Journal of the American Chemical Society
Page 4 of 12
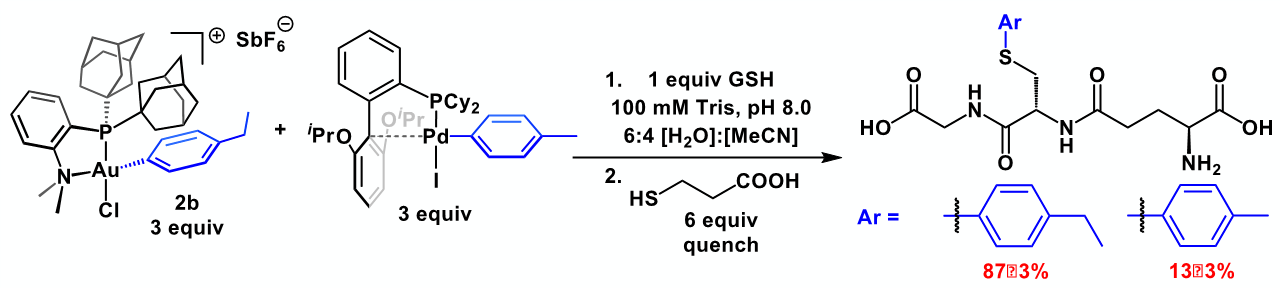
range under mild reaction conditions and displays rapid reaction
2017, 56, 3863–3873.
Vinogradova, E. V. Pure Appl. Chem. 2017, 89, 1619–
1640.
Jbara, M.; Maity, S. K.; Brik, A. Angew. Chem. - Int. Ed.
2017, 56, 10644–10655.
Vara, B. A.; Li, X.; Berritt, S.; Walters, C. R.; Petersson,
E. J.; Molander, G. A. Chem. Sci. 2018, 9, 336–344.
Vinogradova, E. V; Zhang, C.; Spokoyny, A. M.;
Pentelute, B. L.; Buchwald, S. L. Nature 2015, 526, 687–
691.
Rojas, A. J.; Zhang, C.; Vinogradova, E. V.; Buchwald, N.
H.; Reilly, J.; Pentelute, B. L.; Buchwald, S. L. Chem. Sci.
2017, 8, 4257–4263.
1
2
3
4
5
6
7
8
kinetics, high chemoselectivity, and excellent functional group
tolerance. With this work, we expand the scope of biomolecule
modification3,5,38–40 by providing tools that interface bond form-
ing processes characteristic of organometallic complexes18,25,41–
(6)
(7)
(8)
(9)
46
with bioconjugation. The straightforward synthetic proce-
dures and commercially available or otherwise easily accessible
reagents presented should expand the bioconjugation space well
beyond the substrates and peptides reported in this study. This
work also expands on a relatively underrepresented class of or-
ganometallic reagents containing metal-carbon bonds capable
of withstanding relatively harsh environmental conditions.47
9
(10)
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
ASSOCIATED CONTENT
Supporting Information
(11)
(12)
(13)
(14)
(15)
Lee, H. G.; Lautrette, G.; Pentelute, B. L.; Buchwald, S.
L. Angew. Chem. - Int. Ed. 2017, 56, 3177–3181.
Rojas, A. J.; Pentelute, B. L.; Buchwald, S. L. Org. Lett.
2017, 19, 4263–4266.
Kubota, K.; Dai, P.; Pentelute, B. L.; Buchwald, S. L. J.
Am. Chem. Soc. 2018, 140, 3128–3133.
Joost, M.; Amgoune, A.; Bourissou, D. Angew. Chem. -
Int. Ed. 2015, 54, 15022–15045.
Harper, M. J.; Arthur, C. J.; Crosby, J.; Emmett, E. J.;
Falconer, R. L.; Fensham-Smith, A. J.; Gates, P. J.;
Leman, T.; Mcgrady, J. E.; Bower, J. F.; Russell, C. A. J.
Am. Chem. Soc. 2018, 140, 4440–4445.
Livendahl, M.; Goehry, C.; Maseras, F.; Echavarren, A.
M. Chem. Commun. 2014, 50, 1533–1536.
Scott, V. J.; Labinger, J. A.; Bercaw, J. E.
Organometallics 2010, 29, 4090–4096.
Hopkinson, M. N.; Gee, A. D.; Gouverneur, V. Chem. - A
Eur. J. 2011, 17, 8248–8262.
Wegner, H. A.; Auzias, M. Angew. Chem. - Int. Ed. 2011,
50, 8236–8247.
On-Yee Chan, A.; Lui-Lui Tsai, J.; Kar-Yan Lo, V.; Li, G.-
L.; Wong, M.-K.; Che, C.-M. Chem. Commun. 2013, 49,
1428–1430.
Zou, T.; Lum, C. T.; Lok, C.-N.; Zhang, J.-J.; Che, C.-M.
