 1231, 1149,
945, 889, 849, 748, 692.
4-(Benzothiazol-2-yl)phenol (3c).23 Following general proce-
dure A, the title compound was prepared using 2-(4-chlorophenyl)-
benzothiazole (246 mg, 1.0 mmol); EtOAc/hexanes (1:6), then (1:4);
223 mg, 98%; pale-yellow solid. m.p.: 227−229 °C (lit: 228−229
°C).23 1H NMR (400 MHz, DMSO-d6) δ: 10.23 (br s, 1 H), 8.05 (dd,
J = 7.6 Hz, 1 H), 7.98 (dd, J = 8.0 Hz, 1 H), 7.93 (d, J = 8.4 Hz, 2 H),
7.49 (td, J = 8.0 Hz, J = 1.2 Hz, 1 H), 7.39 (td, J = 8.0 Hz, J = 1.2 Hz, 1
H), 6.94 (d, J = 8.8 Hz, 2 H). 13C NMR (100 MHz, DMSO-d6) δ:
167.5, 160.6, 153.7, 134.1, 129.1, 126.4, 124.9, 124.1, 122.3, 122.1,
116.1. Anal. Calcd for C13H9NOS: C, 68.70; H, 3.99. Found: C, 68.83
H, 4.13. IR (neat cm−1) 1604, 1480, 1429, 1283, 1224, 1167, 977, 826,
756, 723.
4-Methoxy-2,5-dimethylphenol (3d).24 Following general
procedure A, the title compound was prepared using 1-bromo-4-
methoxy-2,5-dimethylbenzene (215.1 mg, 1.0 mmol); EtOAc/hexanes
(1:6); 127 mg, 83%; white solid. m.p.: 89−90 °C (lit: 86−88 °C).24
1H NMR (400 MHz, DMSO-d6) δ: 8.58 (br s, 1 H), 6.62 (s, 1 H),
6.55 (s, 1 H), 3.66 (s, 3 H), 2.08 (s, 3 H), 2.03 (s, 3 H). 13C NMR
(100 MHz, DMSO-d6) δ: 150.0, 148.5, 123.1, 121.1, 117.1, 113.1,
55.6, 16.0, 15.7. HRMS (ESI) Calcd for C9H13O2 [M + H]: 153.0910.
Found: 153.0915. IR (neat cm−1) 3238, 1519, 1452, 1412, 1198, 1103,
1018, 864, 834, 671.
Benzofuran-7-ol (3k).29 Following general procedure A, the title
compound was prepared using 7-chlorobenzofuran (153 mg, 1.0
mmol); EtOAc/hexanes (1:4); 125 mg, 93%; pale-yellow oil. 1H NMR
(400 MHz, DMSO-d6) δ: 9.96 (br s, 1 H), 7.91 (d, J = 2.0 Hz, 1 H),
7.06 (d, J = 7.6 Hz, 1 H), 7.02 (t, J = 7.6 Hz, 1 H), 6.87 (d, J = 2.0 Hz,
1 H), 6.74 (d, J = 7.6 Hz, 1 H). 13C NMR (100 MHz, DMSO-d6) δ:
145.4, 143.4, 142.8, 129.1, 123.5, 111.6, 110.3, 107.0. HRMS (ESI)
Calcd for C8H7O2 [M + H]: 135.0441. Found: 135.0440. IR (neat
cm−1) 3323, 1593, 1443, 1297, 1217, 1122, 841.
Benzo-2,1,3-thiadiazol-5-ol (3l). Following general procedure A,
the title compound was prepared using 5-chlorobenzo-2-1-3-
thiadiazole (171 mg, 1.0 mmol); EtOAc/hexanes (1:6); 145 mg,
95%; pale-yellow solid. 1H NMR (400 MHz, DMSO-d6) δ: 10.55 (br s,
1 H), 7.90 (d, J = 9.2 Hz, 1 H), 7.33 (dd, J = 9.2 Hz, J = 2.4 Hz, 1 H),
7.15 (dd, J = 2.4 Hz, J = 0.4 Hz, 1 H). 13C NMR (100 MHz, DMSO-
d6) δ: 159.3, 156.0, 150.1, 125.2, 121.5, 100.4. m.p.: 161−162 °C.
