
Journal of Molecular Liquids p. 153 - 160 (2017)
Update date:2022-08-11
Topics:
 Fareghi-Alamdari, Reza
Fareghi-Alamdari, Reza
 Nadiri Niri, Mehri
Nadiri Niri, Mehri
 Hazarkhani, Hassan
Hazarkhani, Hassan
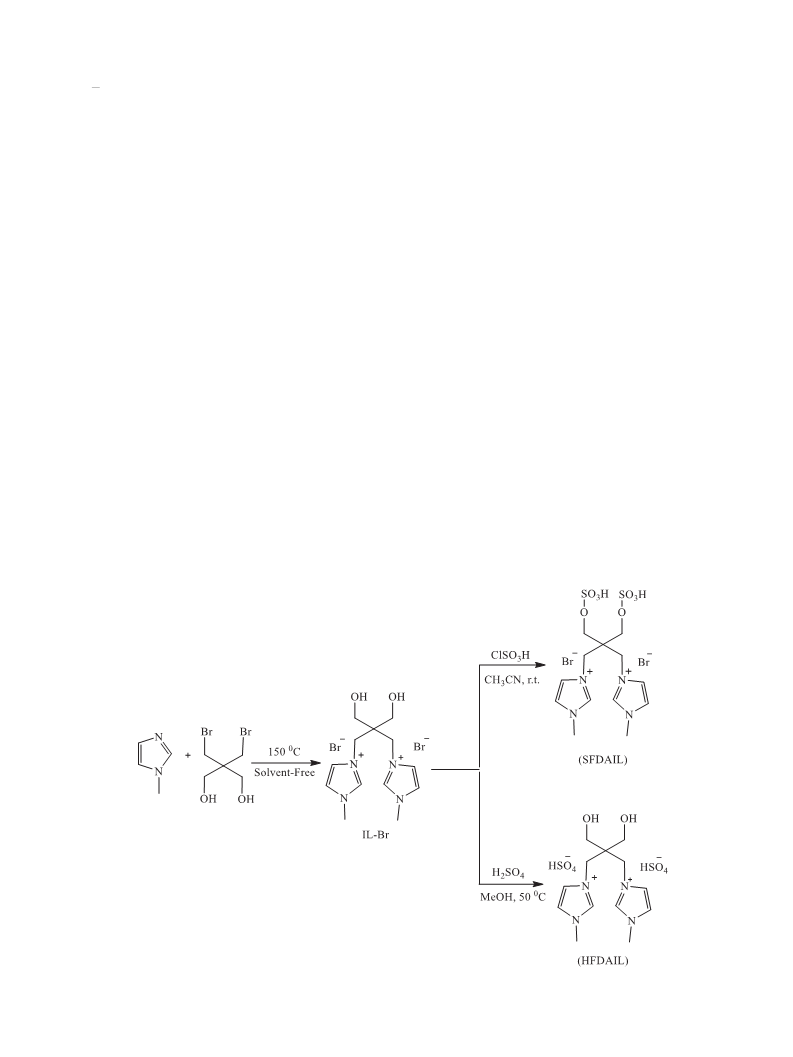
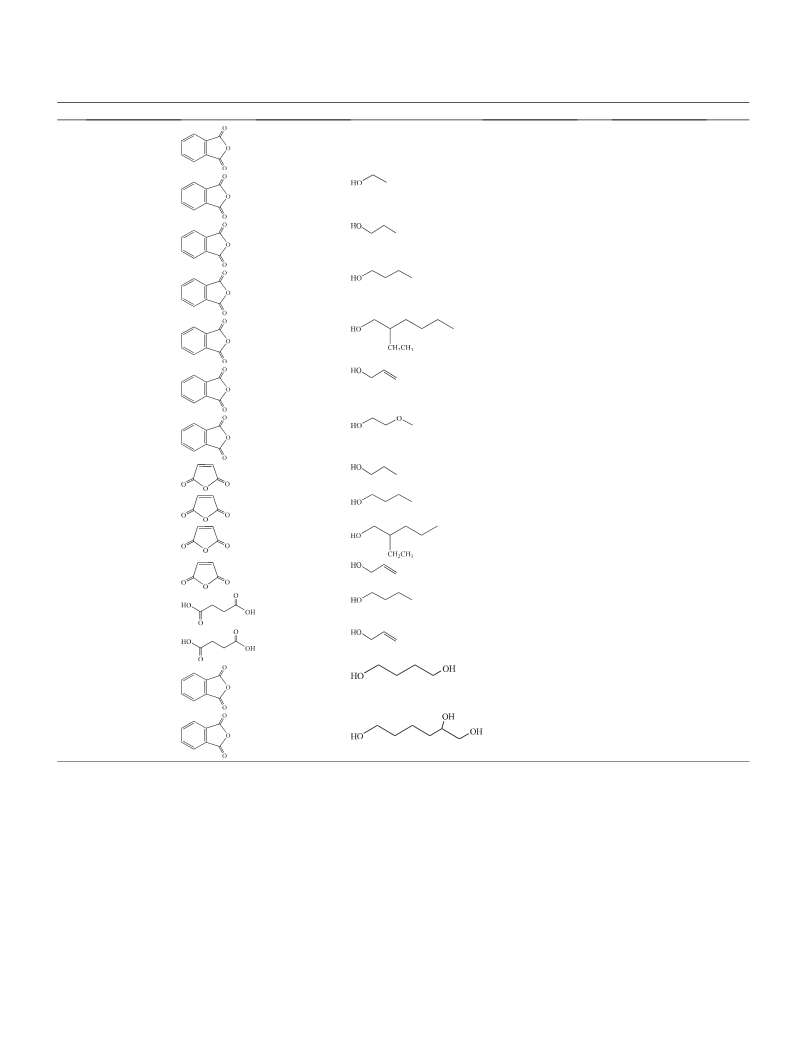
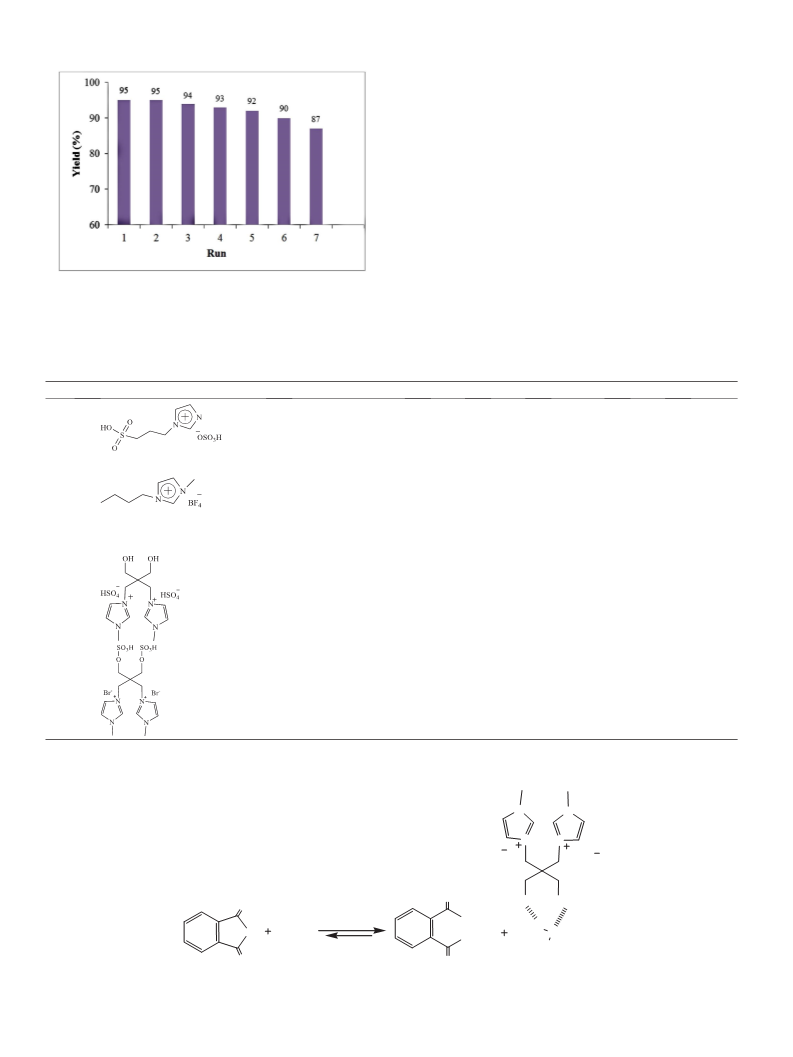
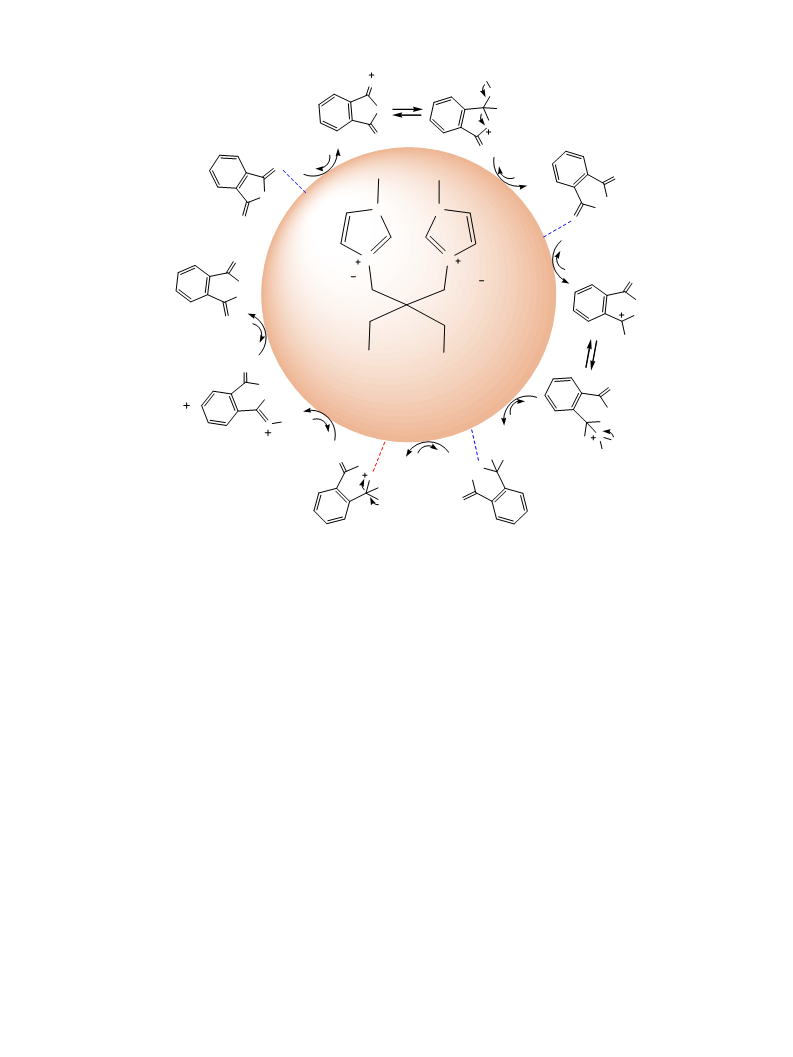
Two new functionalized diacidic ionic liquids (FDAILs) including hydroxyl functionalized diacidic ionic liquid (HFDAIL) and sulfonated diacidic ionic liquid (SFDAIL) were synthesized and characterized by 1HNMR, 13CNMR and FT-IR. The catalytic activities of these FDAILs were examined in esterification reaction of anhydrides