Chem. Soc. Rev. 2015, 44, 8786–8801.
Kung, K. K.-Y.; Ko, H.-M.; Cui, J.-F.; Chong, H.-C.;
Leung, Y.-C.; Wong, M.-K. Chem. Commun. 2014, 50,
11899–11902.
Joost, M.; Zeineddine, A.; Estévez, L.; Mallet-Ladeira,
S.; Miqueu, K.; Amgoune, A.; Bourissou, D. J. Am. Chem.
Soc. 2014, 136, 14654–14657.
Zeineddine, A.; Estévez, L.; Mallet-Ladeira, S.; Miqueu,
K.; Amgoune, A.; Bourissou, D. Nat. Commun. 2017, 8
(1).
Detailed experimental procedures, characterization data, and
crystallographic data are available in the Supporting Infor-
mation. The Supporting Information is available free of charge
on the ACS Publications website.
AUTHOR INFORMATION
Corresponding Author
(16)
(17)
(18)
(19)
(20)
Author Contributions
‡These authors contributed equally.
Notes
The authors declare no competing financial interests.
ACKNOWLEDGMENT
A.M.S. thanks UCLA Department of Chemistry and Biochemistry
for start-up funds, 3M for a Non-Tenured Faculty Award, Alfred
P. Sloan Foundation for a Fellowship in Chemistry and the Na-
tional Institutes of Health (NIH) for a Maximizing Investigators
Research Award (MIRA, R35GM124746). H.D.M thanks the Na-
tional Science Foundation (NSF, CHE-1507735) for funding.
M.S.M thanks the NSF for the Bridge-to-Doctorate (HRD-
1400789) and the Predoctoral (GRFP) (DGE-0707424) Fellow-
ships and UCLA for the Christopher S. Foote Fellowship. Dr.
Jacquelin Kammeyer (UCLA) is acknowledged for helpful discus-
sions and Mr. Nicholas Bernier (UCLA) is thanked for assistance
with ICP-AES measurements. The authors thank Dr. Chi Zhang
and Prof. Bradley L. Pentelute (MIT) for generously sharing the
plasmid used for DARPin expression and the UCLA-DOE Insti-
tute Protein Expression Technology Center (PETC) for expres-
sion of DARPin and Dr. Yu Chen (UCLA) for help with mass spec-
trometry.
(21)
(22)
(23)
(24)
(25)
(26)
(27)
(28)
(29)
(30)
Wolf, W. J.; Winston, M. S.; Toste, F. D. Nat. Chem.
2014, 6, 159–164.
Kang, K.; Liu, S.; Xu, T.; Wang, D.; Leng, X.; Bai, R.; Lan,
Y.; Shen, Q. Organometallics 2017, 36, 4727–4740.
Bachman, R. E.; Bodolosky-Bettis, S. A.; Pyle, C. J.;
Gray, M. A. J. Am. Chem. Soc. 2008, 130, 14303–14310.
Hesp, K. D.; Stradiotto, M. J. Am. Chem. Soc. 2010, 132,
18026–18029.
Lundgren, R. J.; Sappong-Kumankumah, A.; Stradiotto,
M. Chem. - A Eur. J. 2010, 16, 1983–1991.
Winston, M. S.; Wolf, W. J.; Toste, F. D. J. Am. Chem.
Soc. 2015, 137, 7921–7928.
REFERENCES
(1)
(2)
Kalia, J.; Raines, R. T. Curr Org Chem. 2010, 14, 138–147.
Boutureira, O.; Bernardes, G. J. L. Chem. Rev. 2015, 115,
2174–2195.
(3)
(4)
Chalker, J. M.; Bernardes, G. J. L.; Lin, Y. A.; Davis, B. G.
Chem. - An Asian J. 2009, 4, 630–640.
Spokoyny, A. M.; Zou, Y.; Ling, J. J.; Yu, H.; Lin, Y. S.;
Pentelute, B. L. J. Am. Chem. Soc. 2013, 135, 5946–
5949.
(31)
(32)
(33)
Itoh, N.; Ornitz, D. M. J. Biochem. 2011, 149, 121–130.
Yudin, A. K. Chem. Sci. 2015, 6, 30–49.
Kaspar, A. A.; Reichert, J. M. Drug Discov. Today 2013,
18, 807–817.
(5)
DeGruyter, J. N.; Malins, L. R.; Baran, P. S. Biochemistry
ACS Paragon Plus Environment

 Messina, Marco S.
Messina, Marco S.
 Stauber, Julia M.
Stauber, Julia M.
 Waddington, Mary A.
Waddington, Mary A.
 Rheingold, Arnold L.
Rheingold, Arnold L.
 Maynard, Heather D.
Maynard, Heather D.
 Spokoyny, Alexander M.
Spokoyny, Alexander M.