HRMS (ESI) Calcd for C6H5N2OS [M + H]: 153.0117. Found:
153.0114. IR (neat cm−1) 3133, 1613, 1495, 1403, 1226, 1187, 1127,
874, 833, 814, 753, 664, 626.
2,6-Dimethoxyphenol (3e).8e Following general procedure A,
the title compound was prepared using 2-bromo-1,3-dimethoxyben-
zene (217 mg, 1.0 mmol); EtOAc/hexanes (1:6), then (1:4); 134 mg,
86%; pale-brown solid. m.p.: 56−57 °C (lit: 54−56 °C).25 1H NMR
(400 MHz, DMSO-d6) δ: 8.25 (br s, 1 H), 6.69 (t, J = 8.4 Hz, 1 H),
6.59 (d, J = 8.4 Hz, 2 H), 3.74 (s, 6 H). 13C NMR (100 MHz, DMSO-
d6) δ: 148.2, 135.7, 118.1, 105.7, 55.9. HRMS (ESI) Calcd for
C8H11O3 [M + H]: 155.0703. Found: 155.0706. IR (neat cm−1) 3452,
1612, 1508, 1478, 1438, 1360, 1279, 1210, 1097, 1027, 825, 767, 726,
706, 609.
Naphthalen-1-ol (3f).8c Following general procedure A, the title
compound was prepared using 1-bromonaphthalene (207 mg, 1.0
mmol); EtOAc/hexanes (1:6); 133 mg, 92%; pale-brown solid. m.p.:
95 °C (lit: 91−93 °C).8c 1H NMR (400 MHz, DMSO-d6) δ: 10.10 (br
s, 1 H), 8.14 (dd, J = 7.2 Hz, J = 1.2 Hz, 1 H), 7.80 (dd, J = 7.2 Hz, J =
1.6 Hz, 1 H), 7.48−7.41 (ovrlp, 2 H), 7.34−7.28 (ovrlp, 2 H), 6.88
(dd, J = 7.2 Hz, J = 1.6 Hz, 1 H). 13C NMR (100 MHz, DMSO-d6) δ:
153.2, 134.4, 127.4, 126.4, 126.1, 124.6, 124.5, 122.0, 118.3, 108.0.
HRMS (ESI) Calcd for C10H9O [M + H]: 145.0648. Found:
145.0646. IR (neat cm−1) 3277, 1598, 1457, 1385, 1268, 1148, 1083,
1043, 1015, 876, 860, 789, 764, 710.
Dibenzothiophen-2-ol (3m).30 Following general procedure A,
the title compound was prepared using 2-bromodibenzothiophene
(263 mg, 1.0 mmol). After work up, the crude product was purified by
flash chromatography using EtOAc/hexanes (1:6) as an eluent to
afford dibenzothiophen-2-ol (3m) (185 mg, 92%) as a pale-brown
solid. 1H NMR (400 MHz, DMSO-d6) δ: 9.68 (br s, 1 H), 8.23−8.18
(m, 1 H), 7.97−7.91 (m, 1 H), 7.78 (d, J = 8.8 Hz, 1 H), 7.68 (d, J =
2.4 Hz, 1 H), 7.49−7.42 (ovrlp, 2 H), 7.03 (dd, J = 8.4 Hz, J = 2.4 Hz,
1 H). 13C NMR (100 MHz, DMSO-d6) δ: 155.4, 139.7, 136.3, 135.0,
128.5, 126.8, 124.4, 123.6, 123.0, 121.9, 116.7, 107.4. m.p.: 156−157
°C (lit: 158 °C).31 Anal. Calcd for C12H8OS: C, 71.97; H, 4.03.