with some alcohols to give corresponding dialkyl plasticizers under solvent-free conditions. The results indicate that HFDAIL, as hydroxyl-bearing catalyst, show better catalytic performance. Under the optimum conditions, using HFDAIL, the conversion of phthalic anhydride was high and diester plasticizers were obtained with good to excellent yields in the presence of only 10?mol% of ionic liquid. All the produced diesters could be easily recovered due to their immiscibility with the ionic liquid. Recycling experiments suggests that these ionic liquids can be reused several times without remarkable loss in their catalytic activity.
View More


Contact:+852 83038667
Address:Room 1502, 15th Floor, SPA Centre,53-55 Lockhart Road, Wanchai, Hong Kong
Contact:+86 21 34123252
Address:14, 4580 Dushi, Shanghai, China
Contact:0086 533 2282832
Address:Zibo,Shandong
Contact:+86-574- 87178138; 87297407
Address:No. 809, Liudingxingzuo, cangsong road, Ningbo, China
Junkai (Tianjin) Chemical Co., Ltd.
website:http://www.junkaichem.com
Contact:86-22-85689515
Address:Room 8-501, Building K2, The Hi-Tech Green Industrial Base, No.6, 6th Road, Hi-Tech Development Road, Tianjin Hi-Tech IndustrialPark, Tianjin, China.
Doi:10.1021/ja01039a005
(1969)Doi:10.1039/JS8631600051
(1863)Doi:10.1007/BF01912719
(1987)Doi:10.1039/c6ra01364c
(2016)Doi:10.1039/CT8915901012
(1891)Doi:10.1246/cl.1977.1487
(1977)