Found: C, 71.73; H, 4.04. IR (neat cm−1) 3385, 1602, 1470, 1427,
1336, 1187, 849, 810, 755, 733.
1-Trityl-1H-indazol-3-ol (3n). Following general procedure A, the
title compound was prepared using 3-chloro-1-trityl-1H-indazole (S1)
(395 mg, 1.0 mmol); EtOAc/hexanes (1:6); 297 mg, 0.79 mmol, 79%;
pale-brown solid. m.p.: 196−197 °C. 1H NMR (400 MHz, DMSO-d6)
δ: 10.98 (s, 1 H), 7.58 (d, J = 8.0 Hz, 1 H), 7.36 (d, J = 7.2 Hz, 6 H),
7.28 (t, J = 7.2 Hz, 6 H), 7.22 (t, J = 7.2 Hz, 3 H), 7.00 (t, J = 7.2 Hz, 1
H), 6.93 (t, J = 7.2 Hz, 1 H), 6.40 (d, J = 8.4 Hz, 1 H). 13C NMR (100
MHz, DMSO-d6) δ: 144.9, 133.2, 132.7, 119.2, 117.6, 116.9, 116.3,
110.1, 109.5, 105.8, 103.6, 67.0. HRMS (ESI) Calcd for C26H19N2O
[M − H]: 375.1503. Found: 375.1507. IR (neat cm−1) 1671, 1489,
1450, 1217, 1001, 906, 746, 705, 632.
2,3-Dimethoxynaphthalen-1-ol (3g). Following general proce-
dure A, the title compound was prepared using 1-bromo-2,3-
dimethoxynaphthalene (267 mg, 1.0 mmol); EtOAc/hexanes (1:6);
202 mg, 99%; yellow oil. 1H NMR (400 MHz, DMSO-d6) δ: 9.53 (br
s, 1 H), 8.01 (d, J = 8.4 Hz, 1 H), 7.69 (d, J = 8.0 Hz, 1 H), 7.35 (ddd,
J = 8.0 Hz, J = 6.8 Hz, J = 1.2 Hz, 1 H), 7.28 (ddd, J = 8.4 Hz, J = 6.8
Hz, J = 1.2 Hz, 1 H), 6.87 (s, 1 H), 3.89 (s, 3 H), 3.77 (s, 3 H). 13C
NMR (100 MHz, DMSO-d6) δ: 152.7, 144.6, 133.8, 130.7, 126.3,
125.5, 122.5, 121.6, 121.1, 98.2, 60.5, 55.5. HRMS (ESI) Calcd for
C12H13O3 [M + H]: 205.0859. Found: 205.0852. IR (neat cm−1) 3500,
1739, 1635, 1600, 1509, 1476, 1295, 1265, 1231, 1195, 1120, 1095,
1032, 1015, 975, 859, 812, 735, 700.
Quinoxalin-6-ol (3h).15f Following general procedure A, the title
compound was prepared using 6-bromoquinoxaline (209 mg, 1.0
mmol); EtOAc/MeOH (20:1); 130.5 mg, 89%; brown solid. m.p.:
251−253 °C (lit: 252−254 °C).26 1H NMR (400 MHz, DMSO-d6) δ:
10.53 (br s, 1 H), 8.77 (d, J = 2.0 Hz, 1 H), 8.68 (d, J = 1.6 Hz, 1 H),
7.93 (d, J = 9.2 Hz, 1 H), 7.41 (dd, J = 9.2 Hz, J = 2.8 Hz, 1 H), 7.27
(d, J = 2.8 Hz, 1 H). 13C NMR (100 MHz, DMSO-d6) δ: 158.8, 145.4,
5355
dx.doi.org/10.1021/jo500662s | J. Org. Chem. 2014, 79, 5351−5358

 Cheung, Chi Wai
Cheung, Chi Wai
 Buchwald, Stephen L.
Buchwald, Stephen L.